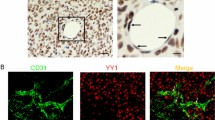
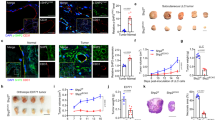
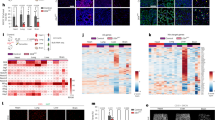
Abstract
Deleted in liver cancer 1 (DLC1) is a RhoGTPase activation protein-containing tumor suppressor that associates with various types of cancer. Although DLC2 shares a similar domain structure with that of DLC1, the function of DLC2 is not well characterized. Here, we describe the expression and ablation of DLC2 in mice using a reporter-knockout approach. DLC2 is expressed in several tissues and in endothelial cells (ECs) of blood vessels. Although ECs and blood vessels show no histological abnormalities and mice appear overall healthy, DLC2-mutant mice display enhanced angiogenic responses induced by matrigel and by tumor cells. Silencing of DLC2 in human ECs has reduced cell attachment, increased migration, and tube formation. These changes are rescued by silencing of RhoA, suggesting that the process is RhoA pathway dependent. These results indicate that DLC2 is not required for mouse development and normal vessel formation, but may protect mouse from unwanted angiogenesis induced by, for example, tumor cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Ching YP, Wong CM, Chan SF, Leung TH, Ng DC, Jin DY et al. (2003). Deleted in liver cancer (DLC) 2 encodes a RhoGAP protein with growth suppressor function and is underexpressed in hepatocellular carcinoma. J Biol Chem 278: 10824–10830.
Durkin ME, Avner MR, Huh CG, Yuan BZ, Thorgeirsson SS, Popescu NC . (2005). DLC-1, a Rho GTPase-activating protein with tumor suppressor function, is essential for embryonic development. FEBS Lett 579: 1191–1196.
Durkin ME, Ullmannova V, Guan M, Popescu NC . (2007a). Deleted in liver cancer 3 (DLC-3), a novel Rho GTPase-activating protein, is downregulated in cancer and inhibits tumor cell growth. Oncogene 26: 4580–4589.
Durkin ME, Yuan BZ, Thorgeirsson SS, Popescu NC . (2002). Gene structure, tissue expression, and linkage mapping of the mouse DLC-1 gene (Arhgap7). Gene 288: 119–127.
Durkin ME, Yuan BZ, Zhou X, Zimonjic DB, Lowy DR, Thorgeirsson SS et al. (2007b). DLC-1: a Rho GTPase-activating protein and tumour suppressor. J Cell Mol Med 11: 1185–1207.
Leung TH, Ching YP, Yam JW, Wong CM, Yau TO, Jin DY et al. (2005). Deleted in liver cancer 2 (DLC2) suppresses cell transformation by means of inhibition of RhoA activity. Proc Natl Acad Sci USA 102: 15207–15212.
Liao YC, Lo SH . (2008). Deleted in liver cancer-1 (DLC-1): a tumor suppressor not just for liver. Int J Biochem Cell Biol 40: 843–847.
Liao YC, Shih YP, Lo SH . (2008). Mutations in the focal adhesion targeting region of deleted in liver cancer-1 attenuate their expression and function. Cancer Res 68: 7718–7722.
Liao YC, Si L, Devere White RW, Lo SH . (2007). The phosphotyrosine-independent interaction of DLC-1 and the SH2 domain of cten regulates focal adhesion localization and growth suppression activity of DLC-1. J Cell Biol 176: 43–49.
Masson V, de la Ballina LR, Munaut C, Wielockx B, Jost M, Maillard C et al. (2005). Contribution of host MMP-2 and MMP-9 to promote tumor vascularization and invasion of malignant keratinocytes. FASEB J 19: 234–236.
Qian X, Li G, Asmussen HK, Asnaghi L, Vass WC, Braverman R et al. (2007). Oncogenic inhibition by a deleted in liver cancer gene requires cooperation between tensin binding and Rho-specific GTPase-activating protein activities. Proc Natl Acad Sci USA 104: 9012–9017.
Testa G, Schaft J, van der Hoeven F, Glaser S, Anastassiadis K, Zhang Y et al. (2004). A reliable lacZ expression reporter cassette for multipurpose, knockout-first alleles. Genesis 38: 151–158.
Ullmannova V, Popescu NC . (2006). Expression profile of the tumor suppressor genes DLC-1 and DLC-2 in solid tumors. Int J Oncol 29: 1127–1132.
Xiaorong L, Wei W, Liyuan Q, Kaiyan Y . (2008). Underexpression of deleted in liver cancer 2 (DLC2) is associated with overexpression of RhoA and poor prognosis in hepatocellular carcinoma. BMC Cancer 8: 205.
Xue W, Krasnitz A, Lucito R, Sordella R, Vanaelst L, Cordon-Cardo C et al. (2008). DLC1 is a chromosome 8p tumor suppressor whose loss promotes hepatocellular carcinoma. Genes Dev 22: 1439–1444.
Yau TO, Leung TH, Lam S, Cheung OF, Tung EK, Khong PL et al. (2009). Deleted in liver cancer 2 (DLC2) was dispensable for development and its deficiency did not aggravate hepatocarcinogenesis. PLoS One 4: e6566.
Yuan BZ, Miller MJ, Keck CL, Zimonjic DB, Thorgeirsson SS, Popescu NC . (1998). Cloning, characterization, and chromosomal localization of a gene frequently deleted in human liver cancer (DLC-1) homologous to rat RhoGAP. Cancer Res 58: 2196–2199.
Acknowledgements
We thank the Knockout Mouse Project Repository for DLC2 ES cells. This work is supported in part by grants from the National Institutes of Health (CA102537) to SHL.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Lin, Y., Chen, NT., Shih, YP. et al. DLC2 modulates angiogenic responses in vascular endothelial cells by regulating cell attachment and migration. Oncogene 29, 3010–3016 (2010). https://doi.org/10.1038/onc.2010.54
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2010.54
Keywords
This article is cited by
-
Functional analysis of the Drosophila RhoGAP Cv-c protein and its equivalence to the human DLC3 and DLC1 proteins
Scientific Reports (2018)
-
The cellular and molecular determinants of emphysematous destruction in COPD
Scientific Reports (2017)
-
The RhoGAP protein Deleted in Liver Cancer 3 (DLC3) is essential for adherens junctions integrity
Oncogenesis (2012)