Abstract
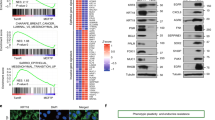
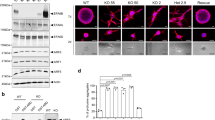
Acquisition of independence from anchorage to the extracellular matrix is a critical event for onset and progression of solid cancers. To identify and characterize new genes conferring anchorage independence, we transduced MCF10A human normal breast cells with a retroviral cDNA expression library and selected them by growth in suspension. Microarray analysis targeted on library-derived transcripts revealed robust and reproducible enrichment, after selection, of cDNAs encoding the scaffolding adaptor Gab2. Gab2 was confirmed to strongly promote anchorage-independent growth when overexpressed. Interestingly, downregulation by RNA interference of endogenous Gab2 in neoplastic cells did not affect their adherent growth, but abrogated their growth in soft agar. Gab2-driven anchorage independence was found to specifically involve activation of the Src-Stat3 signaling axis. A transcriptional ‘signature’ of 205 genes was obtained from GAB2-transduced, anchorage-independent MCF10A cells, and found to contain two main functional modules, controlling proliferation and cell adhesion/migration/invasion, respectively. Extensive validation on breast cancer data sets showed that the GAB2 signature provides a robust prognostic classifier for breast cancer metastatic relapse, largely independent from existing clinical and genomic indicators and from estrogen receptor status. This work highlights a pivotal role for GAB2 and its transcriptional targets in anchorage-independent growth and breast cancer metastatic progression.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Ariga N, Sato E, Ohuchi N, Nagura H, Ohtani H . (2001). Stromal expression of fibroblast activation protein/seprase, a cell membrane serine proteinase and gelatinase, is associated with longer survival in patients with invasive ductal carcinoma of breast. Int J Cancer 95: 67–72.
Bennett HL, Brummer T, Jeanes A, Yap AS, Daly RJ . (2007). Gab2 and Src co-operate in human mammary epithelial cells to promote growth factor independence and disruption of acinar morphogenesis. Oncogene 27: 2693–2704.
Berrier AL, Yamada KM . (2007). Cell-matrix adhesion. J Cell Physiol 213: 565–573.
Bouscary D, Lecoq-Lafon C, Chretien S, Zompi S, Fichelson S, Muller O et al. (2001). Role of Gab proteins in phosphatidylinositol 3-kinase activation by thrombopoietin (Tpo). Oncogene 20: 2197–2204.
Brummer T, Schramek D, Hayes VM, Bennett HL, Caldon CE, Musgrove EA et al. (2006). Increased proliferation and altered growth factor dependence of human mammary epithelial cells overexpressing the Gab2 docking protein. J Biol Chem 281: 626–637.
Buyse M, Loi S, Van't VL, Viale G, Delorenzi M, Glas AM et al. (2006). Validation and clinical utility of a 70-gene prognostic signature for women with node-negative breast cancer. J Natl Cancer Inst 98: 1183–1192.
Cooley J, Takayama TK, Shapiro SD, Schechter NM, Remold-O'Donnell E . (2001). The serpin MNEI inhibits elastase-like and chymotrypsin-like serine proteases through efficient reactions at two active sites. Biochemistry 40: 15762–15770.
Daly RJ, Gu H, Parmar J, Malaney S, Lyons RJ, Kairouz R et al. (2002). The docking protein Gab2 is overexpressed and estrogen regulated in human breast cancer. Oncogene 21: 5175–5181.
Danen EH, Yamada KM . (2001). Fibronectin, integrins, and growth control. J Cell Physiol 189: 1–13.
Debnath J, Brugge JS . (2005). Modelling glandular epithelial cancers in three-dimensional cultures. Nat Rev Cancer 5: 675–688.
DerMardirossian C, Bokoch GM . (2005). GDIs: central regulatory molecules in Rho GTPase activation. Trends Cell Biol 15: 356–363.
Desmedt C, Piette F, Loi S, Wang Y, Lallemand F, Haibe-Kains B et al. (2007). Strong time dependence of the 76-gene prognostic signature for node-negative breast cancer patients in the TRANSBIG multicenter independent validation series. Clin Cancer Res 13: 3207–3214.
ffrench-Constant C, Colognato H . (2004). Integrins: versatile integrators of extracellular signals. Trends Cell Biol 14: 678–686.
Garcia R, Bowman TL, Niu G, Yu H, Minton S, Muro-Cacho CA et al. (2001). Constitutive activation of Stat3 by the Src and JAK tyrosine kinases participates in growth regulation of human breast carcinoma cells. Oncogene 20: 2499–2513.
Geiger TR, Peeper DS . (2007). Critical role for TrkB kinase function in anoikis suppression, tumorigenesis, and metastasis. Cancer Res 67: 6221–6229.
Gu H, Pratt JC, Burakoff SJ, Neel BG . (1998). Cloning of p97/Gab2, the major SHP2-binding protein in hematopoietic cells, reveals a novel pathway for cytokine-induced gene activation. Mol Cell 2: 729–740.
Guo W, Giancotti FG . (2004). Integrin signalling during tumour progression. Nat Rev Mol Cell Biol 5: 816–826.
Hess V . (2008). Adjuvant!Online—an Internet-based decision tool for adjuvant chemotherapy in early breast cancer. Ther Umsch 65: 201–205.
Horst B, Gruvberger-Saal SK, Hopkins BD, Bordone L, Yang Y, Chernoff KA et al. (2009). Gab2-mediated signaling promotes melanoma metastasis. Am J Pathol 174: 1524–1533.
Ivshina AV, George J, Senko O, Mow B, Putti TC, Smeds J et al. (2006). Genetic reclassification of histologic grade delineates new clinical subtypes of breast cancer. Cancer Res 66: 10292–10301.
Ke Y, Wu D, Princen F, Nguyen T, Pang Y, Lesperance J et al. (2007). Role of Gab2 in mammary tumorigenesis and metastasis. Oncogene 26: 4951–4960.
Kitamura T, Koshino Y, Shibata F, Oki T, Nakajima H, Nosaka T et al. (2003). Retrovirus-mediated gene transfer and expression cloning: powerful tools in functional genomics. Exp Hematol 31: 1007–1014.
Kong M, Mounier C, Dumas V, Posner BI . (2003). Epidermal growth factor-induced DNA synthesis. Key role for Src phosphorylation of the docking protein Gab2. J Biol Chem 278: 5837–5844.
Laird AD, Li G, Moss KG, Blake RA, Broome MA, Cherrington JM et al. (2003). Src family kinase activity is required for signal tranducer and activator of transcription 3 and focal adhesion kinase phosphorylation and vascular endothelial growth factor signaling in vivo and for anchorage-dependent and -independent growth of human tumor cells. Mol Cancer Ther 2: 461–469.
Liu Y, Jenkins B, Shin JL, Rohrschneider LR . (2001). Scaffolding protein Gab2 mediates differentiation signaling downstream of Fms receptor tyrosine kinase. Mol Cell Biol 21: 3047–3056.
Martelli ML, Isella C, Mira A, Fu L, Cantarella D, Medico E . (2008). Exploiting orthologue diversity for systematic detection of gain-of-function phenotypes. BMC Genomics 9: 254.
Mason JM, Morrison DJ, Basson MA, Licht JD . (2006). Sprouty proteins: multifaceted negative-feedback regulators of receptor tyrosine kinase signaling. Trends Cell Biol 16: 45–54.
Miller LD, Smeds J, George J, Vega VB, Vergara L, Ploner A et al. (2005). An expression signature for p53 status in human breast cancer predicts mutation status, transcriptional effects, and patient survival. Proc Natl Acad Sci USA 102: 13550–13555.
Miranti CK, Brugge JS . (2002). Sensing the environment: a historical perspective on integrin signal transduction. Nat Cell Biol 4: E83–E90.
Ni S, Zhao C, Feng GS, Paulson RF, Correll PH . (2007). A novel Stat3 binding motif in Gab2 mediates transformation of primary hematopoietic cells by the Stk/Ron receptor tyrosine kinase in response to Friend virus infection. Mol Cell Biol 27: 3708–3715.
Nishida K, Yoshida Y, Itoh M, Fukada T, Ohtani T, Shirogane T et al. (1999). Gab-family adapter proteins act downstream of cytokine and growth factor receptors and T- and B-cell antigen receptors. Blood 93: 1809–1816.
Playford MP, Schaller MD . (2004). The interplay between Src and integrins in normal and tumor biology. Oncogene 23: 7928–7946.
Reginato MJ, Mills KR, Paulus JK, Lynch DK, Sgroi DC, Debnath J et al. (2003). Integrins and EGFR coordinately regulate the pro-apoptotic protein Bim to prevent anoikis. Nat Cell Biol 5: 733–740.
Roux KJ, Amici SA, Fletcher BS, Notterpek L . (2005). Modulation of epithelial morphology, monolayer permeability, and cell migration by growth arrest specific 3/peripheral myelin protein 22. Mol Biol Cell 16: 1142–1151.
Sattler M, Mohi MG, Pride YB, Quinnan LR, Malouf NA, Podar K et al. (2002). Critical role for Gab2 in transformation by BCR/ABL. Cancer Cell 1: 479–492.
Schwartz MA, Baron V . (1999). Interactions between mitogenic stimuli, or, a thousand and one connections. Curr Opin Cell Biol 11: 197–202.
Sotiriou C, Wirapati P, Loi S, Harris A, Fox S, Smeds J et al. (2006). Gene expression profiling in breast cancer: understanding the molecular basis of histologic grade to improve prognosis. J Natl Cancer Inst 98: 262–272.
Soule HD, Maloney TM, Wolman SR, Peterson Jr WD, Brenz R, McGrath CM et al. (1990). Isolation and characterization of a spontaneously immortalized human breast epithelial cell line, MCF-10. Cancer Res 50: 6075–6086.
tires-Alj M, Gil SG, Chan R, Wang ZC, Wang Y, Imanaka N et al. (2006). A role for the scaffolding adapter GAB2 in breast cancer. Nat Med 12: 114–121.
Tsatsanis C, Spandidos DA . (2004). Oncogenic kinase signaling in human neoplasms. Ann NY Acad Sci 1028: 168–175.
van 't Veer LJ, Dai H, van d V, He YD, Hart AA, Mao M et al. (2002). Gene expression profiling predicts clinical outcome of breast cancer. Nature 415: 530–536.
van de Vijver MJ, He YD, van 't Veer LJ, Dai H, Hart AA, Voskuil DW et al. (2002). A gene-expression signature as a predictor of survival in breast cancer. N Engl J Med 347: 1999–2009.
Wang Y, Klijn JG, Zhang Y, Sieuwerts AM, Look MP, Yang F et al. (2005). Gene-expression profiles to predict distant metastasis of lymph-node-negative primary breast cancer. Lancet 365: 671–679.
Wessels LF, Reinders MJ, Hart AA, Veenman CJ, Dai H, He YD et al. (2005). A protocol for building and evaluating predictors of disease state based on microarray data. Bioinformatics 21: 3755–3762.
Wirapati P, Sotiriou C, Kunkel S, Farmer P, Pradervand S, Haibe-Kains B et al. (2008). Meta-analysis of gene expression profiles in breast cancer: toward a unified understanding of breast cancer subtyping and prognosis signatures. Breast Cancer Res 10: R65.
Yu CL, Meyer DJ, Campbell GS, Larner AC, Carter-Su C, Schwartz J et al. (1995). Enhanced DNA-binding activity of a Stat3-related protein in cells transformed by the Src oncoprotein. Science 269: 81–83.
Yu M, Luo J, Yang W, Wang Y, Mizuki M, Kanakura Y et al. (2006). The scaffolding adapter Gab2, via Shp-2, regulates kit-evoked mast cell proliferation by activating the Rac/JNK pathway. J Biol Chem 281: 28615–28626.
Yuan K, Listinsky CM, Singh RK, Listinsky JJ, Siegal GP . (2008). Cell surface associated alpha-L: -fucose moieties modulate human breast cancer neoplastic progression. Pathol Oncol Res 14: 145–156.
Zatkova A, Schoch C, Speleman F, Poppe B, Mannhalter C, Fonatsch C et al. (2006). GAB2 is a novel target of 11q amplification in AML/MDS. Genes Chromosomes Cancer 45: 798–807.
Zhang SQ, Yang W, Kontaridis MI, Bivona TG, Wen G, Araki T et al. (2004). Shp2 regulates SRC family kinase activity and Ras/Erk activation by controlling Csk recruitment. Mol Cell 13: 341–355.
Zhang W, Zong CS, Hermanto U, Lopez-Bergami P, Ronai Z, Wang LH . (2006). RACK1 recruits STAT3 specifically to insulin and insulin-like growth factor 1 receptors for activation, which is important for regulating anchorage-independent growth. Mol Cell Biol 26: 413–424.
Acknowledgements
We thank Barbara Martinoglio and Roberta Porporato for technical assistance. We thank Simona Destefanis, Antonella Cignetto and Michela Bruno for secretarial assistance. This research was supported by grants from AIRC, the EC (contract no. 503438 ‘TRANSFOG’), CNR-MIUR, FIRB-MIUR, Ministero della Salute, Regione Piemonte and the Foundations CRT and ‘Compagnia di San Paolo.’
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc)
Rights and permissions
About this article
Cite this article
Mira, A., Isella, C., Renzulli, T. et al. The GAB2 signaling scaffold promotes anchorage independence and drives a transcriptional response associated with metastatic progression of breast cancer. Oncogene 28, 4444–4455 (2009). https://doi.org/10.1038/onc.2009.296
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2009.296
Keywords
This article is cited by
-
Expression of signaling adaptor proteins predicts poor prognosis in pancreatic ductal adenocarcinoma
Diagnostic Pathology (2017)
-
Gab2 facilitates epithelial-to-mesenchymal transition via the MEK/ERK/MMP signaling in colorectal cancer
Journal of Experimental & Clinical Cancer Research (2016)
-
Mulcom: a multiple comparison statistical test for microarray data in Bioconductor
BMC Bioinformatics (2011)