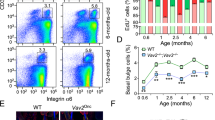
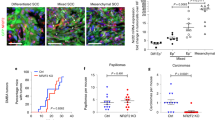
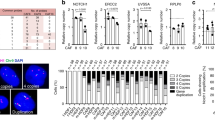
Abstract
Cancer development results from deregulated control of stem cell populations and alterations in their surrounding environment. Notch signaling is an important form of direct cell–cell communication involved in cell fate determination, stem cell potential and lineage commitment. The biological function of this pathway is critically context dependent. Here we review the pro-differentiation role and tumor suppressing function of this pathway, as revealed by loss-of-function in keratinocytes and skin, downstream of p53 and in cross-connection with other determinants of stem cell potential and/or tumor formation, such as p63 and Rho/CDC42 effectors. The possibility that Notch signaling elicits a duality of signals, involved in growth/differentiation control and cell survival will be discussed, in the context of novel approaches for cancer therapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Alimirah F, Panchanathan R, Davis FJ, Chen J, Choubey D . (2007). Restoration of p53 expression in human cancer cell lines upregulates the expression of Notch1: implications for cancer cell fate determination after genotoxic stress. Neoplasia 9: 427–434.
Altucci L, Gronemeyer H . (2001). The promise of retinoids to fight against cancer. Nat Rev Cancer 1: 181–193.
Armstrong BK, Kricker A . (2001). The epidemiology of UV induced skin cancer. J Photochem Photobiol B 63: 8–18.
Artavanis-Tsakonas S, Rand MD, Lake RJ . (1999). Notch signaling: cell fate control and signal integration in development. Science 284: 770–776.
Axelrod JD, Matsuno K, Artavanis-Tsakonas S, Perrimon N . (1996). Interaction between wingless and Notch signaling pathways mediated by dishevelled. Science 271: 1826–1832.
Balint K, Xiao M, Pinnix CC, Soma A, Veres I, Juhasz I et al. (2005). Activation of Notch1 signaling is required for beta-catenin-mediated human primary melanoma progression. J Clin Invest 115: 3166–3176.
Benitah SA, Frye M, Glogauer M, Watt FM . (2005). Stem cell depletion through epidermal deletion of Rac1. Science 309: 933–935.
Beverly LJ, Felsher DW, Capobianco AJ . (2005). Suppression of p53 by Notch in lymphomagenesis: implications for initiation and regression. Cancer Res 65: 7159–7168.
Blanpain C, Lowry WE, Pasolli HA, Fuchs E . (2006). Canonical notch signaling functions as a commitment switch in the epidermal lineage. Genes Dev 20: 3022–3035.
Blokzijl A, Dahlqvist C, Reissmann E, Falk A, Moliner A, Lendahl U et al. (2003). Cross-talk between the Notch and TGF-beta signaling pathways mediated by interaction of the Notch intracellular domain with Smad3. J Cell Biol 163: 723–728.
Bray SJ . (2006). Notch signalling: a simple pathway becomes complex. Nat Rev Mol Cell Biol 7: 678–689.
Brunet A, Bonni A, Zigmond MJ, Lin MZ, Juo P, Hu LS et al. (1999). Akt promotes cell survival by phosphorylating and inhibiting a Forkhead transcription factor. Cell 96: 857–868.
Burgering BM, Kops GJ . (2002). Cell cycle and death control: long live Forkheads. Trends Biochem Sci 27: 352–360.
Carpten J, Nupponen N, Isaacs S, Sood R, Robbins C, Xu J et al. (2002). Germline mutations in the ribonuclease L gene in families showing linkage with HPC1. Nat Genet 30: 181–184.
Chen Y, De Marco MA, Graziani I, Gazdar AF, Strack PR, Miele L et al. (2007). Oxygen concentration determines the biological effects of NOTCH-1 signaling in adenocarcinoma of the lung. Cancer Res 67: 7954–7959.
Couso JP, Martinez Arias A . (1994). Notch is required for wingless signaling in the epidermis of Drosophila. Cell 79: 259–272.
D’Errico M, Lemma T, Calcagnile A, Proietti De Santis L, Dogliotti E . (2007). Cell type and DNA damage specific response of human skin cells to environmental agents. Mutat Res 614: 37–47.
Dajee M, Lazarov M, Zhang JY, Cai T, Green CL, Russell AJ et al. (2003). NF-kappaB blockade and oncogenic Ras trigger invasive human epidermal neoplasia. Nature 421: 639–643.
de Gruijl FR . (1999). Skin cancer and solar UV radiation. Eur J Cancer 35: 2003–2009.
Devgan V, Mammucari C, Millar SE, Brisken C, Dotto GP . (2005). p21WAF1/Cip1 is a negative transcriptional regulator of Wnt4 expression downstream of Notch1 activation. Genes Dev 19: 1485–1495.
Dotto GP, Cotsarelis G . (2005). Developmental biology. Rac1 up for epidermal stem cells. Science 309: 890–891.
Dotto GP, Moellmann G, Ghosh S, Edwards M, Halaban R . (1989). Transformation of murine melanocytes by basic fibroblast growth factor cDNA and oncogenes and selective suppression of the transformed phenotype in a reconstituted cutaneous environment. J Cell Biol 109: 3115–3128.
Estrach S, Cordes R, Hozumi K, Gossler A, Watt FM . (2008). Role of the Notch ligand Delta1 in embryonic and adult mouse epidermis. J Invest Dermatol 128: 825–832.
Galceran J, Sustmann C, Hsu SC, Folberth S, Grosschedl R . (2004). LEF1-mediated regulation of Delta-like1 links Wnt and Notch signaling in somitogenesis. Genes Dev 18: 2718–2723.
Gat U, DasGupta R, Degenstein L, Fuchs E . (1998). De novo hair follicle morphogenesis and hair tumors in mice expressing a truncated beta-catenin in skin. Cell 95: 605–614.
Ghosh A, Sarkar SN, Rowe TM, Sen GC . (2001). A specific isozyme of 2′–5′ oligoadenylate synthetase is a dual function proapoptotic protein of the Bcl-2 family. J Biol Chem 276: 25447–25455.
Gius D, Cui H, Bradbury CM, Cook J, Smart DK, Zhao S et al. (2004). Distinct effects on gene expression of chemical and genetic manipulation of the cancer epigenome revealed by a multimodality approach. Cancer Cell 6: 361–371.
Gomis RR, Alarcon C, He W, Wang Q, Seoane J, Lash A et al. (2006). A FoxO-Smad synexpression group in human keratinocytes. Proc Natl Acad Sci USA 103: 12747–12752.
Hahn WC, Weinberg RA . (2002). Modelling the molecular circuitry of cancer. Nat Rev Cancer 2: 331–341.
Hofmann M, Schuster-Gossler K, Watabe-Rudolph M, Aulehla A, Herrmann BG, Gossler A . (2004). WNT signaling, in synergy with T/TBX6, controls Notch signaling by regulating Dll1 expression in the presomitic mesoderm of mouse embryos. Genes Dev 18: 2712–2717.
Iso T, Kedes L, Hamamori Y . (2003). HES and HERP families: multiple effectors of the Notch signaling pathway. J Cell Physiol 194: 237–255.
Itoh F, Itoh S, Goumans MJ, Valdimarsdottir G, Iso T, Dotto GP et al. (2004). Synergy and antagonism between Notch and BMP receptor signaling pathways in endothelial cells. EMBO J 23: 541–551.
Jehn BM, Bielke W, Pear WS, Osborne BA . (1999). Cutting edge: protective effects of notch-1 on TCR-induced apoptosis. J Immunol 162: 635–638.
Keyes WM, Wu Y, Vogel H, Guo X, Lowe SW, Mills AA . (2005). p63 deficiency activates a program of cellular senescence and leads to accelerated aging. Genes Dev 19: 1986–1999.
Kim JW, Kim MJ, Kim KJ, Yun HJ, Chae JS, Hwang SG et al. (2005). Notch interferes with the scaffold function of JNK-interacting protein 1 to inhibit the JNK signaling pathway. Proc Natl Acad Sci USA 102: 14308–14313.
King KE, Ponnamperuma RM, Yamashita T, Tokino T, Lee LA, Young MF et al. (2003). deltaNp63alpha functions as both a positive and a negative transcriptional regulator and blocks in vitro differentiation of murine keratinocytes. Oncogene 22: 3635–3644.
Kolev V, Mandinova A, Guinea-Viniegra J, Hu B, Lefort K, Lambertini et al. (2008). EGFR signaling as negative regulator of Notch1 gene transcription and function in poliferating keratinocytes and cancer. Nature Cell Biol (doi:10.1038/ncb1750).
Koster MI, Roop DR . (2004). p63 and epithelial appendage development. Differentiation 72: 364–370.
Kunnimalaiyaan M, Vaccaro AM, Ndiaye MA, Chen H . (2006). Overexpression of the NOTCH1 intracellular domain inhibits cell proliferation and alters the neuroendocrine phenotype of medullary thyroid cancer cells. J Biol Chem 281: 39819–39830.
Lai EC . (2002). Keeping a good pathway down: transcriptional repression of Notch pathway target genes by CSL proteins. EMBO Rep 3: 840–845.
Latonen L, Laiho M . (2005). Cellular UV damage responses—functions of tumor suppressor p53. Biochim Biophys Acta 1755: 71–89.
Laurikkala J, Mikkola ML, James M, Tummers M, Mills AA, Thesleff I . (2006). p63 regulates multiple signalling pathways required for ectodermal organogenesis and differentiation. Development 133: 1553–1563.
Laws AM, Osborne BA . (2004). p53 regulates thymic Notch1 activation. Eur J Immunol 34: 726–734.
Lee J, Basak JM, Demehri S, Kopan R . (2007). Bi-compartmental communication contributes to the opposite proliferative behavior of Notch1-deficient hair follicle and epidermal keratinocytes. Development 134: 2795–2806.
Lefort K, Mandinova A, Ostano P, Kolev V, Calpini V, Kolfschoten I et al. (2007). Notch1 is a p53 target gene involved in human keratinocyte tumor suppression through negative regulation of ROCK1/2 and MRCKalpha kinases. Genes Dev 21: 562–577.
Li T, Wen H, Brayton C, Das P, Smithson LA, Fauq A et al. (2007). Epidermal growth factor receptor and notch pathways participate in the tumor suppressor function of gamma-secretase. J Biol Chem 282: 32264–32273.
Lin MH, Leimeister C, Gessler M, Kopan R . (2000). Activation of the Notch pathway in the hair cortex leads to aberrant differentiation of the adjacent hair-shaft layers. Development 127: 2421–2432.
Lowell S, Jones P, Le Roux I, Dunne J, Watt FM . (2000). Stimulation of human epidermal differentiation by delta-notch signalling at the boundaries of stem-cell clusters. Curr Biol 10: 491–500.
Luo B, Aster JC, Hasserjian RP, Kuo F, Sklar J . (1997). Isolation and functional analysis of a cDNA for human Jagged2, a gene encoding a ligand for the Notch1 receptor. Mol Cell Biol 17: 6057–6067.
MacKenzie F, Duriez P, Wong F, Noseda M, Karsan A . (2004). Notch4 inhibits endothelial apoptosis via RBP-Jkappa-dependent and -independent pathways. J Biol Chem 279: 11657–11663.
Maillard I, Weng AP, Carpenter AC, Rodriguez CG, Sai H, Xu L et al. (2004). Mastermind critically regulates Notch-mediated lymphoid cell fate decisions. Blood 104: 1696–1702.
Malanchi I, Peinado H, Kassen D, Hussenet T, Metzger D, Chambon P et al. (2008). Cutaneous cancer stem cell maintenance is dependent on beta-catenin signalling. Nature 452: 650–653.
Mammucari C, Tommasi di Vignano A, Sharov AA, Havrda MC, Roop DR, Botchkarev VA et al. (2005). Integration of Notch1 and Calcineurin/NFAT signaling pathways in keratinocyte growth and differentiation control. Dev Cell 8: 665–676.
Mandinova A, Lefort K, Tommasi di Vignano A, Stonely W, Ostano P, Chiorino G et al. (2008). The FoxO3a gene is a key negative target of canonical Notch signalling in the keratinocyte UVB response. EMBO J 27: 1243–1254.
Merlo LM, Pepper JW, Reid BJ, Maley CC . (2006). Cancer as an evolutionary and ecological process. Nat Rev Cancer 6: 924–935.
Miele L, Miao H, Nickoloff BJ . (2006). NOTCH signaling as a novel cancer therapeutic target. Curr Cancer Drug Targets 6: 313–323.
Mills AA, Zheng B, Wang XJ, Vogel H, Roop DR, Bradley A . (1999). p63 is a p53 homologue required for limb and epidermal morphogenesis. Nature 398: 708–713.
Missero C, Di Cunto F, Kiyokawa H, Koff A, Dotto GP . (1996). The absence of p21Cip1/WAF1 alters keratinocyte growth and differentiation and promotes ras-tumor progression. Genes Dev 10: 3065–3075.
Moriyama M, Durham AD, Moriyama H, Hasegawa K, Nishikawa S, Radtke F et al. (2008). Multiple roles of Notch signaling in the regulation of epidermal development. Dev Cell 14: 594–604.
Mumm JS, Kopan R . (2000). Notch signaling: from the outside in. Dev Biol 228: 151–165.
Mungamuri SK, Yang X, Thor AD, Somasundaram K . (2006). Survival signaling by Notch1: mammalian target of rapamycin (mTOR)-dependent inhibition of p53. Cancer Res 66: 4715–4724.
Nair P, Somasundaram K, Krishna S . (2003). Activated Notch1 inhibits p53-induced apoptosis and sustains transformation by human papillomavirus type 16 E6 and E7 oncogenes through a PI3K-PKB/Akt-dependent pathway. J Virol 77: 7106–7112.
Nguyen BC, Lefort K, Mandinova A, Antonini D, Devgan V, Della Gatta G et al. (2006). Cross-regulation between Notch and p63 in keratinocyte commitment to differentiation. Genes Dev 20: 1028–1042.
Nickoloff B, Qin J, Chaturvedi V, Denning M, Bonish B, Miele L . (2002). Jagged-1 mediated activation of notch signaling induces complete maturation of human keratinocytes through NF-kappaB and PPARgamma. Cell Death Differ 9: 842–855.
Nicolas M, Wolfer A, Raj K, Kummer JA, Mill P, Van Noort M et al. (2003). Notch1 functions as a tumor suppressor in mouse skin. Nat Genet 33: 416–421.
Niimi H, Pardali K, Vanlandewijck M, Heldin CH, Moustakas A . (2007). Notch signaling is necessary for epithelial growth arrest by TGF-beta. J Cell Biol 176: 695–707.
Oishi K, Kamakura S, Isazawa Y, Yoshimatsu T, Kuida K, Nakafuku M et al. (2004). Notch promotes survival of neural precursor cells via mechanisms distinct from those regulating neurogenesis. Dev Biol 276: 172–184.
Okuyama R, Ogawa E, Nagoshi H, Yabuki M, Kurihara A, Terui T et al. (2007). p53 homologue, p51/p63, maintains the immaturity of keratinocyte stem cells by inhibiting Notch1 activity. Oncogene 26: 4478–4488.
Osipo C, Golde TE, Osborne BA, Miele LA . (2008). Off the beaten pathway: the complex cross talk between Notch and NF-kappaB. Lab Invest 88: 11–17.
Oswald F, Liptay S, Adler G, Schmid RM . (1998). NF-kappaB2 is a putative target gene of activated Notch-1 via RBP-Jkappa. Mol Cell Biol 18: 2077–2088.
Oswald F, Tauber B, Dobner T, Bourteele S, Kostezka U, Adler G et al. (2001). p300 acts as a transcriptional coactivator for mammalian Notch-1. Mol Cell Biol 21: 7761–7774.
Pan Y, Lin MH, Tian X, Cheng HT, Gridley T, Shen J et al. (2004). Gamma-secretase functions through Notch signaling to maintain skin appendages but is not required for their patterning or initial morphogenesis. Dev Cell 7: 731–743.
Parsa R, Yang A, McKeon F, Green H . (1999). Association of p63 with proliferative potential in normal and neoplastic human keratinocytes. J Invest Dermatol 113: 1099–1105.
Pellegrini G, Dellambra E, Golisano O, Martinelli E, Fantozzi I, Bondanza S et al. (2001). p63 identifies keratinocyte stem cells. Proc Natl Acad Sci USA 98: 3156–3161.
Perera RJ, Koo S, Bennett CF, Dean NM, Gupta N, Qin JZ et al. (2006). Defining the transcriptome of accelerated and replicatively senescent keratinocytes reveals links to differentiation, interferon signaling, and Notch related pathways. J Cell Biochem 98: 394–408.
Petcherski AG, Kimble J . (2000). Mastermind is a putative activator for Notch. Curr Biol 10: R471–R473.
Philipp J, Vo K, Gurley KE, Seidel K, Kemp CJ . (1999). Tumor suppression by p27Kip1 and p21Cip1 during chemically induced skin carcinogenesis. Oncogene 18: 4689–4698.
Proweller A, Tu L, Lepore JJ, Cheng L, Lu MM, Seykora J et al. (2006). Impaired notch signaling promotes de novo squamous cell carcinoma formation. Cancer Res 66: 7438–7444.
Ramdass B, Maliekal TT, Lakshmi S, Rehman M, Rema P, Nair P et al. (2007). Coexpression of Notch1 and NF-kappaB signaling pathway components in human cervical cancer progression. Gynecol Oncol 104: 352–361.
Rangarajan A, Hong SJ, Gifford A, Weinberg RA . (2004). Species- and cell type-specific requirements for cellular transformation. Cancer Cell 6: 171–183.
Rangarajan A, Talora C, Okuyama R, Nicolas M, Mammucari C, Oh H et al. (2001). Notch signaling is a direct determinant of keratinocyte growth arrest and entry into differentiation. EMBO J 20: 3427–3436.
Sade H, Krishna S, Sarin A . (2004). The anti-apoptotic effect of Notch-1 requires p56lck-dependent, Akt/PKB-mediated signaling in T cells. J Biol Chem 279: 2937–2944.
Sahai E, Marshall CJ . (2002). RHO-GTPases and cancer. Nat Rev Cancer 2: 133–142.
Shin HM, Minter LM, Cho OH, Gottipati S, Fauq AH, Golde TE et al. (2006). Notch1 augments NF-kappaB activity by facilitating its nuclear retention. EMBO J 25: 129–138.
Sriuranpong V, Borges MW, Ravi RK, Arnold DR, Nelkin BD, Baylin SB et al. (2001). Notch signaling induces cell cycle arrest in small cell lung cancer cells. Cancer Res 61: 3200–3205.
Sriuranpong V, Borges MW, Strock CL, Nakakura EK, Watkins DN, Blaumueller CM et al. (2002). Notch signaling induces rapid degradation of achaete-scute homolog 1. Mol Cell Biol 22: 3129–3139.
Talora C, Cialfi S, Segatto O, Morrone S, Kim Choi J, Frati L et al. (2005). Constitutively active Notch1 induces growth arrest of HPV-positive cervical cancer cells via separate signaling pathways. Exp Cell Res 305: 343–354.
Talora C, Sgroi DC, Crum CP, Dotto GP . (2002). Specific down-modulation of Notch1 signaling in cervical cancer cells is required for sustained HPV-E6/E7 expression and late steps of malignant transformation. Genes Dev 16: 2252–2263.
Thelu J, Rossio P, Favier B . (2002). Notch signalling is linked to epidermal cell differentiation level in basal cell carcinoma, psoriasis and wound healing. BMC Dermatol 2: 7.
Topley GI, Okuyama R, Gonzales JG, Conti C, Dotto GP . (1999). p21(WAF1/Cip1) functions as a suppressor of malignant skin tumor formation and a determinant of keratinocyte stem-cell potential. Proc Natl Acad Sci USA 96: 9089–9094.
Truong AB, Kretz M, Ridky TW, Kimmel R, Khavari PA . (2006). p63 regulates proliferation and differentiation of developmentally mature keratinocytes. Genes Dev 20: 3185–3197.
Vauclair S, Majo F, Durham A, Ghyselinck N, Barrandon Y, Radtke F . (2007). Corneal epithelial cell fate is maintained during repair by Notch1 signaling via the regulation of vitamin A metabolism. Dev Cell 13: 242–253.
Wang L, Qin H, Chen B, Xin X, Li J, Han H . (2007). Overexpressed active Notch1 induces cell growth arrest of HeLa cervical carcinoma cells. Int J Gynecol Cancer 17: 1283–1292.
Wasylyk C, Schlumberger SE, Criqui-Filipe P, Wasylyk B . (2002). Sp100 interacts with ETS-1 and stimulates its transcriptional activity. Mol Cell Biol 22: 2687–2702.
Wei CL, Wu Q, Vega VB, Chiu KP, Ng P, Zhang T et al. (2006). A global map of p53 transcription-factor binding sites in the human genome. Cell 124: 207–219.
Weinberg WC, Fernandez-Salas E, Morgan DL, Shalizi A, Mirosh E, Stanulis E et al. (1999). Genetic deletion of p21WAF1 enhances papilloma formation but not malignant conversion in experimental mouse skin carcinogenesis. Cancer Res 59: 2050–2054.
Weng AP, Nam Y, Wolfe MS, Pear WS, Griffin JD, Blacklow SC et al. (2003). Growth suppression of pre-T acute lymphoblastic leukemia cells by inhibition of notch signaling. Mol Cell Biol 23: 655–664.
Westfall MD, Pietenpol JA . (2004). p63: molecular complexity in development and cancer. Carcinogenesis 25: 857–864.
Wu L, Aster JC, Blacklow SC, Lake R, Artavanis-Tsakonas S, Griffin JD . (2000). MAML1, a human homologue of Drosophila Mastermind, is a transcriptional co-activator for NOTCH receptors. Nat Genet 26: 484–489.
Wu X, Quondamatteo F, Lefever T, Czuchra A, Meyer H, Chrostek A et al. (2006). Cdc42 controls progenitor cell differentiation and beta-catenin turnover in skin. Genes Dev 20: 571–585.
Xia X, Qian S, Soriano S, Wu Y, Fletcher AM, Wang XJ et al. (2001). Loss of presenilin 1 is associated with enhanced beta-catenin signaling and skin tumorigenesis. Proc Natl Acad Sci USA 98: 10863–10868.
Xu X, Lyle S, Liu Y, Solky B, Cotsarelis G . (2003). Differential expression of cyclin D1 in the human hair follicle. Am J Pathol 163: 969–978.
Yamamoto N, Tanigaki K, Han H, Hiai H, Honjo T . (2003). Notch/RBP-J signaling regulates epidermis/hair fate determination of hair follicular stem cells. Curr Biol 13: 333–338.
Yang A, Kaghad M, Wang Y, Gillett E, Fleming MD, Dotsch V et al. (1998). p63, a p53 homolog at 3q27-29, encodes multiple products with transactivating, death-inducing, and dominant-negative activities. Mol Cell 2: 305–316.
Yang A, Schweitzer R, Sun D, Kaghad M, Walker N, Bronson RT et al. (1999). p63 is essential for regenerative proliferation in limb, craniofacial and epithelial development. Nature 398: 714–718.
Yang X, Klein R, Tian X, Cheng HT, Kopan R, Shen J . (2004). Notch activation induces apoptosis in neural progenitor cells through a p53-dependent pathway. Dev Biol 269: 81–94.
Yao J, Duan L, Fan M, Yuan J, Wu X . (2007). Notch1 induces cell cycle arrest and apoptosis in human cervical cancer cells: involvement of nuclear factor kappa B inhibition. Int J Gynecol Cancer 17: 502–510.
Yugawa T, Handa K, Narisawa-Saito M, Ohno S, Fujita M, Kiyono T . (2007). Regulation of Notch1 gene expression by p53 in epithelial cells. Mol Cell Biol 27: 3732–3742.
Yuspa SH (ed) (1991). Cutaneous Carcinogenesis: Natural and Experimental in Physiology Biochemistry and Molecular Biology of the Skin. Oxford University Press: New York.
Zagouras P, Stifani S, Blaumueller CM, Carcangiu ML, Artavanis-Tsakonas S . (1995). Alterations in Notch signaling in neoplastic lesions of the human cervix. Proc Natl Acad Sci USA 92: 6414–6418.
Zavadil J, Cermak L, Soto-Nieves N, Bottinger EP . (2004). Integration of TGF-beta/Smad and Jagged1/Notch signalling in epithelial-to-mesenchymal transition. EMBO J 23: 1155–1165.
Zhang JY, Green CL, Tao S, Khavari PA . (2004). NF-kappaB RelA opposes epidermal proliferation driven by TNFR1 and JNK. Genes Dev 18: 17–22.
Zhang L, Pagano JS . (2002). Structure and function of IRF-7. J Interferon Cytokine Res 22: 95–101.
Zheng Q, Qin H, Zhang H, Li J, Hou L, Wang H et al. (2007). Notch signaling inhibits growth of the human lung adenocarcinoma cell line A549. Oncol Rep 17: 847–852.
Zhu AJ, Watt FM . (1999). Beta-catenin signalling modulates proliferative potential of human epidermal keratinocytes independently of intercellular adhesion. Development 126: 2285–2298.
zur Hausen H . (2000). Papillomaviruses causing cancer: evasion from host-cell control in early events in carcinogenesis. J Natl Cancer Inst 92: 690–698.
Zweidler-McKay PA, He Y, Xu L, Rodriguez CG, Karnell FG, Carpenter AC et al. (2005). Notch signaling is a potent inducer of growth arrest and apoptosis in a wide range of B-cell malignancies. Blood 106: 3898–3906.
Acknowledgements
I thank Dr Cathrin Brisken for careful reading of the paper. This study was supported by NIH Grants AR39190, AR054856, the Swiss National Foundation, a grant from the European Union (Epistem, Sixth Framework Program, LSHB-CT-2005-019067).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dotto, G. Notch tumor suppressor function. Oncogene 27, 5115–5123 (2008). https://doi.org/10.1038/onc.2008.225
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2008.225
Keywords
This article is cited by
-
Lentinan enhances the antitumor effects of Delta-like 1 via neutrophils
BMC Cancer (2022)
-
Generation of a lung squamous cell carcinoma three-dimensional culture model with keratinizing structures
Scientific Reports (2021)
-
Identification of novel fusion transcripts in meningioma
Journal of Neuro-Oncology (2020)
-
A prospect of cell immortalization combined with matrix microenvironmental optimization strategy for tissue engineering and regeneration
Cell & Bioscience (2019)
-
Deciphering the cells of origin of squamous cell carcinomas
Nature Reviews Cancer (2018)