Abstract
Objectives:
To determine age-related associations between fat mass (FM), regional fat depots and cardiometabolic traits in normal- and overweight children, adolescents and adults.
Methods:
Detailed body composition (regional subcutaneous and visceral adipose tissue; SAT, VAT) by whole-body magnetic resonance imaging (MRI), FM and fat-free mass by air-displacement plethysmography, systolic and diastolic blood pressure (SBP, DBP), triglycerides (TG), high-density lipoprotein cholesterol (HDL), plasma glucose and plasma insulin were measured in 433 subjects (BMI: 23.6 (21.0–27.7); 151 children and adolescents, aged 6–18 years, 150 young adults, aged 18–30 years and 132 adults, aged 30–60 years). Data were derived from pooled data of the ‘Reference Center for Body Composition’ in Kiel, Germany. Insulin resistance was determined by the homeostatic model assessment of insulin resistance (HOMA-IR). Partial correlations and multivariate linear regression analyses were used to evaluate the associations between body composition and cardiometabolic traits. A descriptive approach was used to demonstrate age-dependent differences in associations between body fat depots and insulin resistance, independent of BMI.
Results:
FM, SAT, and VAT increased from childhood to adulthood with low VAT in children and adolescents. When compared to children, TG was higher in adults. HDL and DBP did not differ between age groups. Insulin resistance was highest in male adolescents and female young adults. Associations between body fat depots and cardiometabolic traits were seen after puberty with no associations in pre- and intrapubertal children. When compared to FM, SAT and VAT had the strongest association with insulin resistance in adults. This association was independent of BMI.
Conclusions:
Associations between individual body fat depots and most cardiometabolic traits became evident after puberty only. The strongest associations were observed between insulin resistance and abdominal fat in adults. The impact of VAT was independent of BMI.
Similar content being viewed by others
Introduction
To address the impact of overweight on cardiometabolic risk most studies are based on body mass index (BMI).1, 2, 3 However, BMI can neither differentiate between fat mass (FM) and fat-free mass (FFM) nor give detailed information about body fat distribution.4, 5 To go beyond BMI, detailed body composition and appropriate modeling taking into account body function have to be considered.6 Magnetic resonance imaging (MRI) and computed tomography are the gold-standard methods to determine trunk and peripheral subcutaneous adipose tissue (SAT) and visceral adipose tissue (VAT). In children and adolescents MRI is the most suitable measurement because it does not expose them to radiation.
Our present knowledge about the association between individual body fat depots and cardio-metabolic risk factors is mainly based on studies on adults. In addition, there is only limited knowledge about regional fat depots in children and adolescents. In a cross-sectional study SAT and VAT were lower in children compared to adults.7 In a longitudinal study, SAT increased during adolescence, whereas VAT remained low.8 By contrast, both, SAT and VAT, increased between age of 11 and 13 years.9 With regard to gender differences, girls compared with boys had greater masses of VAT and SAT.10 This was contrary to other authors who observed that VAT was greater in boys than in girls.11
In obese adolescents and adults cardiometabolic traits and insulin resistance have been mainly related to VAT and liver fat.12, 13, 14, 15, 16 In children and adolescents the degree of insulin resistance rises from pre- to intrapuberty and decreases after puberty.17, 18 However, the association between cardiometabolic traits and insulin resistance with detailed body composition has not been well characterized in children and adolescents. As in adults, SAT and VAT were differently associated with cardiometabolic traits in children and adolescents.19, 20, 21, 22 Both, SAT and VAT were independently correlated with blood pressure,19 whereas VAT was positively associated with plasma triglycerides (TG) and negatively associated with high-density lipoprotein cholesterol (HDL).20 In addition, there was a positive relationship between either VAT or SAT and insulin resistance.20, 21, 22, 23
Untill now, there is no systematic study of the associations between individual body fat depots and cardiometabolic risk in different age groups. The aim of this study was to determine the associations between MRI-derived individual fat depots and cardiometabolic traits in children and adolescents compared to young and older adults. Furthermore, we investigated the associations between insulin resistance, relative to BMI, and regional fat depots.
Methods
All studies had been approved by the Ethical Committee of Christian-Albrechts-University of Kiel, Germany, and conducted according to the guidelines laid down in the ‘Declaration of Helsinki’. Written informed consent was obtained from each subject and, in state of minority, from its legal guardian before participation.
Subjects
This investigation was a secondary analysis of data collected at the ‘Reference Center for Body Composition’ (Institute of Human Nutrition and Food Science at the University of Kiel, Germany) between 2005 and 2016. In this cross sectional study, data of 433 healthy Caucasian subjects (151 children and adolescents, aged 6–18 years, 150 young adults, aged 18–30 years and 132 adults, aged 30–60 years) were analyzed. Exclusion criteria were metallic implants, smoking, pregnancy, chronic or acute diseases, and medication intake that influences body composition. Subjects were recruited by notice boards postings and advertisements in the local newspaper. Study measurements of anthropometry, body composition, detailed body composition by whole body MRI and cardiometabolic variables were performed in children, adolescents and adults. Self-assessment of pubertal stage was determined according to the definition of Marshall and Tanner.24 On the basis of breast and genital stages pubertal status was categorized into three groups (prepubertal: Tanner I; intrapubertal: Tanner II–III, postpubertal: Tanner⩾IV).18 In this analysis prepubertal and intrapubertal subjects were grouped together. This was done because they did not differ in associations between body composition and cardiometabolic traits (data not shown), and because of a low number of prepubertal children (n=26).
Anthropometric measures
Body weight was measured to the nearest 0.01 kg using an electronic Tanita scale coupled to the BOD POD Body Composition Tracking System (Life Measurement Instruments, Concord, CA, USA). Body height was assessed to the nearest 0.5 cm by using a stadiometer (SECA, Modell 220, Hamburg, Germany). BMI was calculated as body weight (kg)/body height (m)2. To define overweight and obesity for children and adolescents (>90th percentile; >97th percentile) German age- and sex-specific references were used.25 Waist circumference (WC) was measured midway between the lowest rib and the top of the iliac crest with the subjects standing in upright position. The measurement was made with a nonelastic plastic tape placed parallel to the floor at the end of a normal expiration.
Densitometry
Air-displacement plethysmography was perfomed by BOD POD device to assess body composition. Child-specific equations were used to determine FM, as in detail described elsewhere.26 In adults, FM was calculated using the equation by Siri et al.27 FFM was then calculated from the difference between body weight and FM. Because of reported difficulties in BOD POD calibration in one of the children studies cross-validated gender specific equations of FM were developed for children and adolescents, as procedure is described elsewhere.28 Around 222 children and adolescents were randomly split into two parts; 155 (91 female, 64 male) were assigned to the derivation group, with 67 (31 female, 36 male) subjects in the cross-validation group. When compared to derivation group, the cross-validation group did not differ in age, gender, pubertal status, height, weight, BMI, WC, and FM measured by bio-impedance analysis. The correlation coefficients between FM measured by bio-impedance analysis and predicted FM based on BOD POD algorithm did not differ between the two groups. Following equations based on stepwise multiple regression analysis were developed for children and adolescents:


On the basis of calculated FM and FFM, FM-Index (FMI) and FFM-Index (FFMI) were calculated for children and adolescents as FM (kg)/height (m)n and FFM (kg)/height (m)n. Height-independent FM is confounded by age in adolescents, whereas age was not included as a significant independent variable in children. By contrast, height-independent FFM is confounded by age in children but not in adolescents. Age-adjusted power of height (n) was determined using log–log regression analysis29 (FMI: n=6.1, 2.3 and FFMI: n=2.3, 2.9, respectively for children and adolescents). In adults FMI and FFMI were calculated, as FM (kg)/height (m)2 and FFM (kg)/height (m)2.
Magnetic resonance imaging (MRI)
As described previously, detailed body composition of all subjects was performed by using whole-body MRI with a 1.5T scanner (Magnetom Vision or Magnetom Avanto, Siemens Medical Systems, Erlangen, Germany).30, 31 Briefly, participants were examined in a supine position with their arms extended above their heads and they were asked to hold their breath during measurement of abdominal and thoracic regions for image acquisition. In the present analysis volumes of SAT and VAT were manually segmented by using segmentation software (SliceOmatic 4.3 and 5.0, Tomovision, Montreal, Canada). SAT was analyzed from ankle to wrist. In this case SATlegs were defined from ankle to femoral heads and SATarms were defined from humerus heads to wrist. Between femoral and humerus heads volume of SATtrunk was assessed. VAT was defined as intra-abdominal fat between femoral heads and the top of the liver. Volumes of SAT and VAT were determined by the sum of all adipose tissue areas (cm2) multiplied by slice thickness. Volume data were then transformed into tissue masses by using the following density: 0.923 g cm−3 for SAT and VAT.
Cardiometabolic risk factors
Systolic and diastolic blood pressure were measured by manual sphygmomanometer in a seated position. After an 8-h overnight fast blood samples were taken and analyzed by standard procedures. HDL was assessed enzymatically by Konelab-20i-Analyzer (Konelab, Espoo, Finland). Triglyceride and plasma glucose were analyzed enzymatically using a Konelab-Test-Kit (Thermo Clinical Labsystems, Frankfurt, Germany). Plasma insulin was measured by radioimmunoassay (Adaltis, Freiburg, Germany). Insulin resistance was then calculated by homeostasis model assessment of insulin resistance (HOMA-IR):
HOMA-IR=plasma glucose (mmol/L)xplasma insulin (μU/ml)/22.5.32
Statistical analysis
Statistical analyses were performed with SPSS statistical software (SPSS 24.0, Inc., Chicago, Illinois, USA). All data are given as median and interquartil range because of not normally distributed data. Kruskal-wallis test with bonferroni correction was performed for more than two groups (pre- and intrapubertal subjects, postpubertal subjects, young adults and adults). To examine the association of individual body components on cardiometabolic traits, age- and gender-adjusted partial correlations were performed. Differences in correlation coefficients between age groups were tested by using the method of Eid, Gollwitzer, Schmitt.33 Further, stepwise multivariate linear regression analyses were done. Four models were conducted. Model 1 included FMI, Model 2 SATtrunk, Model 3 VAT and Model 4 SATtrunk and VAT as independent variables. All models were adjusted for age and gender. In addition, a descriptive approach was used to demonstrate age-dependent differences in individual body components between three different HOMA-IR per BMI groups (low, normal and high). These three groups were stratified according to their age dependent deviation from the regression line. Cut-offs were chosen from tertiles between HOMA-IR and BMI (low: residuals<−0.48,<−0.90,<−0.92,<−0.69; normal: residuals⩽0.16 and⩾−0.48,⩽0.25 and⩾−0.90,⩽0.00 and⩾−0.92,⩽0.29 and⩾−0.69; high: residuals>0.16,>0.25,>0.00, >0.29, respectively for children, adolescents, young adults, and adults). All tests were two-sided and level of significance was set at P<0.05.
Results
Detailed body composition: differences between age groups
The characteristics of the study population are shown in Table 1. When compared with children, height, weight, FM, and FFM were significantly higher in adolescents and with no significant differences to adults. When compared with young adults, VAT was lower in children and adolescents. In males, VAT was significantly higher in older compared with young adults. In females, SATtotal, SATarms, and SATtrunk were higher in young adults compared with children and adolescents. By contrast, SATtrunk was the only subcutaneous fat depot in males which was significantly higher in young adults compared to children and adolescents.
Cardiometabolic risks: Differences between age groups
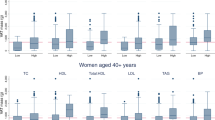
In females, plasma insulin and HOMA-IR were highest in young adults when compared to other age groups (Table 1). In males, plasma insulin and HOMA-IR were highest in adolescents. In both genders, HOMA-IR did not differ between adolescents and adults. There were no differences in SBP, DBP and HDL between the different age groups, except for SBP in male adolescents, and high TG level in female young adults and male adults.
Associations between body composition and cardiometabolic traits: Differences between age groups
Age- and gender-adjusted partial correlation coefficients between individual body fat depots and cardiometabolic traits are shown in Table 2. In general, correlation coefficients were low to moderate. As for SBP, DBP, TG and plasma glucose, significant associations were seen after puberty only. The associations between SATtrunk and either SBP or DBP or TG did not differ in adolescents, young adults and adults. Strongest associations were observed between plasma insulin levels or HOMA-IR and truncal fat depots (SATtrunk, VAT). When compared with adults, the associations between individual body fat depots and HDL were higher in children and adolescents.
Using stepwise multivariate linear regression analysis, the explained variance and respective beta coefficients of cardiometabolic traits as dependent variables and FMI, SATtrunk, VAT and the combination of SATtrunk and VAT, as independent variables are shown in Table 3. The explained variance of cardiometabolic traits was moderate, maximally, 35.2% of the variance of HOMA-IR was explained by SATtrunk and VAT in adults.
When compared with adolescents and adults, the explained variances of SBP, DBP, TG and plasma glucose by body fat depots were low (⩽5.3%) in children. However, the explained variances of HOMA-IR and plasma insulin by parameters of body composition differed only slightly between children, adolescents and adults. The explained variance of HDL by body fat depots decreased from children to adults.
When compared to FMI, the explained variance of plasma insulin and HOMA-IR by SATtrunk and VAT were moderately increased in young and older adults. When compared to FMI, SATtrunk and VAT did not exceed the explained variance of HDL, whereas abdominal fat depots added to the variance of TG in adolescents.
Association between detailed body composition and insulin resistance
Investigating subjects with low HOMA-IR (insulin sensitive), normal HOMA-IR or high HOMA-IR (insulin resistance) relative to their BMI, age-dependent differences in individual body components between these groups became evident (Table 4). In children and adolescents, no differences were found in body fat depots between groups differing in insulin resistance. By contrast, in young adults, adipose tissue, SATtotal, SATtrunk and SATlegs were significantly higher in insulin resistant subjects. In older adults, VAT was highest in insulin resistant subjects.
Discussion
Up to now, there are limited data on the association between regional fat depots and metabolic disturbance in normal- and overweight children and adolescents compared with adults.34, 35 To our knowledge, the present study is first to systematically investigate detailed body composition by age group and gender as well as its associations to cardiometabolic traits in 6–60 year old subjects.
Detailed body Composition in different age groups
In both genders, VAT was low in children and adolescents compared with adults (Table 1). This is in line with previous data.7, 8 However, other authors reported that during childhood, growth of VAT decelerated with increasing age.34 In our study sexual dimorphism was apparent after puberty only with males having more VAT than females. By contrast, other authors observed higher masses of VAT in girls compared to boys.10
Cardiometabolic risks in different age groups
As shown in Table 1, in males, insulin resistance increased from pre- and intrapuberty to postpuberty, with changes to young adulthood in females only. These findings support previous data.36, 37 By contrast, other authors reported that the increase in insulin resistance from childhood to adulthood was independent of gender.38 Studies on blood pressure showed that SBP and DBP increased with age.38, 39 These results are contrary to our findings.
Associations between body composition and cardiometabolic traits in different age groups
Low and moderate associations were observed between cardiometabolic traits and body fat depots in normal- and overweight children, adolescents and adults (Table 2). This is in line with previous data.38 However, moderate associations were seen after puberty only. These findings indicate that in pre- and intrapubertal children individual body components are not related to cardiometabolic risk, as it is in older subjects. Accordingly, a study on pre- and intrapubertal children found no associations between abdominal fat depots vs blood pressure and blood lipids in European Americans.40 Positive associations became evident between SATtrunk, VAT, and cardiometabolic traits in adolescents.19
Overall, there was a limited explained variance of cardiometabolic traits by FMI and different regional fat depots (Table 3). When compared to FMI, abdominal fat depots, especially VAT, were stronger associated to insulin resistance in adults than in children and adolescents. The associations between HDL and body fat depots decreased with age. As HDL is positively associated to physical activity in children, adolescents and adults,41, 42 a possible explanation may be increasing sedentary behavior with age which is due to school entry and entering the workplace.43, 44 This is in line with the finding that children improved HDL by increased physical activity which was independent of weight change.45
Associations between detailed body composition and insulin resistance
To address the role of individual body fat depots on insulin resistance, independent of BMI, differences in age groups became evident (Table 4). In children and adolescents, there were no differences in body fat depots between groups differing in insulin resistance. By contrast, regional SAT depots were significantly different in young adults, with VAT as associated to insulin resistance in older adults only. Our findings suggest that VAT becomes an important determinant of insulin resistance as age increased to adulthood. In our study, VAT was very low in children and adolescents and started to increase in young adults (Table 1). This is in line with other data that VAT is a primary determinant of insulin resistance in adults only.46, 47, 48
Study strengths and limitations
To our knowledge, this is the first study to systematically investigate age-related associations between individual body components and cardiometabolic traits in children and adolescents compared to adults. Detailed body composition was measured by MRI. This is the gold-standard method of body composition. We have used two different approaches. First, an analytic approach was chosen to examine age-dependent associations between cardiometabolic traits and body fat depots. Second, a descriptive approach was conducted to investigate age-dependent differences in body fat depots in groups differing in insulin resistance. A limitation of this study is that we have used cross-sectional data only. Thus, no causal relationships can be derived from our data.
Conclusion
Associations between regional fat depots and cardiometabolic traits were seen after puberty only. The impact of FMI on cardiometabolic traits is limited. With regard to HOMA-IR, this was improved by measuring individual fat depots in adults. The strongest associations were observed between abdominal fat depots and insulin resistance. When compared with adults, VAT was low in normal- and overweight children and adolescents. VAT has no association with insulin resistance groups in children, adolescents and young adults. The role of VAT is apparent in adults only.
References
Janssen I, Katzmarzyk PT, Srinivasan SR, Chen W, Malina RM, Bouchard C et al. Utility of childhood BMI in the prediction of adulthood disease: comparison of national and international references. Obes Res 2005; 13: 1106–1115.
Goodman E, Dolan LM, Morrison JA, Daniels SR . Factor analysis of clustered cardiovascular risks in adolescence: obesity is the predominant correlate of risk among youth. Circulation 2005; 111: 1970–1977.
Bluher S, Molz E, Wiegand S, Otto K-P, Sergeyev E, Tuschy S et al. Body mass index, waist circumference, and waist-to-height ratio as predictors of cardiometabolic risk in childhood obesity depending on pubertal development. J Clin Endocrinol Metab 2013; 98: 3384–3393.
Wells JCK . The Evolutionary Biology of Human Body Fatness: Thrift and Control 1st edn. Cambridge University Press: Cambridge, UK, 2010.
Prentice AM, Jebb SA . Beyond body mass index. Obes Rev 2001; 2: 141–147.
Müller MJ, Braun W, Enderle J, Bosy-Westphal A . Beyond BMI: conceptual issues related to overweight and obese patients. Obes Facts 2016; 9: 193–205.
Shen W, Punyanitya M, Silva AM, Chen J, Gallagher D, Sardinha LB et al. Sexual dimorphism of adipose tissue distribution across the lifespan: a cross-sectional whole-body magnetic resonance imaging study. Nutr Metab (Lond) 2009; 6: 17.
Brambilla P, Manzoni P, Agostini G, Beccaria L, Ruotolo G, Sironi S et al. Persisting obesity starting before puberty is associated with stable intraabdominal fat during adolescence. Int J Obes Relat Metab Disord 1999; 23: 299–303.
Fox KR, Peters DM, Sharpe P, Bell M . Assessment of abdominal fat development in young adolescents using magnetic resonance imaging. Int J Obes Relat Metab Disord 2000; 24: 1653–1659.
Benfield LL, Fox KR, Peters DM, Blake H, Rogers I, Grant C et al. Magnetic resonance imaging of abdominal adiposity in a large cohort of British children. Int J Obes (Lond) 2008; 32: 91–99.
Staiano AE, Broyles ST, Gupta AK, Katzmarzyk PT . Ethnic and sex differences in visceral, subcutaneous, and total body fat in children and adolescents. Obesity 2013; 21: 1251–1255.
Gyllenhammer LE, Alderete TL, Toledo-Corral CM, Weigensberg M, Goran MI . Saturation of subcutaneous adipose tissue expansion and accumulation of ectopic fat associated with metabolic dysfunction during late and post-pubertal growth. Int J Obes (Lond) 2016; 40: 601–606.
Kelly AS, Dengel DR, Hodges J, Zhang L, Moran A, Chow L et al. The relative contributions of the abdominal visceral and subcutaneous fat depots to cardiometabolic risk in youth. Clin Obes 2014; 4: 101–107.
Kim SK, Kim HJ, Hur KY, Choi SH, Ahn CW, Lim SK et al. Visceral fat thickness measured by ultrasonography can estimate not only visceral obesity but also risks of cardiovascular and metabolic diseases. Am J Clin Nutr 2004; 79: 593–599.
King RJ, Ajjan RA . Vascular risk in obesity: facts, misconceptions and the unknown. Diab Vasc Dis Res 2017; 14: 2–13.
Yatsuya H, Nihashi T, Li Y, Hotta Y, Matsushita K, Muramatsu T et al. Independent association of liver fat accumulation with insulin resistance. Obes Res Clin Pract 2014; 8: e350–e355.
Tobisch B, Blatniczky L, Barkai L . Cardiometabolic risk factors and insulin resistance in obese children and adolescents: relation to puberty. Pediatr Obes 2015; 10: 37–44.
Reinehr T, Wolters B, Knop C, Lass N, Holl RW . Strong effect of pubertal status on metabolic health in obese children: a longitudinal study. J Clin Endocrinol Metab 2015; 100: 301–308.
Kim JA, Park HS . Association of abdominal fat distribution and cardiometabolic risk factors among obese Korean adolescents. Diabetes Metab 2008; 34: 126–130.
Owens S, Gutin B, Ferguson M, Allison J, Karp W, Le NA . Visceral adipose tissue and cardiovascular risk factors in obese children. J Pediatr 1998; 133: 41–45.
Syme C, Abrahamowicz M, Leonard GT, Perron M, Pitiot A, Qiu X et al. Intra-abdominal adiposity and individual components of the metabolic syndrome in adolescence: sex differences and underlying mechanisms. Arch Pediatr Adolesc Med 2008; 162: 453–461.
He F, Rodriguez-Colon S, Fernandez-Mendoza J, Vgontzas AN, Bixler EO, Berg A et al. Abdominal obesity and metabolic syndrome burden in adolescents—Penn State Children Cohort study. J Clin Densitom 2015; 18: 30–36.
Maffeis C, Manfredi R, Trombetta M, Sordelli S, Storti M, Benuzzi T et al. Insulin sensitivity is correlated with subcutaneous but not visceral body fat in overweight and obese prepubertal children. J Clin Endocrinol Metab 2008; 93: 2122–2128.
Duke PM, Litt IF, Gross RT . Adolescents' self-assessment of sexual maturation. Pediatrics 1980; 66: 918–920.
Kromeyer-Hauschild K, Wabitsch M, Kunze D, Geller F, Geiß HC, Hesse V et al. Perzentile für den Body-mass-Index für das Kindes- und Jugendalter unter Heranziehung verschiedener deutscher Stichproben. Monatsschrift Kinderheilkunde 2001; 149: 807–818.
Bosy-Westphal A, Danielzik S, Becker C, Geisler C, Onur S, Korth O et al. Need for optimal body composition data analysis using air-displacement plethysmography in children and adolescents. J Nutr 2005; 135: 2257–2262.
Siri WE . Body composition from fluid spaces and density: analysis of methods. 1961. Nutrition 1993; 9: 480–491 discussion 480, 492.
Huang TT-K, Watkins MP, Goran MI . Predicting total body fat from anthropometry in Latino children. Obes Res 2003; 11: 1192–1199.
Wells JCK, Cole TJ . Adjustment of fat-free mass and fat mass for height in children aged 8 y. Int J Obes Relat Metab Disord 2002; 26: 947–952.
Bosy-Westphal A, Booke C-A, Blocker T, Kossel E, Goele K, Later W et al. Measurement site for waist circumference affects its accuracy as an index of visceral and abdominal subcutaneous fat in a Caucasian population. J Nutr 2010; 140: 954–961.
Schautz B, Later W, Heller M, Muller MJ, Bosy-Westphal A . Total and regional relationship between lean and fat mass with increasing adiposity—impact for the diagnosis of sarcopenic obesity. Eur J Clin Nutr 2012; 66: 1356–1361.
Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC . Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985; 28: 412–419.
Eid M, Gollwitzer M, Schmitt M . Statistik und Forschungsmethoden: Lehrbuch; mit Online-Materialien 2nd edn. Beltz: Weinheim, 2011.
Huang TT, Johnson MS, Figueroa-Colon R, Dwyer JH, Goran MI . Growth of visceral fat, subcutaneous abdominal fat, and total body fat in children. Obes Res 2001; 9: 283–289.
Johnson MS, Figueroa-Colon R, Huang TT, Dwyer JH, Goran MI . Longitudinal changes in body fat in African American and Caucasian children: influence of fasting insulin and insulin sensitivity. J Clin Endocrinol Metab 2001; 86: 3182–3187.
Raab J, Haupt F, Kordonouri O, Scholz M, Wosch A, Ried C et al. Continuous rise of insulin resistance before and after the onset of puberty in children at increased risk for type 1 diabetes–a cross-sectional analysis. Diabetes Metab Res Rev 2013; 29: 631–635.
Ballerini MG, Bergada I, Rodriguez ME, Keselman A, Bengolea VS, Pipman V et al. Concentracion de insulina e indices de insulinosensibilidad en ninos y adolescentes sanos. Arch Argent Pediatr 2016; 114: 329–336.
Ali O, Cerjak D, Kent JW JR, James R, Blangero J, Zhang Y . Obesity, central adiposity and cardiometabolic risk factors in children and adolescents: a family-based study. Pediatr Obes 2014; 9: e58–e62.
Wills AK, Lawlor DA, Matthews FE, Sayer AA, Bakra E, Ben-Shlomo Y et al. Life course trajectories of systolic blood pressure using longitudinal data from eight UK cohorts. PLoS Med 2011; 8: e1000440.
Casazza K, Dulin-Keita A, Gower BA, Fernandez JR . Intrabdominal fat is related to metabolic risk factors in Hispanic Americans, African Americans and in girls. Acta Paediatr 2009; 98: 1965–1971.
Heshmat R, Qorbani M, Shahr Babaki AE, Djalalinia S, Ataei-Jafari A, Motlagh ME et al. Joint Association of Screen Time and Physical Activity with Cardiometabolic Risk Factors in a National Sample of Iranian Adolescents: The CASPIANIII Study. PLoS One 2016; 11: e0154502.
Bakrania K, Edwardson CL, Bodicoat DH, Esliger DW, Gill JMR, Kazi A et al. Associations of mutually exclusive categories of physical activity and sedentary time with markers of cardiometabolic health in English adults: a cross-sectional analysis of the Health Survey for England. BMC Public Health 2016; 16: 25.
von Kries R, Beyerlein A, Muller MJ, Heinrich J, Landsberg B, Bolte G et al. Different age-specific incidence and remission rates in pre-school and primary school suggest need for targeted obesity prevention in childhood. Int J Obes (Lond) 2012; 36: 505–510.
Mackenzie K, Goyder E, Eves F . Acceptability and feasibility of a low-cost, theory-based and co-produced intervention to reduce workplace sitting time in desk-based university employees. BMC Public Health 2015; 15: 1294.
Lee YH, Song YW, Kim HS, Lee SY, Jeong HS, Suh S-H et al. The effects of an exercise program on anthropometric, metabolic, and cardiovascular parameters in obese children. Korean Circ J 2010; 40: 179–184.
Faria AN, Ribeiro Filho FF, Gouveia Ferreira SR, Zanella MT . Impact of visceral fat on blood pressure and insulin sensitivity in hypertensive obese women. Obes Res 2002; 10: 1203–1206.
Matsuzawa Y, Shimomura I, Nakamura T, Keno Y, Kotani K, Tokunaga K . Pathophysiology and pathogenesis of visceral fat obesity. Obes Res 1995; 3 (Suppl 2): 187S–194S.
Phillips GB, Jing T, Heymsfield SB . Relationships in men of sex hormones, insulin, adiposity, and risk factors for myocardial infarction. Metabolism 2003; 52: 784–790.
Acknowledgements
We thank Britta Jux and the Clinic for Diagnostic Radiology, University Medical Center Schleswig-Holstein, Kiel (Germany) for the help with MRI scanning. We acknowledge financial support by Land Schleswig-Holstein within the funding programme Open Access Publikationsfonds. This work was supported by the Deutsche Forschungsgemeinschaft (DFG), the Federal Ministry of Education and Research (BMBF) (DFG Mü 714/8-3; DFG Bo 3296/1-1; BMBF 01EA1336, 01GI1121A, 01GI1125, 01GI0821) and OMRON, Kyoto, Japan.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Hübers, M., Geisler, C., Plachta-Danielzik, S. et al. Association between individual fat depots and cardio-metabolic traits in normal- and overweight children, adolescents and adults. Nutr. Diabetes 7, e267 (2017). https://doi.org/10.1038/nutd.2017.20
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nutd.2017.20
This article is cited by
-
Determinants of bone mass in older adults with normal- and overweight derived from the crosstalk with muscle and adipose tissue
Scientific Reports (2023)
-
Changes in overall and regional body fatness from childhood to early adolescence
Scientific Reports (2019)
-
Associations of adult genetic risk scores for adiposity with childhood abdominal, liver and pericardial fat assessed by magnetic resonance imaging
International Journal of Obesity (2018)
-
Body composition and insulin resistance in children
European Journal of Clinical Nutrition (2018)