Abstract
Background:
African-Americans have higher rates of obesity-associated chronic diseases. Serum 25-hydroxyvitamin D (25(OH)D) shows an inverse association with obesity status. We investigated whether vitamin D supplementation changes body mass index (BMI).
Subjects:
In total, 328 overweight African-Americans were enrolled over three consecutive winter periods (2007–2010) into a randomized, double-blind, placebo-controlled trial to receive cholecalciferol supplementation (0, 1000 international units (IU), 2000 IU or 4000 IU per day) for 3 months. Plasma concentrations of 25(OH)D and anthropometric measurements were done at baseline, 3 and 6 months.
Results:
At 3 months, vitamin D supplementation in three dose groups (1000 IU, 2000 IU or 4000 IU per day) did not cause any significant changes in BMI as compared with placebo group 3-month change in BMI per 1000 IU per day estimate (SE): 0.01 (0.039); P=0.78.
Conclusions:
In overweight African-Americans, short-term high-dose vitamin D supplementation did not alter BMI.
Similar content being viewed by others
Introduction
An urgent need exists to identify modifiable dietary risk factors for obesity in African-Americans. Human observations since the 1980’s of lower levels of 25-hydroxyvitamin D25(OH)D in obese than in nonobese individuals highlight a possible inverse relation between vitamin D and obesity.1
African-Americans have consistent associations of vitamin D deficiency with obesity and obesity-associated chronic diseases, such as hypertension, coronary heart disease, diabetes and certain cancers,2 although of potentially lower magnitude than Whites.3, 4 Furthermore, African-Americans have lower vitamin D and calcium dietary intake.5 The mechanisms underlying this association may include a role for 25(OH)D in regulating lipid metabolism in adipose cells. In vivo studies suggest that vitamin D and calcium administration increase fatty-acid oxidation and decrease lipogenesis.6, 7
Results from clinical trials measuring the effect of vitamin D supplementation on obesity are limited and mixed. Some trials show no association of vitamin D supplementation with weight loss.8, 9, 10 Other trials show that vitamin D supplementation may be significantly associated with less weight gain but that this association may be dependent on adjunctive calcium supplementation and a particular region of fat.11, 12 A recent meta-analysis showed no significant effect of vitamin D supplementation on body mass index (BMI), weight or other adiposity measures.13 This meta-analysis did not assess potential ethnic/racial differences in the effect of vitamin D supplementation on adiposity measures. Studies evaluating the effect of vitamin D supplementation on measures such as insulin resistance in African-Americans14 have not reported on changes in adiposity measures. Thus, this ancillary analysis was undertaken to test the hypothesis that supplementation with vitamin D3 (cholecalciferol) leads to weight loss in African-Americans.
Materials and methods
Study design and participants
This is a prospective, randomized, double-blind, placebo-controlled clinical trial of oral cholecalciferol (vitamin D3) in a community-based overweight African-American population (ClinicalTrials.gov NCT00585637). The protocol has been described in detail elsewhere.15 The primary goal of the trial was to examine the effect of daily supplementation of 1000 international units (IU) of vitamin D3, 2000 IU of vitamin D3 and 4000 IU of vitamin D3 and placebo on plasma 25(OH)D levels. All participants provided written informed consent. This trial focused on African-Americans because African-Americans have higher rates of vitamin D deficiency compared with Whites5 or Africans.16 The project was approved by the Institutional Review Boards of Harvard School of Public Health and Dana-Farber Cancer Institute. All procedures were followed in accordance with institutional guidelines.
Recruitment and randomization
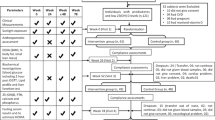
Participants in Open Doors to Health, a colorectal cancer prevention intervention study conducted in 12 public housing communities in the Boston metropolitan area,17 were invited to participate if they were 30–80 years old, understood written and spoken English, self-identified as Black18, 19, 20 and had permission from their primary care doctors. We also recruited participants from community and faith-based organizations and refer-a-friend program. A total of 328 individuals were enrolled into the parent trial (Figure 1). Exclusion criteria included pregnancy, renal disease, pre-existing parathyroid, thyroid, or calcium metabolism disorders, sarcoidosis, requirement for calcium channel blockers, type I diabetes and active malignancies (other than non-melanoma skin cancer). Those taking vitamin D supplementation were enrolled if they agreed to discontinue these medications for 6 months prior to enrollment and during the study.
Treatment
Participants were assigned to four arms consisting of placebo, 1000 IU per day, 2000 IU per day or 4000 IU per day of vitamin D3 for 3 months in a 1:1:1:1 ratio using block randomization stratified by age, sex and enrollment month. Study statisticians generated the random allocation sequence and subjects were enrolled by research assistants. All capsules also contained 200 mg of calcium carbonate (Pharmavite LLC, Mission Hill, CA, USA). Calcium was included because prior studies have shown that African-Americans have low calcium intake.21 All capsules were indistinguishable, and both participants and research staff were blinded to treatment assignment. Study medications were started in early winter (November or December) and were taken orally once daily for 3 months (completed in February or March).
Compliance and safety
All participants were assessed for adverse events by study staff over the phone at week 2 of each month and in-person at the beginning of each month when the next month’s supply of vitamins was provided. Participants were educated on the warning signs and symptoms of hypercalcemia. Serum calcium was measured in subjects taking hydrochlorothiazide at 1 and 3 months. An additional subset of 44 ‘control’ participants not taking hydrochlorothiazide also underwent calcium assays at 3 months. Electronic pill dispenser systems and pill counts were used to track compliance with study supplementation in addition to biweekly phone calls. Any subject found to have calcium >2.62 mmol l−1 (10.5 mg dl−1) was immediately discontinued from the study and the primary care physician was notified.
Measurement of 25(OH)D levels
Blood samples collected at baseline, 3 and 6 months were separated, and plasma was stored in liquid nitrogen in the Dana-Farber Cancer Institute Clinical Research Laboratory (Boston, MA, USA). Once study was completed, all plasma samples that were sent as a single batch to the laboratory of Dr Bruce Hollis (Medical University of South Carolina, Charleston, SC, USA) were 25(OH)D concentrations were measured using the Diasorin (DiaSorin, Inc., Stillwater, MN, USA) radioimmunoassay.22 Masked quality control samples were interspersed among the cases and all laboratory personnel were blinded. The mean coefficient of variation of 25(OH)D measurements was 9%.
Anthropometric measurements
Weight was measured using electronic digital scales to the nearest 0.2 kg. Height was measured using a wall-mounted stadiometer. BMI was subsequently calculated as weight (kg) per height (m2).
Dietary measurements and physical activity
Dietary and calcium intake was assessed at baseline and at 3 months with an abbreviated form of the semiquantitative Food Frequency Questionnaire.23, 24 The responses were recoded to equivalent servings per week and summed to obtain total servings of calcium-rich food groups and vitamin D-rich food groups per week. Dietary vitamin D per serving included (100 IU for milk and soymilk; 400 IU for multivitamin or vitamin D supplement, 100 IU for ice cream, 100 IU for yogurt, 560 IU for dark fish, 100 IU for fortified orange juice and 100 IU for cold cereal).
Statistical analysis
The primary end point for this study was the change in BMI and weight for the participants from 0 to 3 months. We analyzed the change within each dose group from month 0 to month 3 using linear regression. Statistical power for this trial was based on the intent-to-treat population of 80 subjects per arm. Using a two-sided t-test at the 0.05 significance level, the minimum detectable difference in 25(OH)D between treatment arms was 13.2 and 15.5 nmol l−1 (5.3 and 6.2 ng ml−1) with 80% and 90% power, respectively. We estimated that the minimum detectable difference in weight between treatment arms was 4 kg and 6 kg with 80% and 90% power, respectively. All statistical analyses were performed using SAS 9.2 (SAS Institute, Cary, NC, USA).
Results
Subject characteristics according to supplementation arm
Baseline characteristics for the 328 randomized participants are presented in Table 1. The participants had moderately active lifestyles. Mean exercise frequency was 3 times per week. Approximately 32 (9.8%), 24 (7.3%) and 65 (19.8%) participants were taking supplemental calcium, vitamin D and multivitamin, respectively, at baseline. Approximately 99 (30.2%) of the participants were current smokers. More participants in the placebo and 1000 IU per day arms had a past history of cancer than those assigned to 2000 or 4000 IU per day. Otherwise, there were no significant differences in any of the subject characteristics between the supplementation arms. Compliance was 96.6% across the cohort.
Impact of vitamin D3 supplementation on plasma 25(OH)D
Among 328 subjects, 25(OH)D level at baseline was median interquartile range (IQR):15.3 ng ml−1 (12.5) with 25(OH)D >20 ng ml−1 in 34.3% of participants. The effect of the various doses of vitamin D3 supplementation on 25(OH)D levels after 3 months is shown in Table 2. At 3 months, median 25(OH)D levels rose to 29.7, 34.8 and 45.9 ng ml−1, respectively, in participants assigned to 1000, 2000 and 4000 IU per day. After 3 months of vitamin D3 supplementation, 292 subjects (89%) provided plasma for follow-up 25(OH)D determination. The compliance rate with randomized therapy in the whole cohort was 96.6%.
Association of baseline 25(OH)D and BMI and weight
There was no significant association between BMI or weight and 25(OH)D at the beginning of the study. Baseline weight and baseline BMI were not significantly associated with 25(OH)D (estimate (SE), P-value: BMI, −0.04 (0.07), P=0.55; weight, −0.02 (0.01), P=0.06). Baseline 25(OH)D was not significantly associated with baseline BMI or baseline weight (BMI, −0.03 (0.05), P=0.55; weight, −0.56 (0.30), P=0.06).
Impact of vitamin D supplementation on BMI and weight
The primary efficacy analyses of vitamin D3 supplementation on weight and BMI are shown in Table 2. In linear regression with the dose of vitamin D3 (per 1000 IU per day) as the independent variable and the 3-month change in weight (or 3-month change in BMI) as the dependent variable, no effect of vitamin D3 on change in BMI or weight was observed. For each additional 1000 IU per day of vitamin D3, BMI increased by estimate (SE): 0.01(0.039); P=0.78 and weight increased by estimate (SE):0.06 (0.24); P=0.81. At 6 months, no significant change in BMI or weight was observed (Table 2).
Adverse events
There were five isolated incidences of mild hypercalcemia that were in the reference range on repeated sampling. Vitamin D supplements were discontinued in the four participants with mild hypercalcemia at 1 month. Vitamin D supplementation was discontinued in the participant who had hypercalcemia at month 3. There were no episodes of nephrolithiasis.
Discussion
On the basis of evidence showing that vitamin D deficiency is associated with obesity, we tested the hypothesis that vitamin D supplementation could reduce BMI in a randomized, placebo-controlled trial of African-Americans. Although circulating 25(OH)D increased with vitamin D supplementation, we found that vitamin D supplementation did not lower BMI or weight among healthy African-Americans. Numerous cross-sectional studies have assessed the relationship between vitamin D status and different measures of adiposity.2 These studies found an inverse relationship between vitamin D and total body fat and regional adipose tissue.2 Others have shown an inverse relationship between vitamin D levels and components of metabolic syndrome, including abdominal obesity,25 which is a known risk factor for several chronic conditions. Furthermore, a recent meta-analysis evaluating the effect of vitamin D supplementation on adiposity biomarkers in randomized controlled trials found no significant reduction in adiposity measures in the absence of calorie restriction.13
The lack of effect of vitamin D supplementation on weight may be due to low dietary calcium intake. Heaney et al.,26 concluded that vitamin D status and benefits associated with vitamin D supplementation appear to be dependent on calcium intakes at or above recommended amounts. The doses of vitamin D tested in this study, as well as the dosing schedule (once per day, rather than intermittent large boluses), may provide an optimal balance of efficacy and safety based on current evidence and the recommended daily allowance and tolerable upper limit set by the Institute of Medicine.27
Although there have been some trials including Whites that suggest that vitamin D supplementation prevents weight gain or promotes weight loss, little is known about African-Americans.28 Some have shown positive effects on weight but these effects may have been due to the adjunctive calcium supplementation. For example, in the Women’s Health Initiative Study,12 women who were randomly assigned to calcium and vitamin D supplementation arm had significantly less weight gain. Although the overall mean weight change difference between groups was small (−0.13 kg), women in the active intervention who had inadequate baseline calcium intakes (<1200 mg per day) had 11% lower risks for substantial weight gain (1–3 kg or >3 kg), whereas those who had calcium intakes >1200 mg per day were unaffected by treatment (P for interaction=0.008).
The vitamin D/calcium trial was imbedded in the other Women's Health Initiative randomized controlled trials, including a diet modification trial that may have led to weight loss, thus influencing the effects of combined vitamin D/calcium supplementation. It was not possible to differentiate between the effect of calcium and vitamin D supplementation. Major et al.,29 showed that among women with calcium intake <800 mg per day, calcium and vitamin D supplementation enhanced the beneficial effect of body weight loss on lipids and lipoprotein profile in over weight and obese women.
In our study, vitamin D supplementation was not associated with any significant adverse effects. This confirms the documentation by others that even higher doses of vitamin D3 supplements are safe.30 Strengths of our study include its prospective design, the use of a double-blind, randomized placebo-controlled intervention and our adjustment for season. Another strength is the similarity of living environments of these urban African-Americans. Relative homogeneity of the population is likely to reduce the influence of confounding factors on the measurement of the vitamin D effect. We considered seasonal changes in vitamin D and body composition by limiting supplementation of participants to the winter months.
Limitations of our study include the relatively short duration of vitamin D supplementation. Yet, this targeted population of obese African-Americans allowed for short duration and small sample size with adequate power to test the hypothesis that vitamin D supplementation reduces BMI. As this was an ancillary analysis, we did not collect other anthropometric data, such as waist or hip circumference or arm fat index. Our primary outcome was change in weight and BMI. These are weaker measures of adiposity than direct measures. Thus, we may be underestimating the association between vitamin D supplementation and adiposity.31 Yet, our finding agree with other published null findings of the effects of vitamin D supplementation on adiposity biomarkers.13 We also recognize that low dietary calcium intake may have contributed to our results. This study is unique in that it evaluates vitamin D supplementation effects in African-Americans with a low intake of calcium.
Other limitations of the trial compared with some trials specifically designed for obesity is the lack of records of energy intake and detailed measurements of physical activity. Small changes in BMI may not be detected because of the small sample size and the measurements of adiposity may not be sufficiently sensitive. BMI includes fat mass, lean mass and bone mass. Furthermore, a variety of hormones including gonadal, thyroidal, adrenal and growth hormones influence adiposity. These hormones are under the influence of the hypothalamic-pituitary axis and have circadian and circannual rhythms in adults.32 Thus, the association between obesity and 25(OH)D status may be related to other hormones that change by season.
In conclusion, supplementation with vitamin D did not significantly alter the weight or the BMI over a relatively short duration in a cohort of overweight or obese African-Americans. It is not known whether the low serum 25(OH)D in African-Americans is due to vitamin D insufficiency secondary to the increase in fat mass or to other factors such as genotype variation in vitamin D binding protein or enzymes involved in vitamin D metabolism. Future trials of longer vitamin D supplementation in African-Americans are needed to examine the biological contributors to the interaction between vitamin D deficiency, obesity, health disparities and obesity-related chronic diseases.
References
Bell NH, Epstein S, Greene A, Shary J, Oexmann MJ, Shaw S . Evidence for alteration of the vitamin D-endocrine system in obese subjects. J Clin Invest 1985; 76: 370–373.
Soares MJ, Chan She Ping-Delfos W, Ghanbari MH . Calcium and vitamin D for obesity: a review of randomized controlled trials. Eur J Clin Nutr 2011; 65: 994–1004.
Nesby-O'Dell S, Scanlon KS, Cogswell ME, Gillespie C, Hollis BW, Looker AC et al. Hypovitaminosis D prevalence and determinants among African American and white women of reproductive age: third National Health and Nutrition Examination Survey, 1988-1994. Am J Clin Nutr 2002; 76: 187–192.
Scragg R, Sowers M, Bell C . Serum 25-hydroxyvitamin D, ethnicity, and blood pressure in the Third National Health and Nutrition Examination Survey. Am J Hypertens 2007; 20: 713–719.
Yetley EA . Assessing the vitamin D status of the US population. Am J Clin Nutr 2008; 88: 558S–564S.
Caron-Jobin M, Morisset AS, Tremblay A, Huot C, Legare D, Tchernof A . Elevated serum 25(OH)D concentrations, vitamin D, and calcium intakes are associated with reduced adipocyte size in women. Obesity 2011; 19: 1335–1341.
Soares MJ, Murhadi LL, Kurpad AV, Chan She Ping-Delfos WL, Piers LS . Mechanistic roles for calcium and vitamin D in the regulation of body weight. Obes Rev 2012; 13: 592–605.
Sneve M, Figenschau Y, Jorde R . Supplementation with cholecalciferol does not result in weight reduction in overweight and obese subjects. Eur J Endocrinol 2008; 159: 675–684.
Zittermann A, Frisch S, Berthold HK, Gotting C, Kuhn J, Kleesiek K et al. Vitamin D supplementation enhances the beneficial effects of weight loss on cardiovascular disease risk markers. Am J Clin Nutr 2009; 89: 1321–1327.
Holecki M, Zahorska-Markiewicz B, Wiecek A, Mizia-Stec K, Nieszporek T, Zak-Golab A . Influence of calcium and vitamin D supplementation on weight and fat loss in obese women. Obes Facts 2008; 1: 274–279.
Zhou J, Zhao LJ, Watson P, Zhang Q, Lappe JM . The effect of calcium and vitamin D supplementation on obesity in postmenopausal women: secondary analysis for a large-scale, placebo controlled, double-blind, 4-year longitudinal clinical trial. Nutr Metab 2010; 7: 62.
Caan B, Neuhouser M, Aragaki A, Lewis CB, Jackson R, LeBoff MS et al. Calcium plus vitamin D supplementation and the risk of postmenopausal weight gain. Arch Int Med 2007; 167: 893–902.
Pathak K, Soares MJ, Calton EK, Zhao Y, Hallett J . Vitamin D supplementation and body weight status: a systematic review and meta-analysis of randomized controlled trials. Obes Rev 2014; 15: 528–537.
Harris SS, Pittas AG, Palermo NJ . A randomized, placebo-controlled trial of vitamin D supplementation to improve glycaemia in overweight and obese African Americans. Diabetes Obes Metab 2012; 14: 789–794.
Ng K, Scott JB, Drake BF, Chan AT, Hollis BW, Chandler PD et al. Dose response to vitamin D supplementation in African Americans: results of a 4-arm, randomized, placebo-controlled trial. Am J Clin Nutr 2013; 24.
Durazo-Arvizu RA, Aloia JF, Dugas LR, Tayo BO, Shoham DA, Bertino AM et al. 25-hydroxyvitamin D levels in African American and Nigerian women. Am J Hum Biol 2013; 25: 560–562.
McNeill LH, Coeling M, Puleo E, Suarez EG, Bennett GG, Emmons KM . Colorectal cancer prevention for low-income, sociodemographically-diverse adults in public housing: baseline findings of a randomized controlled trial. BMC Public Health 2009; 9: 353.
U.S. Bureau of the Census Overview of Race and Hispanic Origin. Census 2000 Brief. U.S. Bureau of the Census: Washington, DC, USA, 2001.
McKenney NR, Bennett CE . Issues regarding data on race and ethnicity: the Census Bureau experience. Public Health Rep 1994; 109: 16–25.
Williams DR . Race/ethnicity and socioeconomic status: measurement and methodological issues. Int J Health Serv 1996; 26: 483–505.
Aloia JF . African Americans, 25-hydroxyvitamin D, and osteoporosis: a paradox. Am J Cin Nutr 2008; 88: 545S–550S.
Hollis BW . Quantitation of 25-hydroxyvitamin D and 1,25-dihydroxyvitamin D by radioimmunoassay using radioiodinated tracers. Methods Enzymol 1997; 282: 174–186.
Willett WC, Sampson L, Stampfer MJ, Rosner B, Bain C, Witschi J et al. Reproducibility and validity of a semiquantitative food frequency questionnaire. Am J Epidemiol 1985; 122: 51–65.
Rimm EB, Giovannucci EL, Stampfer MJ, Colditz GA, Litin LB, Willett WC . Reproducibility and validity of an expanded self-administered semiquantitative food frequency questionnaire among male health professionals. Am J Epidemiol 1992; 135: 1114–1126.
Martini LA, Wood RJ . Vitamin D status and the metabolic syndrome. Nutr Rev 2006; 64: 479–486.
Heaney RP . Vitamin D and calcium interactions: functional outcomes. Am J Clin Nutr 2008; 88: 541S–544S.
Ross AC, Manson JE, Abrams SA, Aloia JF, Brannon PM, Clinton SK et al. The 2011 report on dietary reference intakes for calcium and vitamin D from the Institute of Medicine: what clinicians need to know. J Clin Endocrinol Metab 2011; 96: 53–58.
Valina-Toth AL, Lai Z, Yoo W, Abou-Samra A, Gadegbeku CA, Flack JM . Relationship of vitamin D and parathyroid hormone with obesity and body composition in African Americans. Clin Endocrinol 2010; 72: 595–603.
Major GC, Alarie F, Dore J, Phouttama S, Tremblay A . Supplementation with calcium + vitamin D enhances the beneficial effect of weight loss on plasma lipid and lipoprotein concentrations. Am J Clin Nutr 2007; 85: 54–59.
Vieth R . Vitamin D supplementation, 25-hydroxyvitamin D concentrations, and safety. Am J Clin Nutr 1999; 69: 842–856.
Young KA, Engelman CD, Langefeld CD, Hairston KG, Haffner SM, Bryer-Ash M et al. Association of plasma vitamin D levels with adiposity in Hispanic and African Americans. J Clin Endocrinol Metab 2009; 94: 3306–3313.
Nicolau GY, Haus E, Lakatua D, Bogdan C, Petrescu E, Robu E et al. Circadian time structure of endocrine and biochemical parameters in adult onset (type II) diabetic patients. Endocrinologie 1984; 22: 227–243.
Acknowledgements
We would like to thank Cara Marcus, MSLIS, AHIP, Director of Library Services, Brigham and Women’s Faulkner Hospital for facilitating access to reference articles and Harvard Catalyst for statistical support. The authors’ responsibilities were as follows—CSF, ELG, KME, BFD, GGB: conceived and designed the study; PDC CSF ELG: analyzed the study; and all authors contributed to the manuscript. This trial was funded by the National Cancer Institute (P50CA127003; K07CA148894 [Ng]; K22CA126992; 5K05CA124415 [Emmons]; U01CA138962 [Chandler]), the Department of Defense Prostate Cancer Research Program (PC081669 [Drake]), the American Society of Clinical Oncology Career Development Award (Ng) and Pharmavite LLC. The sponsor had no role in designing, developing the protocol, or conducting the trial; in data collection, analysis, management, or interpretation of the data; or in preparing the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Hollis has support from DiaSorin S.p.A for serving as an academic consultant. The remaining authors declare no conflict of interest.
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/4.0/
About this article
Cite this article
Chandler, P., Scott, J., Drake, B. et al. Impact of vitamin D supplementation on adiposity in African-Americans. Nutr & Diabetes 5, e147 (2015). https://doi.org/10.1038/nutd.2014.44
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nutd.2014.44