Abstract
Background:
Pre-commitment strategies can encourage participants to commit to a healthy food plan and have been suggested as a potential strategy for weight loss. However, it is unclear whether such strategies are cost-effective.
Objective:
To analyse whether pre-commitment interventions that facilitate healthier diets are a cost-effective approach to tackle obesity.
Methods:
Effectiveness evidence was obtained from a systematic review of the literature. For interventions demonstrating a clinically significant change in weight, a Markov model was employed to simulate the long-term health and economic consequences. The review supported modelling just one intervention: grocery shopping to a predetermined list combined with standard behavioural therapy (SBT). SBT alone and do nothing were used as comparators. The target population was overweight or obese adult women. A lifetime horizon for health effects (expressed as quality-adjusted life years (QALYs)) and costs from the perspective of the UK health sector were used to calculate incremental cost-effectiveness ratios (ICERs).
Results:
In the base case analysis, the pre-commitment strategy of shopping to a list was found to be more effective and cost saving when compared against SBT, and cost-effective when compared against ‘do nothing’ (ICER=£166 per QALY gained). A sensitivity analysis indicated that shopping to a list remained dominant or cost-effective under various scenarios.
Conclusion:
Our findings suggest grocery shopping to a predetermined list combined with SBT is a cost-effective means for reducing obesity and its related health conditions.
Similar content being viewed by others
Introduction
Recent European Union statistics revealed that the United Kingdom has more obese women (24%) than any other member state.1 Overweight and obesity was responsible for 7.3% of morbidity and mortality in the United Kingdom in 2002 and contributed an estimated £3 billion in direct health costs to the National Health Service (NHS).2 A systematic review of evidence from mostly high income countries suggests that obesity alone accounts for 1–3% of national health care expenditure.3 Current public health approaches to tackling obesity, which have largely focussed on information provision and health education, have not been particularly effective in reversing the obesity epidemic. Recent findings suggest that behavioural economics may offer alternative approaches to improving everyday decisions people make about the foods they consume, exercise habits and their health more generally.4, 5 The behavioural economics literature recognises that very often, people’s actual behaviour departs significantly from their intended behaviour. For example, many consumers know all too well that overeating and under-exercising will lead to weight gain and increased risk of chronic illness. Many consumers take the further step of forming an intention to improve their diet and increase activity levels. However, in many cases impulsiveness or poor self-control leads people to behave in a manner that departs from their good intentions.5 This has been described in the consumer behaviour literature as a struggle between the want self and the should self,6 or between the psychological forces of desire and willpower.7
Pre-commitment (or commitment) mechanisms could assist individuals who have difficulty sticking to a healthy eating plan, by removing future temptations to deviate from the plan.4, 8 In other words, they could be used to protect long-term health goals from short-term consumption decisions.9 Preselecting menu items in school or work cafeterias, pre-ordering grocery items on supermarket websites, and preselecting the list of food items to buy at the supermarket are examples of strategies to constrain oneself from giving in to future unhealthy temptations. One advantage of such strategies (unlike more paternalistic approaches such as food/drink bans or fat taxes) is that they nudge people towards making better decisions, while safeguarding individuals’ freedom of choice.4 There have been a few attempts to evaluate the impact of these types of pre-commitment mechanisms on consumption behaviour. For example, one study found that shoppers of an online grocer were more likely to purchase ‘should’ choices (healthier foods) when they ordered for home delivery farther into the future than when they ordered for delivery sooner.10 Despite some evidence that pre-commitment devices can assist with healthier decisions, little is known about the long-term cost-effectiveness of such strategies.
The aim of this study is to determine whether pre-commitment strategies from the existing literature are likely to be cost-effective in facilitating healthier diets, weight loss and ultimately better health among overweight individuals.
Materials and methods
Interventions
A systematic review was conducted to identify interventions that employed pre-commitment strategies to influence food-purchasing patterns among overweight individuals. Databases searched included the Cochrane Central Register of Controlled Trials, the Cochrane Health Technology Assessment Database, MEDLINE and EconLit. Full details of the search strategy are presented in Supplementary Table S1 and Supplementary Figure S1. Included studies trialled interventions that employed a pre-commitment mechanism to modify the mode (for example, in-person versus online), frequency, duration, product selection strategy or provider that subjects use for purchasing groceries. Interventions that relied on other mechanisms of action such as changes in the relative price of healthy foods were excluded from consideration. Studies were only included if the subjects in the study were overweight or obese. A further inclusion criteria were that the reported outcome measure reflected: (a) changes in grocery consumption (for example, purchasing volume or consumption pattern); (b) changes in weight or body fatness (for example, weight, body mass index (BMI), skin-folds or body composition) or (c) changes in obesity-related illnesses (for example, obesity risk factors, disease status, or severity or mortality). Only studies in English language were included.
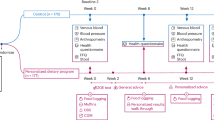
Two papers were identified as meeting the inclusion criteria.11, 12 Wing et al.11 investigated (among other interventions) the impact of shopping to a list among 163 overweight women from the United States (aged 15–55, mean age 41 years). Participants were randomly assigned to 6 months of standard behavioural therapy (SBT) or SBT combined with provision of detailed meal plans and corresponding shopping lists (SBT+list). Average baseline BMI was 32 kg m−2 for the SBT group and 33 kg m−2 for the SBT+list group. SBT in this trial involved weekly group sessions with therapists that focussed on stimulus control, pre-planning and problem solving. Low-calorie and low-fat goals were prescribed and participants received a calorie/fat guide book. Participants recorded all foods consumed and were set exercise goals. SBT participants were given no specific instruction on what they should consume. In addition to SBT, SBT+list participants received meal plans for five breakfasts and five dinners each week and a weekly grocery list that covered all items needed to make these meals. Height and weight were measured at baseline and follow-up. Over the 26-week period, SBT+list participants lost significantly more weight (12.0±7.2 kg) than SBT participants (8.0±6.2 kg), implying a treatment effect of −4 kg or −1.5 kg m−2. At follow-up 1 year later, participants had regained some lost weight but total weight loss from baseline still averaged 6.9 kg for the SBT+list group and 3.3 kg for the SBT group.
Gorin et al.12 studied the effect of using online grocery shopping to modify the household food environment among 28 overweight participants from the United States (aged 21–65, 89% female). Participants were randomly assigned to 8 weeks of standard behavioural weight loss therapy or weight loss therapy combined with online purchase and home delivery of groceries (home delivery). Weight loss therapy involved weekly sessions with therapists that focussed on behavioural and cognitive skills. All participants were set low-calorie, low-fat diets and instructed to exercise for 150 min a week. Calorie intake and exercise were self-monitored. The home delivery participants were instructed to order all of their groceries online for home delivery. A tutorial on ordering online was provided and delivery costs were reimbursed. Home delivery participants demonstrated significantly greater reductions in the total number of food categories in the home (out of 26 possible food categories) with an average change of −3.67±1.05 compared with −0.14±2.63 foods. Home delivery participants also experienced a greater reduction in the number of high-fat foods. However, no significant difference in weight loss was found between the groups after the 8-week intervention.
Long-term intervention effectiveness
The results from the identified trials were extrapolated to estimate long-term weight loss for use in the cost-effectiveness analysis. Shopping to a list (SBT+list)11 was associated with weight loss at the end of the 6-month treatment programme (week 26) and at the final follow-up 18 months after baseline (week 78). Weight change (in kg) was converted to BMI change (in kg m−2) based on relativities between weight and BMI at baseline. At the end of the intervention (week 26), the SBT+list group had a lower BMI (28 kg m−2) than SBT participants (29 kg m−2). Although a between-group difference in weight and BMI persisted at week 78, both groups regained weight between week 26 and week 78. Other studies13, 14 have also shown that weight regain is common following short-term weight loss interventions. Therefore, for extrapolation it was assumed that the weight regain per month observed between week 26 and week 78 in SBT (0.39 kg) and SBT+list (0.43 kg) groups was a linear function of time that continued until participants returned to the baseline BMI of 33 kg m−2. In doing so, the intervention effect was modelled as a temporary (rather than permanent) weight loss. This conservative assumption was made to ensure the model did not overestimate the effectiveness of the intervention. SBT and SBT+list groups had slightly different average baseline BMI values in the trial. In extrapolating the trial evidence, it is assumed that both groups had the same baseline BMI of 33 kg m−2 while still maintaining follow-up BMI values as per weight loss achieved in the trial. Body weight and BMI were estimated for SBT and SBT+list groups at 6-monthly intervals up to 3 years after baseline (see Supplementary Figure S2).
In the Gorin et al. trial,12 home delivery was not associated with a significant treatment effect with respect to weight loss. In the absence of a clinically significant treatment effect, no amount of modelling will allow us to conclude that home delivery is a cost-effective strategy. The modelled cost-effectiveness analysis reported below, therefore, focuses solely on the shopping to a list intervention from the Wing et al.11 trial.
Modelling the health outcomes and costs
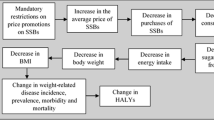
A Markov model was constructed to calculate the lifetime health outcomes and costs associated with the weight loss achieved by the shopping to a list pre-commitment intervention delivered to an overweight population. The Markov model was based on the model developed by Galani et al.15 that compared lifestyle interventions in overweight and obese people. The original model was adjusted to include disease states representing colorectal cancer and osteoarthritis, which are known to be related to obesity16, 17 and to include a state representing death unrelated to weight status. Hypercholesterolaemia per se is not considered to have an impact on quality of life,18 thus this state was absorbed into the overweight state. Where possible, the model was developed so as to allow for relationships between co-morbidities, permitting transition between co-morbidity health states. The model was developed in accordance with good practice guidelines.19, 20 Owing to limitations surrounding available data, additive or multiplicative effects of multiple morbidities were not considered within the model. The state of hypertension was omitted from the model, as data to capture the complex interrelationship between this disease, obesity, stroke and coronary heart disease could not be identified. Finally, a post-intervention weight state was added to illustrate the role of the intervention in reducing weight and to highlight that transition probabilities to disease states are different at lower levels of BMI. The Markov model used in the analysis is detailed in Supplementary Figure S3 of the Supplementary Information section.
Essentially, the model compares two populations over their lifetime: a reference population of overweight individuals who receive SBT and an intervention population who additionally receive the pre-commitment intervention (shopping to a list). The model enables the simulation of participants moving from a baseline weight to post-intervention weight states, disease states related to an elevated BMI (type 2 diabetes, colorectal cancer, osteoarthritis, coronary heart disease and stoke) and death (either related or unrelated to obesity). The disease risks, transition probabilities and mortality rates used to populate the model were obtained from published sources (detailed in Supplementary Tables S2, S3 and S4 of the Supplementary Information). Care was taken to ensure that the best available evidence was used. This meant that where possible, estimates from meta-analyses of large population studies from the United Kingdom (or comparable high income countries) were used. Where meta-analyses were not available, estimates from a single study in which the population matched the target population as closely as possible were used.
The model was built in Microsoft Excel. The individuals were assumed to enter the model aged 40 years old (the average age of the women in the trial) and to have a BMI of 33 kg m−2 (the average baseline BMI in the trial) and be free of any obesity-related illnesses. For a hypothetical cohort of 1000 individuals, the model simulated the effect of the intervention on body weight and the development of co-morbidities over a period of 40 years to estimate lifetime costs and effects of the intervention. The 40-year time horizon was broken down into cycles of 6 months to allow the model to capture the gradual effect of weight regain, which was measured at 6 and 18 months in the trial.
The cost-effectiveness of the intervention was ascertained through comparison of costs and quality-adjusted life years (QALYs) and summarised as incremental cost-effectiveness ratios (ICERs). An ICER estimates the ratio of the difference in costs to the difference in effects between the intervention and the comparator, and here it is expressed in terms of cost (£) per QALY gained. Both costs and effects were discounted at 3.5% per annum in accordance with recommendations from the UK treasury.21 The analysis adopted the perspective of the UK NHS, assuming that—for any given level of resource available—the decision maker wishes to maximise aggregate health benefits. This perspective allows comparability with other services currently funded under the NHS and may motivate debate on whether there is value in the NHS taking responsibility for funding interventions that are outside the standard health care system.
The analysis included a do nothing case, in which it was assumed that all participants enter the model free of any obesity-related illnesses with a mean BMI of 33 kg m−2. Under the do nothing case, participants progress to obesity-related illness and to related and unrelated death at rates specific to their baseline BMI.
Costs
The intervention costs were based on the reported use of resources in the trial11 and are detailed in Supplementary Table S5. It was assumed that weekly group behavioural sessions lasted 60 min and required 30 min of preparation, with each therapist supporting 20 participants per session (as achieved in the trial). For the SBT+list component, 30 min of dietician time (per 20 participants) was added to reflect preparation of meal plans and grocery lists. The total intervention costs for SBT and SBT+list were estimated at £125 and £141 per participant, respectively.
The costs for each disease state were obtained from published sources and are outlined in Supplementary Table S6. The cost of osteoarthritis was obtained in Australian dollars and converted to British pounds using gross domestic product-purchasing power parities.22 The remaining costs were obtained from UK-based studies. The cost of the pre- and post-intervention weight states was based on the cost of treatment for elevated cholesterol (average cost of statin treatment plus monitoring costs),23 weighted by the proportion of obese individuals with elevated cholesterol.24 All costs were obtained for annual treatment and divided in half to obtain the 6-month figure. The exception is the cost of stroke, for which there is a substantial cost at the time of event and lesser costs over subsequent periods. Upfront costs were thus included in the first cycle of stroke, with lesser costs in subsequent cycles. The absorbing states of death and unrelated death were assumed to have zero cost. All costs were adjusted to 2010 prices using the price inflation indices for hospital and community health services.25
Health outcomes
Time spent in each state was weighted using quality of life adjustments and then summed to calculate QALYs. To maintain a consistent approach to valuing health states, utility estimates (on a scale from 0 (dead) to 1 (perfect health)) were derived by subtracting utility decrements associated with each health condition from a reference utility estimate of normal weight individuals in the UK population.26 The underlying assumption here is that normal weight individuals do not suffer from the co-morbidities associated with overweight and obesity. The utility weights were obtained from the EuroQol EQ-5D index score and are detailed in Supplementary Table S7.
Parameter uncertainty
A sensitivity analysis was conducted to characterise uncertainty surrounding the base case estimate of cost-effectiveness. Upper and lower bounds for the treatment effect were calculated assuming that the width of the confidence interval (CI) surrounding initial weight loss was constant throughout weight regain. The treatment effect (95% CI) was estimated to be −1.5 kg m−2 (CI: −2.65, −0.35) at week 26.
Best and worst case scenarios for the intervention were constructed to test the sensitivity of the incremental cost-effectiveness of SBT+list compared with SBT alone (or doing nothing) to extremes of the parameter distributions. This approach effectively allows us to estimate whether the intervention remains cost-effective under a worst case scenario. The best case scenario was calculated by halving the intervention costs; doubling the health-state costs; assuming the upper CI limits for (a) baseline risks of disease, (b) relative risks of disease associated with BMI change, and (c) disease-related mortality risk; and assuming the lower CI limits for (d) the treatment effect of the SBT+list intervention (−2.65 kg m−2 at week 26) and (e) utilities associated with each disease state (while maintaining base case utility values for overweight and obesity). Under the worst case scenario, the intervention costs were doubled, health-state costs were halved and the treatment effect was assumed to be at the lower bound estimate. All remaining parameters were also set to the opposite bound to that set in the best case scenario. Cost ranges and upper and lower CI limits of parameters for the sensitivity analyses are presented in Supplementary Tables S2–S7.
In addition, different scenarios were explored in one-way sensitivity analyses to determine which parameters are the key drivers of the results. The treatment effect of the SBT+list intervention was varied to the upper (scenario 1) and lower (scenario 2) bound estimates. Intervention costs were halved (scenario 3) and doubled (scenario 4), and costs for disease states were halved (scenario 5). Utilities for disease states were set to their upper bound estimates (scenario 6). The model was estimated for different ages including an average age of 50 and 65 (scenarios 7 and 8), and for males (scenario 9). Finally, the discount rate was varied between 0 (scenario 10) and 6% (scenario 11).
It was clear from the sensitivity analyses that the base case conclusions changed little even under extreme scenarios. Therefore, a probabilistic sensitivity analysis was not undertaken because its results would be of little additional value.
Results
The results of the cost-effectiveness analysis for the base case and best/worst case scenarios are presented in Table 1. Results show that the SBT+list pre-commitment intervention dominates the SBT intervention in the base case, producing greater health gains at a lower cost. When compared against the do nothing option, the SBT+list intervention yields an ICER of £166 per QALY. This ICER is well below the threshold of £20 000 per QALY, below which interventions are deemed cost-effective by the National Institute for Health and Care Excellence,27 suggesting the SBT+list intervention is cost-effective.
Under the best case scenario, the SBT+list intervention dominates both SBT and do nothing, improving outcomes at lower cost. Under the worst case scenario, the intervention still yields more QALYs than both SBT and the do nothing alternative, but at a higher cost. The resulting ICERs for the SBT+list intervention (£1720 per QALY against SBT and £2455 per QALY against the do nothing alternative) suggest the intervention remains cost-effective even under the worst case scenario.
The ICERs from one-way sensitivity analyses are shown in Table 2. The additional benefit from these analyses over and above the best/worst case sensitivity analyses is that it allows us to identify parameters that are the key drivers of any uncertainty. Pre-commitment via the SBT+list intervention either dominated or remained cost-effective under all scenarios. The results were more sensitive to changes in the treatment effect, interventions costs and disease state costs. Varying the other parameters in the model made little difference to the base case results.
Discussion
The total incremental weight loss achieved by the pre-commitment intervention of shopping to a list was small and of short duration owing to the tendency for weight regain and (potentially conservative) extrapolation of continued weight regain after trial. Despite the assumption of a return to baseline weight following the intervention, the pre-commitment intervention of shopping to a list, when combined with SBT was found to be cost saving compared with SBT alone and cost-effective compared with the do nothing alternative. These findings provide preliminary evidence that shopping to a list (combined with SBT) is likely to be a cost-effective means through which the UK NHS could reduce obesity and its related illnesses. The sensitivity analysis showed that the SBT+list intervention remained either dominant or cost-effective under a range of scenarios, including under an extreme worst case scenario. The results from this paper are encouraging and provide a valuable starting point for further research into the cost and benefits of obesity reduction strategies drawn from behavioural economics.
In contrast to the evidence for the shopping to a list intervention, pre-commitment via online grocery shopping and home delivery (home delivery)12 was not found to be effective with respect to weight loss, and therefore cannot be considered cost-effective on the current available evidence. Further, primary evidence would be required before home delivery can be recommended as a cost-effective means of reducing obesity and its related illnesses. The systematic review of the literature did not identify any other interventions that employed pre-commitment strategies to influence food-purchasing behaviour for overweight individuals.
The analysis focused on weight change of the trial participant and as such ignored positive spillover or ripple effects to other members of the household. Previous research has shown that weight loss of spouses of participants in a randomised weight loss trial was associated with participant weight loss and decreases in high-fat foods in the home.28 For both interventions evaluated in this present study, the pre-commitment intervention resulted in a significant change to the home food environment. Such changes may have a positive impact on other family members, especially children, for which the benefits of obesity prevention are substantial and are long term. On considering only the costs and health outcomes of the individual receiving the intervention, the benefits of the intervention in households with more than one individual have probably been underestimated.
Generalisability of results
The study population from which the treatment effect of SBT+list was obtained was restricted to overweight women in their early 40s. For the findings to be generalisable to males and other (adult) age groups, it is necessary to assume that advice to shop to a list would influence the home food environment, and therefore weight of these population groups in much the same way as demonstrated in the trial population. Assuming a generalisable treatment effect, the Markov model can be applied to males and different age groups, using age- and gender-specific values for baseline disease risks and utilities where available. Results for some of these populations were provided in the sensitivity analysis. Although the model was for a UK adult population, the results could apply to other western countries that have comparable disease patterns and prices.
Limitations and areas for further research
There are a number of limitations to this analysis. First, there was a lack of effectiveness evidence. Only two studies met the criteria of the systematic review, and only one of these studies produced a clinically significant as well as statistically significant treatment effect. This finding is of itself worth reporting, and highlights the need for further trial evidence. The Wing et al.11 trial itself was not without limitations. It is unclear how participants were randomised and there is no mention of any attempt to conceal randomisation schedules or blind analysts to treatment group status. Despite these limitations, this trial represents the best available evidence on the effects of a pre-commitment mechanism for an overweight population. Further research in this area, particularly if focussed on longer-term interventions would be useful in improving precision of the treatment effect.
There are also limitations surrounding the model. To estimate long-term weight loss, it was assumed that the weight loss achieved from the intervention was temporary and that participants returned to their baseline weight. It was further assumed that the relative risks of chronic disease for participants in the years following weight regain were no different to that of individuals with the same, but relatively constant BMI (that is, those who did not experience a temporary weight loss). This assumption is consistent with the literature that finds an insignificant association between disease risk (or mortality) and intentional weight loss, followed by weight gain (weight cycling) after controlling for BMI.29, 30 Owing to the complex nature of the relationship between obesity and chronic diseases, it was necessary to use a simplified model of disease progression. The model did not account for the possibility of co-morbid conditions (participants could only be in one disease state at a time), and is therefore possible to underestimate the costs and utility decrements of obesity and its related diseases. The positive effects of weight loss were only modelled through reductions in BMI and consequently through known related diseases. However, it is possible for weight loss to have other impacts on wellbeing, such as improved mental health and self-esteem that may not be fully captured by health-state utility values.31 The inclusion of such effects could improve the cost-effectiveness estimates of the intervention.
The analysis adopted the perspective of the UK NHS, and therefore did not consider the costs borne by the individual or their families. The impact of the intervention on household costs would in part depend on whether the (healthier) grocery items on the intervention’s shopping list were in total more costly than the total expenditure on food (including snacks) and meals of individuals (or families) in the comparison group.
Conclusion
Existing evidence suggest that pre-commitment strategies may be effective interventions for facilitating healthier diets,11, 12 and for promoting weight loss among overweight and obese individuals.11 Although the incremental weight loss arising from the shopping to a list pre-commitment intervention was small and of short duration, it persisted for a sufficient length of time to yield gains in quality of life and survival when compared with SBT or the do nothing alternative. Incorporating the costs of the intervention and disease states resulted in the shopping to a list intervention being cheaper than SBT and cost-effective against the do nothing alternative. These findings provide preliminary evidence to suggest that a pre-commitment strategy to alter the home food environment is a cost-effective means for reducing obesity and its related illnesses. This study highlights the need for further evidence, particularly from trials that focus on long-term pre-commitment interventions.
References
Eurostat. News release: European Health Interview Survey - Between 8% and 25% of adults are obese across Member States. Eurostat: Luxembourg, 2011. Available at http://www.epp.eurostat.ec.europa.eu/cache/ITY_PUBLIC/3-24112011-BP/EN/3-24112011-BP-EN.PDF (accessed 1 December 2011).
Allender S, Rayner M . The burden of overweight and obesity-related ill health in the UK. Obe Rev 2007; 8: 467–473.
Withrow D, Alter DA . The economic burden of obesity worldwide: a systematic review of the direct costs of obesity. Obes Rev 2011; 12: 131–141.
Thaler RH, Sunstein CR . Nudge: Improving Decisions About Health, Wealth, and happiness. Yale University Press: New Haven, CT, USA, 2008.
Kooreman P, Prast H . What does behavioral economics mean for policy? challenges to savings and health policies in the Netherlands. Economist 2010; 158: 101–122.
Bazerman MH, Tenbrunsel AE, Wade-Benzoni K . Negotiating with yourself and losing: making decisions with competing internal preferences. Acad Manage Rev 1998; 23: 225–241.
Hoch SJ, Loewenstein GF . Time-inconsistent preferences and consumer self-control. J Consum Res 1991; 17: 492–507.
Rogers T, Bazerman MH . Future lock-in: future implementation increases selection of 'should' choices. Organ Behav Hum Decis Process 2008; 106: 1–20.
Scharff RL . Obesity and hyperbolic discounting: evidence and implications. J Consum Policy 2009; 32: 3–21.
Milkman K, Rogers T, Bazerman M . I’ll have the ice cream soon and the vegetables later: a study of online grocery purchases and order lead time. Mark Lett 2010; 21: 17–35.
Wing R, Jeffery R, Burton L, Thorson C, Sperber Nissinoff K, Baxter J . Food provision vs structured meal plans in the behavioral treatment of obesity. Int J Obes 1996; 20: 56–62.
Gorin A, Raynor H, Niemeier H, Wing R . Home grocery delivery improves the household food environments of behavioral weight loss participants: results of an 8-week pilot study. Int J Behav Nutr Phys Act 2007; 4: 58.
Kramer FM, Jeffery RW, Forster JL, Snell MK . Long-term follow-up of behavioral treatment for obesity: patterns of weight regain among men and women. Int J Obes 1989; 13: 123.
Latner J, Stunkard A, Wilson G, Jackson M, Zelitch D, Labouvie E . Effective long-term treatment of obesity: a continuing care model. Int J Obes 2000; 24: 893–898.
Galani C, Schneider H, Rutten F . Modelling the lifetime costs and health effects of lifestyle intervention in the prevention and treatment of obesity in Switzerland. Int J Public Health 2007; 52: 372–382.
Moghaddam AA, Woodward M, Huxley R . Obesity and risk of colorectal cancer: a meta-analysis of 31 studies with 70,000 events. Cancer Epidemiol Biomarkers Prev 2007; 16: 2533.
Grotle M, Hagen K, Natvig B, Dahl F, Kvien T . Obesity and osteoarthritis in knee, hip and/or hand: an epidemiological study in the general population with 10 years follow-up. BMC Musculoskelet Disord 2008; 9: 132.
Ward S, Lloyd Jones M, Pandor A, Holmes M, Ara R, Ryan A et al. A systematic review and economic evaluation of statins for the prevention of coronary events. Health Technol Assess 2007; 11: 1–160.
Husereau D, Drummond M, Petrou S, Carswell C, Moher D, Greenberg D et al. Consolidated health economic evaluation reporting standards (CHEERS)—explanation and elaboration: a report of the ispor health economic evaluation publication guidelines good reporting practices task force. Value Health 2013; 16: 231–250.
Philips Z, Bojke L, Sculpher M, Claxton K, Golder S . Good practice guidelines for decision-analytic modelling in health technology assessment. PharmacoEconomics 2006; 24: 355–371.
HM Treasury. The Green Book: Appraisal and Evaluation in Central Government. TSO: London, 2007.
OCED. Purchasing power parities (PPP) for OECD Countries. OECD: Paris, 2011. Available at http://www.oecd.org/std/ppp (accessed 2 September 2011).
Matrix Insight. Statins for primary prevention of stroke and heart disease (demonstrating QALYs for two example CVD risk groups). Matrix Insight: London, 2009.
Wilson PWF, D'Agostino RB, Sullivan L, Parise H, Kannel WB . Overweight and obesity as determinants of cardiovascular risk: the Framingham experience. Arch Intern Med 2002; 162: 1867.
Curtis L . Unit costs of health and social care 2010 Report no.: 9781902671 Personal Social Services Research Unit, The University of Kent: Canterbury, 2010.
Kinge JM, Morris S . Socioeconomic variation in the impact of obesity on health related quality of life. Soc Sci Med 2010; 71: 1864–1871.
NICE. Guide to the Methods of Technology Appraisal. National Institute for Health and Clinical Excellence: London, 2008. Available at http://www.nice.org.uk (accessed 19 September 2011).
Gorin AA, Wing RR, Fava JL, Jakicic JM, Jeffery R, West DS et al. Weight loss treatment influences untreated spouses and the home environment: evidence of a ripple effect. Int J Obes 2008; 32: 1678.
Field AE, Manson JE, Laird N, Williamson DF, Willett WC, Colditz GA . Weight cycling and the risk of developing type 2 diabetes among adult women in the United States. Obe Res 2002; 12: 267–274.
Stevens VL, Jacobs EJ, Sun J, Patel AV, McCullough ML, Teras LR et al. Weight cycling and mortality in a large prospective US study. Am J Epidemiol 2012; 175: 785–792.
Walsh R . Lifestyle and mental health. Am Psychol 2011; 66: 579–592.
Drummond MF, Sculpher MJ, Torrance GW . Methods for the Economic Evaluation of Health Care Programmes 3rd edn Oxford University Press: New York, NY, USA, 2005.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Nutrition & Diabetes website
Supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/3.0/
About this article
Cite this article
Au, N., Marsden, G., Mortimer, D. et al. The cost-effectiveness of shopping to a predetermined grocery list to reduce overweight and obesity. Nutr & Diabetes 3, e77 (2013). https://doi.org/10.1038/nutd.2013.18
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nutd.2013.18
Keywords
This article is cited by
-
Multi-dimensional Profiles of Risk and Their Association with Obesity-Severity in Low-Income Black Women
Journal of Immigrant and Minority Health (2023)
-
Does the Structure Matter? An External Validation and Health Economic Results Comparison of Event Simulation Approaches in Severe Obesity
PharmacoEconomics (2022)
-
Replication of Published Health Economic Obesity Models: Assessment of Facilitators, Hurdles and Reproduction Success
PharmacoEconomics (2021)
-
Behavioural Insights and (Un)healthy Dietary Choices: a Review of Current Evidence
Journal of Consumer Policy (2019)