Abstract
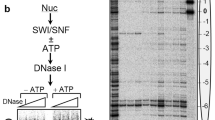
The RSC chromatin remodeler contains Sth1, an ATP-dependent DNA translocase. On DNA substrates, RSC/Sth1 tracks along one strand of the duplex with a 3′ → 5′ polarity and a tracking requirement of one base, properties that may enable directional DNA translocation on nucleosomes. The binding of RSC or Sth1 elicits a DNase I–hypersensitive site approximately two DNA turns from the nucleosomal dyad, and the binding of Sth1 requires intact DNA at this location. Results with various nucleosome substrates suggest that RSC/Sth1 remains at a fixed position on the histone octamer and that Sth1 conducts directional DNA translocation from a location about two turns from the nucleosomal dyad, drawing in DNA from one side of the nucleosome and pumping it toward the other. These studies suggest that nucleosome mobilization involves directional DNA translocation initiating from a fixed internal site on the nucleosome.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Almer, A., Rudolph, H., Hinnen, A. & Horz, W. Removal of positioned nucleosomes from the yeast PHO5 promoter upon PHO5 induction releases additional upstream activating DNA elements. EMBO J. 5, 2689–2696 (1986).
Boeger, H., Griesenbeck, J., Strattan, J.S. & Kornberg, R.D. Nucleosomes unfold completely at a transcriptionally active promoter. Mol. Cell 11, 1587–1598 (2003).
Phelan, M.L., Sif, S., Narlikar, G.J. & Kingston, R.E. Reconstitution of a core chromatin remodeling complex from SWI/SNF subunits. Mol. Cell 3, 247–253 (1999).
Corona, D.F. et al. ISWI is an ATP-dependent nucleosome remodeling factor. Mol. Cell 3, 239–245 (1999).
Tran, H.G., Steger, D.J., Iyer, V.R. & Johnson, A.D. The chromo domain protein chd1p from budding yeast is an ATP-dependent chromatin-modifying factor. EMBO J. 19, 2323–2331 (2000).
Boyer, L.A. et al. Functional delineation of three groups of the ATP-dependent family of chromatin remodeling enzymes. J. Biol. Chem. 275, 18864–18870 (2000).
Cairns, B.R. et al. RSC, an essential, abundant chromatin-remodeling complex. Cell 87, 1249–1260 (1996).
Laurent, B.C., Yang, X. & Carlson, M. An essential Saccharomyces cerevisiae gene homologous to SNF2 encodes a helicase-related protein in a new family. Mol. Cell. Biol. 12, 1893–1902 (1992).
Peterson, C.L., Zhao, Y. & Chait, B.T. Subunits of the yeast SWI/SNF complex are members of the actin-related protein (ARP) family. J. Biol. Chem. 273, 23641–23644 (1998).
Wang, W. et al. Diversity and specialization of mammalian SWI/SNF complexes. Genes Dev. 10, 2117–2130 (1996).
Imbalzano, A.N., Kwon, H., Green, M.R. & Kingston, R.E. Facilitated binding of TATA-binding protein to nucleosomal DNA. Nature 370, 481–485 (1994).
Saha, A., Wittmeyer, J. & Cairns, B.R. Chromatin remodeling by RSC involves ATP-dependent DNA translocation. Genes Dev. 16, 2120–2134 (2002).
Flaus, A. & Owen-Hughes, T. Mechanisms for nucleosome mobilization. Biopolymers 68, 563–578 (2003).
Whitehouse, I., Stockdale, C., Flaus, A., Szczelkun, M.D. & Owen-Hughes, T. Evidence for DNA translocation by the ISWI chromatin-remodeling enzyme. Mol. Cell. Biol. 23, 1935–1945 (2003).
Whitehouse, I. et al. Nucleosome mobilization catalysed by the yeast SWI/SNF complex. Nature 400, 784–787 (1999).
Langst, G., Bonte, E.J., Corona, D.F. & Becker, P.B. Nucleosome movement by CHRAC and ISWI without disruption or trans-displacement of the histone octamer. Cell 97, 843–852 (1999).
Hamiche, A., Sandaltzopoulos, R., Gdula, D.A. & Wu, C. ATP-dependent histone octamer sliding mediated by the chromatin remodeling complex NURF. Cell 97, 833–842 (1999).
Fyodorov, D.V. & Kadonaga, J.T. Dynamics of ATP-dependent chromatin assembly by ACF. Nature 418, 897–900 (2002).
Havas, K. et al. Generation of superhelical torsion by ATP-dependent chromatin remodeling activities. Cell 103, 1133–1142 (2000).
Langst, G. & Becker, P.B. ISWI induces nucleosome sliding on nicked DNA. Mol. Cell 8, 1085–1092 (2001).
Lorch, Y., Zhang, M. & Kornberg, R.D. RSC unravels the nucleosome. Mol. Cell 7, 89–95 (2001).
Kassabov, S.R., Zhang, B., Persinger, J. & Bartholomew, B. SWI/SNF unwraps, slides, and rewraps the nucleosome. Mol. Cell 11, 391–403 (2003).
Narlikar, G.J., Phelan, M.L. & Kingston, R.E. Generation and interconversion of multiple distinct nucleosomal states as a mechanism for catalyzing chromatin fluidity. Mol. Cell 8, 1219–1230 (2001).
Asturias, F.J., Chung, W.H., Kornberg, R.D. & Lorch, Y. Structural analysis of the RSC chromatin-remodeling complex. Proc. Natl. Acad. Sci. USA 99, 13477–13480 (2002).
Leschziner, A.E., Lemon, B., Tjian, R. & Nogales, E. Structural studies of the human PBAF chromatin-remodeling complex. Structure (Camb.) 13, 267–275 (2005).
Lowary, P.T. & Widom, J. New DNA sequence rules for high affinity binding to histone octamer and sequence-directed nucleosome positioning. J. Mol. Biol. 276, 19–42 (1998).
Polach, K.J. & Widom, J. Mechanism of protein access to specific DNA sequences in chromatin: a dynamic equilibrium model for gene regulation. J. Mol. Biol. 254, 130–149 (1995).
Anderson, J.D., Thastrom, A. & Widom, J. Spontaneous access of proteins to buried nucleosomal DNA target sites occurs via a mechanism that is distinct from nucleosome translocation. Mol. Cell. Biol. 22, 7147–7157 (2002).
Lorch, Y., Cairns, B.R., Zhang, M. & Kornberg, R.D. Activated RSC–nucleosome complex and persistently altered form of the nucleosome. Cell 94, 29–34 (1998).
Fan, H.Y., He, X., Kingston, R.E. & Narlikar, G.J. Distinct strategies to make nucleosomal DNA accessible. Mol. Cell 11, 1311–1322 (2003).
Li, G. & Widom, J. Nucleosomes facilitate their own invasion. Nat. Struct. Mol. Biol. 11, 763–769 (2004).
Jaskelioff, M., Van Komen, S., Krebs, J.E., Sung, P. & Peterson, C.L. Rad54p is a chromatin remodeling enzyme required for heteroduplex DNA joint formation with chromatin. J. Biol. Chem. 278, 9212–9218 (2003).
Schwanbeck, R., Xiao, H. & Wu, C. Spatial contacts and nucleosome step movements induced by the NURF chromatin remodeling complex. J. Biol. Chem. 279, 39933–39941 (2004).
Kagalwala, M.N., Glaus, B.J., Dang, W., Zofall, M. & Bartholomew, B. Topography of the ISW2–nucleosome complex: insights into nucleosome spacing and chromatin remodeling. EMBO J. 23, 2092–2104 (2004).
Flaus, A. & Owen-Hughes, T. Dynamic properties of nucleosomes during thermal and ATP-driven mobilization. Mol. Cell. Biol. 23, 7767–7779 (2003).
Lorch, Y., Davis, B. & Kornberg, R.D. Chromatin remodeling by DNA bending, not twisting. Proc. Natl. Acad. Sci. USA 102, 1329–1332 (2005).
Velankar, S.S., Soultanas, P., Dillingham, M.S., Subramanya, H.S. & Wigley, D.B. Crystal structures of complexes of PcrA DNA helicase with a DNA substrate indicate an inchworm mechanism. Cell 97, 75–84 (1999).
Soultanas, P., Dillingham, M.S., Wiley, P., Webb, M.R. & Wigley, D.B. Uncoupling DNA translocation and helicase activity in PcrA: direct evidence for an active mechanism. EMBO J. 19, 3799–3810 (2000).
Durr, H., Korner, C., Muller, M., Hickmann, V. & Hopfner, K.P. X-ray structures of the Sulfolobus solfataricus SWI2/SNF2 ATPase core and its complex with DNA. Cell 121, 363–373 (2005).
Thoma, N.H. et al. Structure of the SWI2/SNF2 chromatin-remodeling domain of eukaryotic Rad54. Nat. Struct. Mol. Biol. 12, 350–356 (2005).
Wittmeyer, J., Saha, A. & Cairns, B. DNA translocation and nucleosome remodeling assays by the RSC chromatin remodeling complex. Methods Enzymol. 377, 322–343 (2004).
Acknowledgements
We thank B. Schackmann for oligonucleotide syntheses, B. Kingston and G. Narlikar for the TGT plasmids, D. Close and C. Hill for purification of the Sth1301–1097 derivative and both M. Gordon and T. Formosa for comments on the manuscript. This work was funded by the US National Institutes of Health grant GM60415 (to B.R.C. and a majority of the support for A.S.), a University of Utah Graduate Research Fellowship (to A.S.), the Huntsman Cancer Institute (to J.W. and grant CA24014 for the support of core facilities) and the Howard Hughes Medical Institute (to B.R.C.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
RSC binding to nucleosomes bearing linker of different lengths. (PDF 88 kb)
Rights and permissions
About this article
Cite this article
Saha, A., Wittmeyer, J. & Cairns, B. Chromatin remodeling through directional DNA translocation from an internal nucleosomal site. Nat Struct Mol Biol 12, 747–755 (2005). https://doi.org/10.1038/nsmb973
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb973
This article is cited by
-
Energy-driven genome regulation by ATP-dependent chromatin remodellers
Nature Reviews Molecular Cell Biology (2023)
-
Structure of SWI/SNF chromatin remodeller RSC bound to a nucleosome
Nature (2020)
-
The BAF complex in development and disease
Epigenetics & Chromatin (2019)
-
Direct observation of coordinated DNA movements on the nucleosome during chromatin remodelling
Nature Communications (2019)
-
Structural rearrangements of the histone octamer translocate DNA
Nature Communications (2018)