Abstract
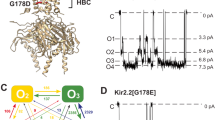
We show that in the potassium channel KcsA, proton-dependent activation is followed by an inactivation process similar to C-type inactivation, and this process is suppressed by an E71A mutation in the pore helix. EPR spectroscopy demonstrates that the inner gate opens maximally at low pH regardless of the magnitude of the single-channel-open probability, implying that stationary gating originates mostly from rearrangements at the selectivity filter. Two E71A crystal structures obtained at 2.5 Å reveal large structural excursions of the selectivity filter during ion conduction and provide a glimpse of the range of conformations available to this region of the channel during gating. These data establish a mechanistic basis for the role of the selectivity filter during channel activation and inactivation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
MacKinnon, R. Potassium channels and the atomic basis of selective ion conduction. Angew. Chem. Int. Edn Engl. 43, 4265–4277 (2004).
Yellen, G. The voltage-gated potassium channels and their relatives. Nature 419, 35–42 (2002).
Schrempf, H. et al. A prokaryotic potassium ion channel with two predicted transmembrane segments from Streptomyces lividans. EMBO J. 14, 5170–5178 (1995).
Cuello, L.G., Romero, J.G., Cortes, D.M. & Perozo, E. pH-dependent gating in the Streptomyces lividans K+ channel. Biochemistry 37, 3229–3236 (1998).
Heginbotham, L., LeMasurier, M., Kolmakova-Partensky, L. & Miller, C. Single Streptomyces lividans K+ channels: functional asymmetries and sidedness of proton activation. J. Gen. Physiol. 114, 551–560 (1999).
Cordero, J.F., Cuello, L.G. & Perozo, E. Voltage-dependent gating at the KcsA selectivity filter. Nat. Struct. Mol. Biol., advance online publication 12 March 2006 (doi:10.1038/nsmb1070).
Liu, Y.S., Sompornpisut, P. & Perozo, E. Structure of the KcsA channel intracellular gate in the open state. Nat. Struct. Biol. 8, 883–887 (2001).
Perozo, E., Cortes, D.M. & Cuello, L.G. Three-dimensional architecture and gating mechanism of a K+ channel studied by EPR spectroscopy. Nat. Struct. Biol. 5, 459–469 (1998).
Perozo, E., Cortes, D.M. & Cuello, L.G. Structural rearrangements underlying K+-channel activation gating. Science 285, 73–78 (1999).
Hackos, D.H., Chang, T.H. & Swartz, K.J. Scanning the intracellular S6 activation gate in the shaker K+ channel. J. Gen. Physiol. 119, 521–532 (2002).
Jin, T. et al. The βγ subunits of G proteins gate a K+ channel by pivoted bending of a transmembrane segment. Mol. Cell 10, 469–481 (2002).
Liu, Y., Holmgren, M., Jurman, M.E. & Yellen, G. Gated access to the pore of a voltage-dependent K+ channel. Neuron 19, 175–184 (1997).
Loussouarn, G., Phillips, L.R., Masia, R., Rose, T. & Nichols, C.G. Flexibility of the Kir6.2 inward rectifier K+ channel pore. Proc. Natl. Acad. Sci. USA 98, 4227–4232 (2001).
Jiang, Y. et al. The open pore conformation of potassium channels. Nature 417, 523–526 (2002).
Jiang, Y. et al. X-ray structure of a voltage-dependent K+ channel. Nature 423, 33–41 (2003).
Meuser, D., Splitt, H., Wagner, R. & Schrempf, H. Exploring the open pore of the potassium channel from Streptomyces lividans. FEBS Lett. 462, 447–452 (1999).
Demo, S.D. & Yellen, G. Ion effects on gating of the Ca2+-activated K+ channel correlate with occupancy of the pore. Biophys. J. 61, 639–648 (1992).
Shapiro, M.S. & Decoursey, T.E. Selectivity and gating of the type-L potassium channel in mouse lymphocytes. J. Gen. Physiol. 97, 1227–1250 (1991).
Spruce, A.E., Standen, N.B. & Stanfield, P.R. Rubidium ions and the gating of delayed rectifier potassium channels of frog skeletal-muscle. J. Physiol. (Lond.) 411, 597–610 (1989).
Swenson, R.P., Jr & Armstrong, C.M. K+ channels close more slowly in the presence of external K+ and Rb+. Nature 291, 427–429 (1981).
Lu, T. et al. Probing ion permeation and gating in a K+ channel with backbone mutations in the selectivity filter. Nat. Neurosci. 4, 239–246 (2001).
Alagem, N., Yesylevskyy, S. & Reuveny, E. The pore helix is involved in stabilizing the open state of inwardly rectifying K+ channels. Biophys. J. 85, 300–312 (2003).
Chapman, M.L., VanDongen, H.M. & VanDongen, A.M. Activation-dependent subconductance levels in the drk1 K channel suggest a subunit basis for ion permeation and gating. Biophys. J. 72, 708–719 (1997).
Proks, P., Capener, C.E., Jones, P. & Ashcroft, F.M. Mutations within the P-loop of Kir6.2 modulate the intraburst kinetics of the ATP-sensitive potassium channel. J. Gen. Physiol. 118, 341–353 (2001).
Zheng, J. & Sigworth, F.J. Selectivity changes during activation of mutant Shaker potassium channels. J. Gen. Physiol. 110, 101–117 (1997).
Hoshi, T., Zagotta, W.N. & Aldrich, R.W. Two types of inactivation in Shaker K+ channels: effects of alterations in the carboxy-terminal region. Neuron 7, 547–556 (1991).
Kiss, L., LoTurco, J. & Korn, S.J. Contribution of the selectivity filter to inactivation in potassium channels. Biophys. J. 76, 253–263 (1999).
Liu, Y., Jurman, M.E. & Yellen, G. Dynamic rearrangement of the outer mouth of a K+ channel during gating. Neuron 16, 859–867 (1996).
Lopez-Barneo, J., Hoshi, T., Heinemann, S.H. & Aldrich, R.W. Effects of external cations and mutations in the pore region on C-type inactivation of Shaker potassium channels. Receptors Channels 1, 61–71 (1993).
Lu, Z., Klem, A.M. & Ramu, Y. Ion conduction pore is conserved among potassium channels. Nature 413, 809–813 (2001).
Gao, L., Mi, X., Paajanen, V., Wang, K. & Fan, Z. Activation-coupled inactivation in the bacterial potassium channel KcsA. Proc. Natl. Acad. Sci. USA 102, 17630–17635 (2005).
Zhou, Y., Morais-Cabral, J.H., Kaufman, A. & MacKinnon, R. Chemistry of ion coordination and hydration revealed by a K+ channel-Fab complex at 2.0 A resolution. Nature 414, 43–48 (2001).
Kuo, A. et al. Crystal structure of the potassium channel KirBac1.1 in the closed state. Science 300, 1922–1926 (2003).
Yang, J., Yu, M., Jan, Y.N. & Jan, L.Y. Stabilization of ion selectivity filter by pore loop ion pairs in an inwardly rectifying potassium channel. Proc. Natl. Acad. Sci. USA 94, 1568–1572 (1997).
Choi, H. & Heginbotham, L. Functional influence of the pore helix glutamate in the KcsA K+ channel. Biophys. J. 86, 2137–2144 (2004).
Ficker, E., Jarolimek, W., Kiehn, J., Baumann, A. & Brown, A.M. Molecular determinants of dofetilide block of HERG K+ channels. Circ. Res. 82, 386–395 (1998).
Kuriyan, J., Petsko, G.A., Levy, R.M. & Karplus, M. Effect of anisotropy and anharmonicity on protein crystallographic refinement. An evaluation by molecular dynamics. J. Mol. Biol. 190, 227–254 (1986).
Doyle, D.A. et al. The structure of the potassium channel: molecular basis of K+ conduction and selectivity. Science 280, 69–77 (1998).
Berneche, S. & Roux, B. Energetics of ion conduction through the K+ channel. Nature 414, 73–77 (2001).
Zhou, Y. & MacKinnon, R. The occupancy of ions in the K+ selectivity filter: charge balance and coupling of ion binding to a protein conformational change underlie high conduction rates. J. Mol. Biol. 333, 965–975 (2003).
Lenaeus, M.J., Vamvouka, M., Focia, P.J. & Gross, A. Structural basis of TEA blockade in a model potassium channel. Nat. Struct. Mol. Biol. 12, 454–459 (2005).
Delcour, A.H., Martinac, B., Adler, J. & Kung, C. Modified reconstitution method used in patch-clamp studies of Escherichia coli ion channels. Biophys. J. 56, 631–636 (1989).
Cortes, D.M. & Perozo, E. Structural dynamics of the Streptomyces lividans K+ channel (SKC1): oligomeric stoichiometry and stability. Biochemistry 36, 10343–10352 (1997).
Cortes, D.M., Cuello, L.G. & Perozo, E. Molecular architecture of full-length KcsA: role of cytoplasmic domains in ion permeation and activation gating. J. Gen. Physiol. 117, 165–180 (2001).
Otwinowski, Z. & Minor, W. Processing of X-ray diffraction data collected in oscillation mode. Methods Enzymol. 276, 307–326 (1997).
Jones, T.A., Zou, J.-Y., Cowans, S.W. & Kjeldgaard, M. Improved methods for building protein models in electron-density maps and the location of errors in these models. Acta Crystallogr. A 47, 110–119 (1991).
Brunger, A.T. et al. Crystallography and NMR system: a new software suite for macromolecular structure determination. Acta Crystallogr. D Biol. Crystallogr. 54, 905–921 (1998).
Berneche, S. & Roux, B. Molecular dynamics of the KcsA K+ channel in a bilayer membrane. Biophys. J. 78, 2900–2917 (2000).
Woo, H.J., Dinner, A.R. & Roux, B. Grand canonical Monte Carlo simulations of water in protein environments. J. Chem. Phys. 121, 6392–6400 (2004).
Pettersen, E.F. et al. Chimera—a visualization system for exploratory research and analysis. J. Comput. Chem. 25, 1605–1612 (2004).
Acknowledgements
We thank J. Bushweller, R. Nakamoto and S. Chakrapani for critically reading the manuscript; the staff at BNL X-4A and X-29 for assistance in data collection; R. MacKinnon (Rockefeller University) for providing the KcsA monoclonal antibody hybridoma cell line and F.W. Garcia for assistance with monoclonal antibodies; and H. Wu for generous access to her laboratory space and facilities. V. Vasquez provided assistance with mutagenesis and channel biochemistry; C. Gonzalez provided comments and experimental advice. This work was supported by grants from the US National Institutes of Health to E.P. and B.R.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Inactivation kinetics depends on the extracellular K+ concentration (PDF 34 kb)
Rights and permissions
About this article
Cite this article
Cordero-Morales, J., Cuello, L., Zhao, Y. et al. Molecular determinants of gating at the potassium-channel selectivity filter. Nat Struct Mol Biol 13, 311–318 (2006). https://doi.org/10.1038/nsmb1069
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb1069
This article is cited by
-
35 years of channelling potassium ions
Nature (2022)
-
Ion-dependent structure, dynamics, and allosteric coupling in a non-selective cation channel
Nature Communications (2021)
-
Conformational equilibrium shift underlies altered K+ channel gating as revealed by NMR
Nature Communications (2020)
-
Structural and dynamic origins of ESR lineshapes in spin-labeled GB1 domain: the insights from spin dynamics simulations based on long MD trajectories
Scientific Reports (2020)
-
Shifts in the selectivity filter dynamics cause modal gating in K+ channels
Nature Communications (2019)