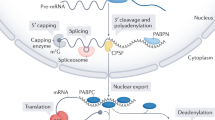
Abstract
Poly(A) tails are important elements in mRNA translation and stability, although recent genome-wide studies have concluded that poly(A) tail length is generally not associated with translational efficiency in nonembryonic cells. To investigate whether poly(A) tail size might be coupled to gene expression in an intact organism, we used an adapted TAIL-seq protocol to measure poly(A) tails in Caenorhabditis elegans. Surprisingly, we found that well-expressed transcripts contain relatively short, well-defined tails. This attribute appears to be dependent on translational efficiency, as transcripts enriched for optimal codons and ribosome association had the shortest tail sizes, whereas noncoding RNAs retained long tails. Across eukaryotes, short tails were a feature of abundant and well-translated mRNAs. This seems to contradict the dogma that deadenylation induces translational inhibition and mRNA decay and suggests that well-expressed mRNAs accumulate with pruned tails that accommodate a minimal number of poly(A)-binding proteins, which may be ideal for protective and translational functions.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Accession codes
References
Mangus, D.A., Evans, M.C. & Jacobson, A. Poly(A)-binding proteins: multifunctional scaffolds for the post-transcriptional control of gene expression. Genome Biol. 4, 223 (2003).
Goldstrohm, A.C. & Wickens, M. Multifunctional deadenylase complexes diversify mRNA control. Nat. Rev. Mol. Cell Biol. 9, 337–344 (2008).
Roy, B. & Jacobson, A. The intimate relationships of mRNA decay and translation. Trends Genet. 29, 691–699 (2013).
Jonas, S. & Izaurralde, E. Towards a molecular understanding of microRNA-mediated gene silencing. Nat. Rev. Genet. 16, 421–433 (2015).
Wahle, E. & Winkler, G.S. RNA decay machines: deadenylation by the Ccr4-not and Pan2-Pan3 complexes. Biochim. Biophys. Acta 1829, 561–570 (2013).
Xie, J., Kozlov, G. & Gehring, K. The “tale” of poly(A) binding protein: the MLLE domain and PAM2-containing proteins. Biochim. Biophys. Acta 1839, 1062–1068 (2014).
Weill, L., Belloc, E., Bava, F.A. & Méndez, R. Translational control by changes in poly(A) tail length: recycling mRNAs. Nat. Struct. Mol. Biol. 19, 577–585 (2012).
Chang, H., Lim, J., Ha, M. & Kim, V.N. TAIL-seq: genome-wide determination of poly(A) tail length and 3′ end modifications. Mol. Cell 53, 1044–1052 (2014).
Park, J.E., Yi, H., Kim, Y., Chang, H. & Kim, V.N. Regulation of Poly(A) tail and translation during the somatic cell cycle. Mol. Cell 62, 462–471 (2016).
Subtelny, A.O., Eichhorn, S.W., Chen, G.R., Sive, H. & Bartel, D.P. Poly(A)-tail profiling reveals an embryonic switch in translational control. Nature 508, 66–71 (2014).
Eichhorn, S.W. et al. mRNA poly(A)-tail changes specified by deadenylation broadly reshape translation in Drosophila oocytes and early embryos. eLife 5, e16955 (2016).
Lim, J., Lee, M., Son, A., Chang, H. & Kim, V.N. mTAIL-seq reveals dynamic poly(A) tail regulation in oocyte-to-embryo development. Genes Dev. 30, 1671–1682 (2016).
Baer, B.W. & Kornberg, R.D. The protein responsible for the repeating structure of cytoplasmic poly(A)-ribonucleoprotein. J. Cell Biol. 96, 717–721 (1983).
Smith, B.L., Gallie, D.R., Le, H. & Hansma, P.K. Visualization of poly(A)-binding protein complex formation with poly(A) RNA using atomic force microscopy. J. Struct. Biol. 119, 109–117 (1997).
Wang, Z., Day, N., Trifillis, P. & Kiledjian, M. An mRNA stability complex functions with poly(A)-binding protein to stabilize mRNA in vitro. Mol. Cell. Biol. 19, 4552–4560 (1999).
Nousch, M., Techritz, N., Hampel, D., Millonigg, S. & Eckmann, C.R. The Ccr4-Not deadenylase complex constitutes the main poly(A) removal activity in C. elegans. J. Cell Sci. 126, 4274–4285 (2013).
Chikina, M.D., Huttenhower, C., Murphy, C.T. & Troyanskaya, O.G. Global prediction of tissue-specific gene expression and context-dependent gene networks in Caenorhabditis elegans. PLOS Comput. Biol. 5, e1000417 (2009).
Jalkanen, A.L., Coleman, S.J. & Wilusz, J. Determinants and implications of mRNA poly(A) tail size--does this protein make my tail look big? Semin. Cell Dev. Biol. 34, 24–32 (2014).
Ingolia, N.T. Ribosome profiling: new views of translation, from single codons to genome scale. Nat. Rev. Genet. 15, 205–213 (2014).
Presnyak, V. et al. Codon optimality is a major determinant of mRNA stability. Cell 160, 1111–1124 (2015).
Quax, T.E., Claassens, N.J., Söll, D. & van der Oost, J. Codon bias as a means to fine-tune gene expression. Mol. Cell 59, 149–161 (2015).
Bazzini, A.A. et al. Codon identity regulates mRNA stability and translation efficiency during the maternal-to-zygotic transition. EMBO J. 35, 2087–2103 (2016).
Mishima, Y. & Tomari, Y. Codon usage and 3′ UTR length determine maternal mRNA stability in zebrafish. Mol. Cell 61, 874–885 (2016).
Radhakrishnan, A. et al. The DEAD-Box protein Dhh1p couples mRNA decay and translation by monitoring codon optimality. Cell 167, 122–132 e9 (2016).
Hendriks, G.J., Gaidatzis, D., Aeschimann, F. & Großhans, H. Extensive oscillatory gene expression during C. elegans larval development. Mol. Cell 53, 380–392 (2014).
Thompson, M.K. & Gilbert, W.V. mRNA length-sensing in eukaryotic translation: reconsidering the “closed loop” and its implications for translational control. Curr. Genet. 63, 613–620 (2017).
Nam, J.W. & Bartel, D.P. Long noncoding RNAs in C. elegans. Genome Res. 22, 2529–2540 (2012).
Pelechano, V., Wei, W. & Steinmetz, L.M. Widespread Co-translational RNA Decay Reveals Ribosome Dynamics. Cell 161, 1400–1412 (2015).
Schwanhäusser, B. et al. Global quantification of mammalian gene expression control. Nature 473, 337–342 (2011).
Coller, J. & Parker, R. Eukaryotic mRNA decapping. Annu. Rev. Biochem. 73, 861–890 (2004).
Hsu, C.L. & Stevens, A. Yeast cells lacking 5′-->3′ exoribonuclease 1 contain mRNA species that are poly(A) deficient and partially lack the 5′ cap structure. Mol. Cell. Biol. 13, 4826–4835 (1993).
Hu, W., Sweet, T.J., Chamnongpol, S., Baker, K.E. & Coller, J. Co-translational mRNA decay in Saccharomyces cerevisiae. Nature 461, 225–229 (2009).
Palatnik, C.M., Storti, R.V., Capone, A.K. & Jacobson, A. Messenger RNA stability in Dictyostelium discoideum: does poly(A) have a regulatory role? J. Mol. Biol. 141, 99–118 (1980).
Palatnik, C.M., Storti, R.V. & Jacobson, A. Fractionation and functional analysis of newly synthesized and decaying messenger RNAs from vegetative cells of Dictyostelium discoideum. J. Mol. Biol. 128, 371–395 (1979).
Beilharz, T.H. & Preiss, T. Widespread use of poly(A) tail length control to accentuate expression of the yeast transcriptome. RNA 13, 982–997 (2007).
Brown, C.E. & Sachs, A.B. Poly(A) tail length control in Saccharomyces cerevisiae occurs by message-specific deadenylation. Mol. Cell. Biol. 18, 6548–6559 (1998).
Tucker, M. et al. The transcription factor associated Ccr4 and Caf1 proteins are components of the major cytoplasmic mRNA deadenylase in Saccharomyces cerevisiae. Cell 104, 377–386 (2001).
Wong, Y.Y. et al. Cordycepin inhibits protein synthesis and cell adhesion through effects on signal transduction. J. Biol. Chem. 285, 2610–2621 (2010).
Gowrishankar, G. et al. Inhibition of mRNA deadenylation and degradation by different types of cell stress. Biol. Chem. 387, 323–327 (2006).
Hilgers, V., Teixeira, D. & Parker, R. Translation-independent inhibition of mRNA deadenylation during stress in Saccharomyces cerevisiae. RNA 12, 1835–1845 (2006).
Kleene, K.C., Cataldo, L., Mastrangelo, M.A. & Tagne, J.B. Alternative patterns of transcription and translation of the ribosomal protein L32 mRNA in somatic and spermatogenic cells in mice. Exp. Cell Res. 291, 101–110 (2003).
Hoshino, S. Mechanism of the initiation of mRNA decay: role of eRF3 family G proteins. Wiley Interdiscip. Rev. RNA 3, 743–757 (2012).
Coller, J.M., Gray, N.K. & Wickens, M.P. mRNA stabilization by poly(A) binding protein is independent of poly(A) and requires translation. Genes Dev. 12, 3226–3235 (1998).
Decker, C.J. & Parker, R. A turnover pathway for both stable and unstable mRNAs in yeast: evidence for a requirement for deadenylation. Genes Dev. 7, 1632–1643 (1993).
Ezzeddine, N. et al. Human TOB, an antiproliferative transcription factor, is a poly(A)-binding protein-dependent positive regulator of cytoplasmic mRNA deadenylation. Mol. Cell. Biol. 27, 7791–7801 (2007).
Funakoshi, Y. et al. Mechanism of mRNA deadenylation: evidence for a molecular interplay between translation termination factor eRF3 and mRNA deadenylases. Genes Dev. 21, 3135–3148 (2007).
Siddiqui, N. et al. Poly(A) nuclease interacts with the C-terminal domain of polyadenylate-binding protein domain from poly(A)-binding protein. J. Biol. Chem. 282, 25067–25075 (2007).
Shoemaker, C.J. & Green, R. Translation drives mRNA quality control. Nat. Struct. Mol. Biol. 19, 594–601 (2012).
Porta-de-la-Riva, M., Fontrodona, L., Villanueva, A. & Cerón, J. Basic Caenorhabditis elegans methods: synchronization and observation. J. Vis. Exp. 64, e4019 (2012).
Sallés, F.J., Richards, W.G. & Strickland, S. Assaying the polyadenylation state of mRNAs. Methods 17, 38–45 (1999).
Van Wynsberghe, P.M., Chan, S.P., Slack, F.J. & Pasquinelli, A.E. Analysis of microRNA expression and function. Methods Cell Biol. 106, 219–252 (2011).
Dobin, A. et al. STAR: ultrafast universal RNA-seq aligner. Bioinformatics 29, 15–21 (2013).
Quinlan, A.R. & Hall, I.M. BEDTools: a flexible suite of utilities for comparing genomic features. Bioinformatics 26, 841–842 (2010).
Bray, N.L., Pimentel, H., Melsted, P. & Pachter, L. Near-optimal probabilistic RNA-seq quantification. Nat. Biotechnol. 34, 525–527 (2016).
Stenico, M., Lloyd, A.T. & Sharp, P.M. Codon usage in Caenorhabditis elegans: delineation of translational selection and mutational biases. Nucleic Acids Res. 22, 2437–2446 (1994).
Akashi, H. Synonymous codon usage in Drosophila melanogaster: natural selection and translational accuracy. Genetics 136, 927–935 (1994).
Huang, W., Sherman, B.T. & Lempicki, R.A. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat. Protoc. 4, 44–57 (2009).
Acknowledgements
We thank V.N. Kim, J. Lim, and H. Chang for providing a detailed TAIL-seq protocol, their algorithm (tailseeker2), and technical assistance, E. Van Nostrand and members of the Yeo lab for assistance with the Illumina MiSeq platform, J. Chen and J. Broughton for programming support, and J. Lykke-Andersen, H. Cook-Andersen, M. Wilkinson, and members of the Pasquinelli lab for suggestions and critical reading of the manuscript. L.B.C. and A.L.N. were supported by the UCSD Cellular and Molecular Genetics Training Program through an institutional grant from the National Institute of General Medicine (T32 GM007240) and NSF Graduate Research Fellowships DGE-1650112 (L.B.C.) and DGE-1650112 (A.L.N.). This work was supported by grants from the NIH (GM071654) and UCSD Academic Senate to A.E.P., NIH (GM118018) to J.C., and NIH (HG004659) to G.W.Y. S.A.L. was supported by an international predoctoral fellowship from the HHMI.
Author information
Authors and Affiliations
Contributions
A.E.P. and S.A.L. designed the project and wrote the paper. S.A.L. conducted the experiments and data analysis with help from L.B.C., A.L.N., B.A.Y. and G.W.Y. Y.-H.C. and J.C. designed and performed experiments for Supplementary Figure 5.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 Validation of mTAIL-seq.
(A) Cumulative frequency plot of poly(A) measurements of spike in controls of varying tail lengths. (B) Pearson correlation between median tail-length per gene in two biological replicates (n = 12,017 genes). (C) Pearson correlation of measured transcript abundance between two biological replicates (n = 12,017 genes). (D) Functional annotations and (E) tissue enrichment profiles for genes with median tail sizes predicted to accommodate 1–2 (29–60 median tail sizes), 2–3 (61–90), 3–4 (91–120), 4–5 (121–150) and 5 or more (>151) PABPs.
Supplementary Figure 2 Range of poly(A) tails.
(A to C) Heat maps show the median maximal tail length reported for each box (A), the median minimal tail length (B) and the tail range (C) (n = 13,421 protein coding genes). Poly(A) tail measurements and frequency of optimal codons (Fop) for C. elegans transcripts are available in Supplementary Data Set 1.
Supplementary Figure 3 Short poly(A) tails are associated with well-expressed genes in Drosophila S2 cells.
(A and B) Cumulative distribution plots showing the relationship between codon optimization, poly(A) length (A) and transcript abundance (B) (n = 4,904 genes). Source data is from Subtelny et al., Nature. 508, 66–71, 2014, and summarized in Supplementary Table 2.
Supplementary Figure 4 Poly(A) tails of ribosomal protein transcripts.
(A) Cumulative distribution plots of median poly(A) tail length for the mRNAs of ribosomal proteins in S. cerevisiae (n = 56 RP and 3,526 all genes), C. elegans (n = 71 RP and 13,601 all genes), NIH3T3 (n = 67 RP and 10,107 all genes) and HeLa (n = 58 RP and 10,213 all genes). (B) Violin distribution plots with inlaid box-plots (white dot represents the median) of all tail-length measurements for transcripts of 8 ribosomal proteins in C. elegans. (C) Northern blot for poly(A) length analysis of rps-5 mRNA. The expected length of the poly(A)-fragment and an RNA marker were used to determine the poly(A) tail size in the three biological replicates. Poly(A) tail measurements and frequency of optimal codons (Fop) for C. elegans transcripts are available in Supplementary Data Set 1. References and details for the source data used in (A) are in Supplementary Table 2.
Supplementary Figure 5 Codon optimality affects poly(A) tail size.
Northern blot for poly(A) length analysis of steady state HIS3 reporter mRNAs with varying percentages of optimal codons (Radhakrishnan et al., Cell. 167, 122–132, 2016). The normalized signal intensity plotted for the 0%, 50% and 100% codon optimality lanes shows an inverse relationship between poly(A) tail size and codon optimality. Results are representative of 3 independent biological replicates.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–5 and Supplementary Table 1 (PDF 824 kb)
Supplementary Table 2
Summary and references for datasets used in the analyses (XLSX 11 kb)
Supplementary Data Set 1
Poly(A) tail measurements, abundance, Fop, DAVID and tissue enrichment analyses - Data related to Fig. 1, 2, 3; Supplementary Fig. 1, 2, 4; Supplementary Table 1 (XLSX 3784 kb)
Supplementary Data Set 2
Ribosome enrichment calculations after removing the first 50nt of ORF - Data related to Fig. 2d, 2h; Supp Table 1 (XLSX 1249 kb)
Supplementary Data Set 3
WT (L4) C. elegans RNA-seq - Data related to Supplementary Table 1 (XLSX 1156 kb)
Rights and permissions
About this article
Cite this article
Lima, S., Chipman, L., Nicholson, A. et al. Short poly(A) tails are a conserved feature of highly expressed genes. Nat Struct Mol Biol 24, 1057–1063 (2017). https://doi.org/10.1038/nsmb.3499
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb.3499
This article is cited by
-
Regulation by the RNA-binding protein Unkempt at its effector interface
Nature Communications (2024)
-
Stress responses of plants through transcriptome plasticity by mRNA alternative polyadenylation
Molecular Horticulture (2023)
-
Advances of mRNA vaccine in tumor: a maze of opportunities and challenges
Biomarker Research (2023)
-
Revolutionizing viral disease vaccination: the promising clinical advancements of non-replicating mRNA vaccines
Virology Journal (2023)
-
Short poly(A) tails are protected from deadenylation by the LARP1–PABP complex
Nature Structural & Molecular Biology (2023)