Abstract
Aggregated forms of α-synuclein play a crucial role in the pathogenesis of synucleinopathies such as Parkinson's disease (PD). However, the molecular mechanisms underlying the pathogenic effects of α-synuclein are not completely understood. Here we show that asparagine endopeptidase (AEP) cleaves human α-synuclein, triggers its aggregation and escalates its neurotoxicity, thus leading to dopaminergic neuronal loss and motor impairments in a mouse model. AEP is activated and cleaves human α-synuclein at N103 in an age-dependent manner. AEP is highly activated in human brains with PD, and it fragments α-synuclein, which is found aggregated in Lewy bodies. Overexpression of the AEP-cleaved α-synuclein1–103 fragment in the substantia nigra induces both dopaminergic neuronal loss and movement defects in mice. In contrast, inhibition of AEP-mediated cleavage of α-synuclein (wild type and A53T mutant) diminishes α-synuclein's pathologic effects. Together, these findings support AEP's role as a key mediator of α-synuclein-related etiopathological effects in PD.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Maries, E., Dass, B., Collier, T.J., Kordower, J.H. & Steece-Collier, K. The role of alpha-synuclein in Parkinson's disease: insights from animal models. Nat. Rev. Neurosci. 4, 727–738 (2003).
Polymeropoulos, M.H. et al. Mutation in the alpha-synuclein gene identified in families with Parkinson's disease. Science 276, 2045–2047 (1997).
Krüger, R. et al. Ala30Pro mutation in the gene encoding α-synuclein in Parkinson's disease. Nat. Genet. 18, 106–108 (1998).
Leroy, E., Boyer, R. & Polymeropoulos, M.H. Intron-exon structure of ubiquitin c-terminal hydrolase-L1. DNA Res. 5, 397–400 (1998).
Kitada, T. et al. Mutations in the parkin gene cause autosomal recessive juvenile parkinsonism. Nature 392, 605–608 (1998).
Spillantini, M.G. et al. α-Synuclein in Lewy bodies. Nature 388, 839–840 (1997).
Olanow, C.W. & Brundin, P. Parkinson's disease and alpha synuclein: is Parkinson's disease a prion-like disorder? Mov. Disord. 28, 31–40 (2013).
Masliah, E. et al. Dopaminergic loss and inclusion body formation in alpha-synuclein mice: implications for neurodegenerative disorders. Science 287, 1265–1269 (2000).
Kahle, P.J., Neumann, M., Ozmen, L. & Haass, C. Physiology and pathophysiology of alpha-synuclein: cell culture and transgenic animal models based on a Parkinson's disease-associated protein. Ann. NY Acad. Sci. 920, 33–41 (2000).
Feany, M.B. & Bender, W.W.A. Drosophila model of Parkinson's disease. Nature 404, 394–398 (2000).
Burré, J. et al. Properties of native brain α-synuclein. Nature 498, E4–E6 (2013).
Lashuel, H.A., Overk, C.R., Oueslati, A. & Masliah, E. The many faces of α-synuclein: from structure and toxicity to therapeutic target. Nat. Rev. Neurosci. 14, 38–48 (2013).
Li, W. et al. Aggregation promoting C-terminal truncation of alpha-synuclein is a normal cellular process and is enhanced by the familial Parkinson's disease-linked mutations. Proc. Natl. Acad. Sci. USA 102, 2162–2167 (2005).
Liu, C.W. et al. A precipitating role for truncated alpha-synuclein and the proteasome in alpha-synuclein aggregation: implications for pathogenesis of Parkinson disease. J. Biol. Chem. 280, 22670–22678 (2005).
Hoyer, W., Cherny, D., Subramaniam, V. & Jovin, T.M. Impact of the acidic C-terminal region comprising amino acids 109-140 on alpha-synuclein aggregation in vitro. Biochemistry 43, 16233–16242 (2004).
Murray, I.V. et al. Role of alpha-synuclein carboxy-terminus on fibril formation in vitro. Biochemistry 42, 8530–8540 (2003).
Mishizen-Eberz, A.J. et al. Distinct cleavage patterns of normal and pathologic forms of alpha-synuclein by calpain I in vitro. J. Neurochem. 86, 836–847 (2003).
Mishizen-Eberz, A.J. et al. Cleavage of alpha-synuclein by calpain: potential role in degradation of fibrillized and nitrated species of alpha-synuclein. Biochemistry 44, 7818–7829 (2005).
Sevlever, D., Jiang, P. & Yen, S.H. Cathepsin D is the main lysosomal enzyme involved in the degradation of alpha-synuclein and generation of its carboxy-terminally truncated species. Biochemistry 47, 9678–9687 (2008).
Takahashi, M. et al. Oxidative stress-induced phosphorylation, degradation and aggregation of alpha-synuclein are linked to upregulated CK2 and cathepsin D. Eur. J. Neurosci. 26, 863–874 (2007).
Halfon, S., Patel, S., Vega, F., Zurawski, S. & Zurawski, G. Autocatalytic activation of human legumain at aspartic acid residues. FEBS Lett. 438, 114–118 (1998).
Gamblin, T.C. et al. Caspase cleavage of tau: linking amyloid and neurofibrillary tangles in Alzheimer's disease. Proc. Natl. Acad. Sci. USA 100, 10032–10037 (2003).
Alvarez-Fernandez, M. et al. Inhibition of mammalian legumain by some cystatins is due to a novel second reactive site. J. Biol. Chem. 274, 19195–19203 (1999).
Liu, Z. et al. Neuroprotective actions of PIKE-L by inhibition of SET proteolytic degradation by asparagine endopeptidase. Mol. Cell 29, 665–678 (2008).
Madeira, A., Pommet, J.M., Prochiantz, A. & Allinquant, B. SET protein (TAF1beta, I2PP2A) is involved in neuronal apoptosis induced by an amyloid precursor protein cytoplasmic subdomain. FASEB J. 19, 1905–1907 (2005).
Basurto-Islas, G., Grundke-Iqbal, I., Tung, Y.C., Liu, F. & Iqbal, K. Activation of asparaginyl endopeptidase leads to Tau hyperphosphorylation in Alzheimer disease. J. Biol. Chem. 288, 17495–17507 (2013).
Herskowitz, J.H. et al. Asparaginyl endopeptidase cleaves TDP-43 in brain. Proteomics 12, 2455–2463 (2012).
Zhang, Z. et al. Cleavage of tau by asparagine endopeptidase mediates the neurofibrillary pathology in Alzheimer's disease. Nat. Med. 20, 1254–1262 (2014).
Zhang, Z. et al. Delta-secretase cleaves amyloid precursor protein and regulates the pathogenesis in Alzheimer's disease. Nat. Commun. 6, 8762 (2015).
Li, D.N., Matthews, S.P., Antoniou, A.N., Mazzeo, D. & Watts, C. Multistep autoactivation of asparaginyl endopeptidase in vitro and in vivo. J. Biol. Chem. 278, 38980–38990 (2003).
Chen, L. et al. Tyrosine and serine phosphorylation of alpha-synuclein have opposing effects on neurotoxicity and soluble oligomer formation. J. Clin. Invest. 119, 3257–3265 (2009).
Ellis, C.E., Schwartzberg, P.L., Grider, T.L., Fink, D.W. & Nussbaum, R.L. α-Synuclein is phosphorylated by members of the Src family of protein-tyrosine kinases. J. Biol. Chem. 276, 3879–3884 (2001).
Beyer, K. & Ariza, A. α-Synuclein posttranslational modification and alternative splicing as a trigger for neurodegeneration. Mol. Neurobiol. 47, 509–524 (2013).
Giasson, B.I. et al. Neuronal alpha-synucleinopathy with severe movement disorder in mice expressing A53T human alpha-synuclein. Neuron 34, 521–533 (2002).
Lee, M.K. et al. Human alpha-synuclein-harboring familial Parkinson's disease-linked Ala-53 → Thr mutation causes neurodegenerative disease with alpha-synuclein aggregation in transgenic mice. Proc. Natl. Acad. Sci. USA 99, 8968–8973 (2002).
Forester, B.P. et al. Age-related changes in brain energetics and phospholipid metabolism. NMR Biomed. 23, 242–250 (2010).
Gauthier, S., Kaur, G., Mi, W., Tizon, B. & Levy, E. Protective mechanisms by cystatin C in neurodegenerative diseases. Front. Biosci. (Schol. Ed.) 3, 541–554 (2011).
Dall, E. & Brandstetter, H. Structure and function of legumain in health and disease. Biochimie 122, 126–150 (2016).
Oueslati, A., Fournier, M. & Lashuel, H.A. Role of post-translational modifications in modulating the structure, function and toxicity of alpha-synuclein: implications for Parkinson's disease pathogenesis and therapies. Prog. Brain Res. 183, 115–145 (2010).
Wang, W. et al. Caspase-1 causes truncation and aggregation of the Parkinson's disease-associated protein α-synuclein. Proc. Natl. Acad. Sci. USA 113, 9587–9592 (2016).
Shirahama-Noda, K. et al. Biosynthetic processing of cathepsins and lysosomal degradation are abolished in asparaginyl endopeptidase-deficient mice. J. Biol. Chem. 278, 33194–33199 (2003).
Volpicelli-Daley, L.A., Luk, K.C. & Lee, V.M. Addition of exogenous α-synuclein preformed fibrils to primary neuronal cultures to seed recruitment of endogenous α-synuclein to Lewy body and Lewy neurite-like aggregates. Nat. Protoc. 9, 2135–2146 (2014).
Zhang, Z. et al. 7,8-dihydroxyflavone prevents synaptic loss and memory deficits in a mouse model of Alzheimer's disease. Neuropsychopharmacology 39, 638–650 (2014).
Pozdeyev, N. et al. Dopamine modulates diurnal and circadian rhythms of protein phosphorylation in photoreceptor cells of mouse retina. Eur. J. Neurosci. 27, 2691–2700 (2008).
Lowry, O.H., Rosebrough, N.J., Farr, A.L. & Randall, R.J. Protein measurement with the Folin phenol reagent. J. Biol. Chem. 193, 265–275 (1951).
Acknowledgements
This work was supported by grants from the Michael J. Fox Foundation (grant ID 11137) to K.Y.; a grant from the National Natural Science Foundation (NSFC) of China (no. 81571249) to Zhentao Zhang; NSFC grant (no. 81528007) to K.Y. and J.-Z.W.; a National Key Basic Research Program of China grant (2010CB945202) to Y.E.S.; an NSFC grant (81330030) to Y.E.S.; and grants from the US Public Health Service (P30EY006360 and R01EY004864) to P.M.I. We thank the ADRC at Emory University for providing human PD, LBD and healthy-control samples, and C. Watts (University of Cambridge) for providing anti-AEP.
Author information
Authors and Affiliations
Contributions
K.Y. conceived the project, designed the experiments and wrote the manuscript. Zhentao Zhang designed and performed most of the experiments. S.S.K. and X.L. prepared primary neurons and assisted with animal experiments. M.J.B. and F.P.M. provided clones and packaged viral vectors. D.M.D. and N.T.S. performed the mass spectrometry analysis. L.H. and P.M.I. performed the HPLC experiments and critically read and edited the manuscript. Zhaohui Zhang, E.H.A., L.J., Y.E.S., F.P.M. and J.-Z.W. designed the experiments, assisted with data analysis and interpretation and critically read the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
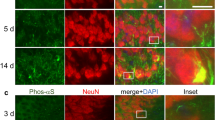
Supplementary Figure 1 α-synuclein is truncated in PD and LBD brains.
(a) Specificity of anti-N103 antibody. Anti-N103 antibody was coated on an ELISA plate and different concentrations of full-length α-synuclein or 1-103 fragment (0-16,000 ng/ml) was added to the the system. The signal was detected using the anti-α-synuclein N-terminal antibody. (b) Localization of α-synuclein N103 fragments in synaptic structures. LBD brain sections were immunostained with anti-α-synuclein N103 (red) and presynaptic marker synapsin I (green). Scale bar, 20 μm. Images are representative of 9 sections from three subjects. SNCA knockout mice brain was used as negative control. (c-d) Western blot showing the presence of α-synuclein N103 fragment in brain lysates from human LBD cortex and PD SN tissues (mean ± SEM; n = 6, *P < 0.05 compared with control, student’s t-test). The shown blots are the representative figures of three independent experiments. (e) The correlation between N103 fragment concentration and AEP activity.
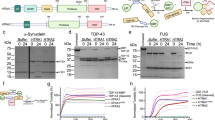
Supplementary Figure 2 α-synuclein is cleaved at N103 by AEP.
(a) Recombinant mammalian glutathione transferase (mGST)-tagged α-synuclein was incubated with purified active AEP, and analyzed by immunoblotting. The AEP-derived α-synuclein fragment shows the same molecular weight as a.a. 1-103 fragment. The AEP-derived α-synuclein fragment was recognized by the anti-α-synuclein N103 antibody. (b) MS/MS spectrum showing that AEP cleaves α-synuclein after N103 in vitro. The purified mGST-α-synuclein was incubated with active AEP for 30 min. The fragment was subject to MS/MS spectrum assay. (c-d) Cleavage of mutant N-terminal GFP-tagged α-synuclein or C-terminal GFP-tagged α-synuclein by AEP. α-synuclein cleavage was analyzed by Western blot after recombinant α-synuclein wide-type, N65A, N103A, or N122A mutants were incubated with active mouse kidney lysates for 15 min. (e) Comparison of protein sequences of human and mouse α-synuclein. (f) Cleavage of human and mouse α-synuclein by recombinant AEP. The shown blots are the representative figures of three independent experiments.
Supplementary Figure 3 AEP interacts with α-synuclein.
(a) Co-immunoprecipitation of α-synuclein and AEP in PD brain samples. α-synuclein was immunoprecipitated with anti-α-synuclein N-terminal antibody, and analyzed by immunoblotting with anti-AEP antibody. (b) Anti-AEP antibody abolishes the cleavage of α-synuclein by AEP. Anti-AEP antibody or control IgG was added into human brain lysates and incubated at pH 6.0 for 15 min. The proteolytic processing of α-synuclein was analyzed using Western blot. The shown blots are the representative figures of three independent experiments. Data represent mean ± SEM of three experiments. n = 3, *P < 0.05, one-way ANOVA. (c) Immunostaining with lysosomal marker LAMP1 and AEP showing strict lysosomal localization of AEP in the control brain slides and diffuse staining of AEP in PD brain slides. Scale bar, 20 μm. (d) AEP activity in lysosomal and cytoplasmic fractions of control and PD brain samples (mean ± SEM; n = 3, *P < 0.05, one-way ANOVA). (e) Cleavage of recombinant α-synuclein by lysosomal and cytoplasmic fraction. Recombinant α-synuclein was incubated in lysosomal and cytoplasmic fraction of human brain tissue at pH 6.0 for 0, 5 and 10 min. The production of α-synuclein N103 fragment was analyzed using Western blot.
Supplementary Figure 4 The cleavage of α-synuclein by AEP is independent of its phosphorylation and mutations.
(a) Cleavage rate of α-synuclein S129A and S129D mutations by AEP. HEK293 cells were transfected with GFP-α-synuclein wild-type, S129A, and S129D mutations of α-synuclein, and incubated with active kidney lysates for 0, 5, 10, or 20 min at pH 6.0, and analyzed by immunoblotting. (b) Cleavage rate of α-synuclein Y125F mutation by AEP. (c) Overexpression of constitutively active Fyn induced the phosphorylation of α-synuclein at Y125. (d) Cleavage assay indicates that constitutively active (CA) Fyn or kinase dead (KD) Fyn does not affect the cleavage rate of α-synuclein. (e) A30P and A53T mutant α-synuclein were cleaved by AEP at a similar rate as wild-type (WT) α-synuclein. (f) Cleavage of α-synuclein by AEP was not blocked by calpain, cathepsin or protease inhibitor cocktail. HEK293 cells were transfected with GFP-α-synuclein. The cell lysates were incubated with active kidney lysates at 37°C for 10 min in the presence of calpain inhibitor ALLN, cathepsin inhibitor E64, protease inhibitor cocktail, or AEP inhibitor AENK. α-synuclein cleavage was analyzed by Western blot. Only AEP inhibitory peptide AENK but not any other small molecular inhibitors antagonized α-synuclein cleavage by AEP. The shown blots are the representative figures of three independent experiments. The shown blots are the representative figures of three independent experiments. Data represent mean ± SEM of three experiments. *P < 0.05, one-way ANOVA.
Supplementary Figure 5 AEP activity assay in wild-type mice.
AEP activity is escalated in the cortex and SN tissues of wild-type mice in an age-dependent style (mean ± SEM; n = 6, *P < 0.05, one-way ANOVA). AFU, arbitrary fluorescence units.
Supplementary Figure 6 Aggregation of full-length α-synuclein and 1–103 fragments in vitro.
(a) Electron microscopy visualizing the aggregated full-length α-synuclein and 1-103 fragment. Scale bar, 100 nm. (b) Immunostaining showing the presence of 1-103 fragment in neurons. Scale bar, 20 μm. (c) Western blots showing the localization of α-synuclein in the cytosol and nuclear fraction. Most of full-length α-synuclein and its 1-103 fragment localizes in the cytosol fraction.
Supplementary Figure 7 Expression of α-synuclein full-length or 1–103 fragments induces intraneuronal inclusions containing ubiquitin and 14-3-3.
(a) Immunohistochemistry using anti-human α-synuclein antibody (LB509) showing the expressing of FL α-synuclein. Scale bar, 200 μm. (b) Immunostaining using anti-α-synuclein N103 antibody showing the expression of α-synuclein 1-103 fragment. Scale bar, 20 μm. (c-f) Brain slides injected with AAVs encoding α-synuclein full-length (c, d) or 1-103 (e, f) were immunostained with α-synuclein antibody (c, d), anti-α-synuclein N103 antibody (e, f), 14-3-3 antibody (c, e) and ubiquitin antibody (d, f). (g) Relative AEP activity in lysosomal and cytosolic fractions of SN tissue from control mice and mice injected with AAV-α-synuclein FL and AAV-α-synuclein A53T (mean ± SEM; n = 3, *P < 0.05, one-way ANOVA).
Supplementary Figure 8 Expression level of N103 fragments.
Western blot shows the expression level of α-synuclein 1-103 fragments in SN tissues from PD patients, mice SN tissue injected with human α-synuclein virus, and mice SN tissue injected with α-synuclein 1-103 fragment virus (mean ± SEM; n = 3, *P < 0.05, student’s t-test).
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8. (PDF 1716 kb)
Supplementary Data Set 1
Uncropped images of gels. (PDF 13158 kb)
Rights and permissions
About this article
Cite this article
Zhang, Z., Kang, S., Liu, X. et al. Asparagine endopeptidase cleaves α-synuclein and mediates pathologic activities in Parkinson's disease. Nat Struct Mol Biol 24, 632–642 (2017). https://doi.org/10.1038/nsmb.3433
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb.3433
This article is cited by
-
Inhibition of asparagine endopeptidase (AEP) effectively treats sporadic Alzheimer’s disease in mice
Neuropsychopharmacology (2024)
-
Toxic interactions between dopamine, α-synuclein, monoamine oxidase, and genes in mitochondria of Parkinson’s disease
Journal of Neural Transmission (2024)
-
Implications of COVID-19 in Parkinson’s disease: the purinergic system in a therapeutic-target perspective to diminish neurodegeneration
Purinergic Signalling (2024)
-
L116 Deletion in CSPα Promotes α-Synuclein Aggregation and Neurodegeneration
Molecular Neurobiology (2024)
-
Development and validation of an expanded antibody toolset that captures alpha-synuclein pathological diversity in Lewy body diseases
npj Parkinson's Disease (2023)