Abstract
Direct interactions with lipids have emerged as key determinants of the folding, structure and function of membrane proteins, but an understanding of how lipids modulate protein dynamics is still lacking. Here, we systematically explored the effects of lipids on the conformational dynamics of the proton-powered multidrug transporter LmrP from Lactococcus lactis, using the pattern of distances between spin-label pairs previously shown to report on alternating access of the protein. We uncovered, at the molecular level, how the lipid headgroups shape the conformational-energy landscape of the transporter. The model emerging from our data suggests a direct interaction between lipid headgroups and a conserved motif of charged residues that control the conformational equilibrium through an interplay of electrostatic interactions within the protein. Together, our data lay the foundation for a comprehensive model of secondary multidrug transport in lipid bilayers.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Singer, S.J. & Nicolson, G.L. The fluid mosaic model of the structure of cell membranes. Science 175, 720–731 (1972).
Heijne, G. The distribution of positively charged residues in bacterial inner membrane proteins correlates with the trans-membrane topology. EMBO J. 5, 3021–3027 (1986).
Cantor, R.S. The influence of membrane lateral pressures on simple geometric models of protein conformational equilibria. Chem. Phys. Lipids 101, 45–56 (1999).
Powl, A.M., East, J.M. & Lee, A.G. Anionic phospholipids affect the rate and extent of flux through the mechanosensitive channel of large conductance MscL. Biochemistry 47, 4317–4328 (2008).
Phillips, R., Ursell, T., Wiggins, P. & Sens, P. Emerging roles for lipids in shaping membrane-protein function. Nature 459, 379–385 (2009).
Dowhan, W. & Bogdanov, M. Lipid-protein interactions as determinants of membrane protein structure and function. Biochem. Soc. Trans. 39, 767–774 (2011).
Hanson, M.A. et al. A specific cholesterol binding site is established by the 2.8 A structure of the human beta2-adrenergic receptor. Structure 16, 897–905 (2008).
Hunte, C. & Richers, S. Lipids and membrane protein structures. Curr. Opin. Struct. Biol. 18, 406–411 (2008).
Lee, A.G. Lipid-protein interactions in biological membranes: a structural perspective. Biochim. Biophys. Acta 1612, 1–40 (2003).
Ellena, J.F., Blazing, M.A. & McNamee, M.G. Lipid-protein interactions in reconstituted membranes containing acetylcholine receptor. Biochemistry 22, 5523–5535 (1983).
Fernandez-Ballester, G. et al. Role of cholesterol as a structural and functional effector of the nicotinic acetylcholine receptor. Biochem. Soc. Trans. 22, 776–780 (1994).
Contreras, F.X. et al. Molecular recognition of a single sphingolipid species by a protein's transmembrane domain. Nature 481, 525–529 (2012).
Lee, A.G. Lipid-protein interactions. Biochem. Soc. Trans. 39, 761–766 (2011).
Vitrac, H., Bogdanov, M. & Dowhan, W. Proper fatty acid composition rather than an ionizable lipid amine is required for full transport function of lactose permease from Escherichia coli. J. Biol. Chem. 288, 5873–5885 (2013).
Laganowsky, A. et al. Membrane proteins bind lipids selectively to modulate their structure and function. Nature 510, 172–175 (2014).
Pliotas, C. et al. The role of lipids in mechanosensation. Nat. Struct. Mol. Biol. 22, 991–998 (2015).
Koshy, C. et al. Structural evidence for functional lipid interactions in the betaine transporter BetP. EMBO J. 32, 3096–3105 (2013).
Dawaliby, R. et al. Allosteric regulation of G protein-coupled receptor activity by phospholipids. Nat. Chem. Biol. 12, 35–39 (2016).
Hamilton, P.J. et al. PIP2 regulates psychostimulant behaviors through its interaction with a membrane protein. Nat. Chem. Biol. 10, 582–589 (2014).
Khelashvili, G. & Weinstein, H. Functional mechanisms of neurotransmitter transporters regulated by lipid-protein interactions of their terminal loops. Biochim. Biophys. Acta 1848, 1765–1774 (2015).
Khelashvili, G., Galli, A. & Weinstein, H. Phosphatidylinositol 4,5-biphosphate (PIP2) lipids regulate the phosphorylation of syntaxin N-terminus by modulating both its position and local structure. Biochemistry 51, 7685–7698 (2012).
Sachs, J.N. & Engelman, D.M. Introduction to the membrane protein reviews: the interplay of structure, dynamics, and environment in membrane protein function. Annu. Rev. Biochem. 75, 707–712 (2006).
Bolhuis, H. et al. Energetics and mechanism of drug transport mediated by the lactococcal multidrug transporter LmrP. J. Biol. Chem. 271, 24123–24128 (1996).
Putman, M., van Veen, H.W., Degener, J.E. & Konings, W.N. The lactococcal secondary multidrug transporter LmrP confers resistance to lincosamides, macrolides, streptogramins and tetracyclines. Microbiology 147, 2873–2880 (2001).
Gottesman, M.M. & Pastan, I. Biochemistry of multidrug resistance mediated by the multidrug transporter. Annu. Rev. Biochem. 62, 385–427 (1993).
Putman, M., van Veen, H.W. & Konings, W.N. Molecular properties of bacterial multidrug transporters. Microbiol. Mol. Biol. Rev. 64, 672–693 (2000).
Fluman, N., Ryan, C.M., Whitelegge, J.P. & Bibi, E. Dissection of mechanistic principles of a secondary multidrug efflux protein. Mol. Cell 47, 777–787 (2012).
Schaedler, T.A. & van Veen, H.W. A flexible cation binding site in the multidrug major facilitator superfamily transporter LmrP is associated with variable proton coupling. FASEB J. 24, 3653–3661 (2010).
Schuldiner, S. EmrE, a model for studying evolution and mechanism of ion-coupled transporters. Biochim. Biophys. Acta 1794, 748–762 (2009).
Paulsen, I.T., Brown, M.H., Littlejohn, T.G., Mitchell, B.A. & Skurray, R.A. Multidrug resistance proteins QacA and QacB from Staphylococcus aureus: membrane topology and identification of residues involved in substrate specificity. Proc. Natl. Acad. Sci. USA 93, 3630–3635 (1996).
Kaatz, G.W. & Seo, S.M. Inducible NorA-mediated multidrug resistance in Staphylococcus aureus. Antimicrob. Agents Chemother. 39, 2650–2655 (1995).
Masureel, M. et al. Protonation drives the conformational switch in the multidrug transporter LmrP. Nat. Chem. Biol. 10, 149–155 (2014).
Claxton, D.P., Kazmier, K., Mishra, S. & Mchaourab, H.S. Navigating membrane protein structure, dynamics, and energy landscapes using spin labeling and EPR spectroscopy. Methods Enzymol. 564, 349–387 (2015).
Pao, S.S., Paulsen, I.T. & Saier, M.H. Jr. Major facilitator superfamily. Microbiol. Mol. Biol. Rev. 62, 1–34 (1998).
Jiang, D. et al. Structure of the YajR transporter suggests a transport mechanism based on the conserved motif A. Proc. Natl. Acad. Sci. USA 110, 14664–14669 (2013).
Zou, P. & McHaourab, H.S. Increased sensitivity and extended range of distance measurements in spin-labeled membrane proteins: Q-band double electron-electron resonance and nanoscale bilayers. Biophys. J. 98, L18–L20 (2010).
Bayburt, T.H. & Sligar, S.G. Self-assembly of single integral membrane proteins into soluble nanoscale phospholipid bilayers. Protein Sci. 12, 2476–2481 (2003).
Putman, M., van Veen, H.W., Poolman, B. & Konings, W.N. Restrictive use of detergents in the functional reconstitution of the secondary multidrug transporter LmrP. Biochemistry 38, 1002–1008 (1999).
Gbaguidi, B. et al. Proton motive force mediates a reorientation of the cytosolic domains of the multidrug transporter LmrP. Cell. Mol. Life Sci. 61, 2646–2657 (2004).
Stein, R.A., Beth, A.H. & Hustedt, E.J. A straightforward approach to the analysis of double electron-electron resonance data. Methods Enzymol. 563, 531–567 (2015).
Mishra, S. et al. Conformational dynamics of the nucleotide binding domains and the power stroke of a heterodimeric ABC transporter. eLife 3, e02740 (2014).
Hakizimana, P., Masureel, M., Gbaguidi, B., Ruysschaert, J.M. & Govaerts, C. Interactions between phosphatidylethanolamine headgroup and LmrP, a multidrug transporter: a conserved mechanism for proton gradient sensing? J. Biol. Chem. 283, 9369–9376 (2008).
Denisov, I.G., McLean, M.A., Shaw, A.W., Grinkova, Y.V. & Sligar, S.G. Thermotropic phase transition in soluble nanoscale lipid bilayers. J. Phys. Chem. B 109, 15580–15588 (2005).
Lanucara, F., Holman, S.W., Gray, C.J. & Eyers, C.E. The power of ion mobility-mass spectrometry for structural characterization and the study of conformational dynamics. Nat. Chem. 6, 281–294 (2014).
Zhou, M. et al. Mass spectrometry of intact V-type ATPases reveals bound lipids and the effects of nucleotide binding. Science 334, 380–385 (2011).
Bechara, C. et al. A subset of annular lipids is linked to the flippase activity of an ABC transporter. Nat. Chem. 7, 255–262 (2015).
Adamberg, K., Kask, S., Laht, T.M. & Paalme, T. The effect of temperature and pH on the growth of lactic acid bacteria: a pH-auxostat study. Int. J. Food Microbiol. 85, 171–183 (2003).
Bibal, B., Goma, G., Vayssier, Y. & Pareilleux, A. Influence of pH, lactose and lactic acid on the growth of Streptococcus cremoris: a kinetic study. Appl. Microbiol. Biotechnol. 28, 340–344 (1988).
Chen, C.C. & Wilson, T.H. The phospholipid requirement for activity of the lactose carrier of Escherichia coli. J. Biol. Chem. 259, 10150–10158 (1984).
Driessen, A.J., Zheng, T., In't Veld, G., Op den Kamp, J.A. & Konings, W.N. Lipid requirement of the branched-chain amino acid transport system of Streptococcus cremoris. Biochemistry 27, 865–872 (1988).
Powl, A.M., East, J.M. & Lee, A.G. Importance of direct interactions with lipids for the function of the mechanosensitive channel MscL. Biochemistry 47, 12175–12184 (2008).
Haines, T.H. A new look at Cardiolipin. Biochim. Biophys. Acta 1788, 1997–2002 (2009).
Shintre, C.A. et al. Structures of ABCB10, a human ATP-binding cassette transporter in apo- and nucleotide-bound states. Proc. Natl. Acad. Sci. USA 110, 9710–9715 (2013).
Nury, H. et al. Structural basis for lipid-mediated interactions between mitochondrial ADP/ATP carrier monomers. FEBS Lett. 579, 6031–6036 (2005).
Ruprecht, J.J. et al. Structures of yeast mitochondrial ADP/ATP carriers support a domain-based alternating-access transport mechanism. Proc. Natl. Acad. Sci. USA 111, E426–E434 (2014).
Lange, C., Nett, J.H., Trumpower, B.L. & Hunte, C. Specific roles of protein-phospholipid interactions in the yeast cytochrome bc1 complex structure. EMBO J. 20, 6591–6600 (2001).
Pöyry, S. et al. Atomistic simulations indicate cardiolipin to have an integral role in the structure of the cytochrome bc1 complex. Biochim. Biophys. Acta 1827, 769–778 (2013).
Kates, M., Syz, J.Y., Gosser, D. & Haines, T.H. pH-dissociation characteristics of cardiolipin and its 2′-deoxy analogue. Lipids 28, 877–882 (1993).
Haines, T.H. & Dencher, N.A. Cardiolipin: a proton trap for oxidative phosphorylation. FEBS Lett. 528, 35–39 (2002).
Lensink, M.F., Govaerts, C. & Ruysschaert, J.M. Identification of specific lipid-binding sites in integral membrane proteins. J. Biol. Chem. 285, 10519–10526 (2010).
Yin, Y., He, X., Szewczyk, P., Nguyen, T. & Chang, G. Structure of the multidrug transporter EmrD from Escherichia coli. Science 312, 741–744 (2006).
Guan, L., Mirza, O., Verner, G., Iwata, S. & Kaback, H.R. Structural determination of wild-type lactose permease. Proc. Natl. Acad. Sci. USA 104, 15294–15298 (2007).
Dang, S. et al. Structure of a fucose transporter in an outward-open conformation. Nature 467, 734–738 (2010).
Thompson, J.D., Higgins, D.G. & Gibson, T.J. CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res. 22, 4673–4680 (1994).
Webb, B. & Sali, A. Comparative protein structure modeling using MODELLER. Curr. Protoc. Bioinformatics 47, 5.6 (2014).
Pettersen, E.F. et al. UCSF Chimera: a visualization system for exploratory research and analysis. J. Comput. Chem. 25, 1605–1612 (2004).
Mazurkiewicz, P., Driessen, A.J. & Konings, W.N. Energetics of wild-type and mutant multidrug resistance secondary transporter LmrP of Lactococcus lactis. Biochim. Biophys. Acta 1658, 252–261 (2004).
Putman, M., Koole, L.A., van Veen, H.W. & Konings, W.N. The secondary multidrug transporter LmrP contains multiple drug interaction sites. Biochemistry 38, 13900–13905 (1999).
Mierau, I. & Kleerebezem, M. 10 years of the nisin-controlled gene expression system (NICE) in Lactococcus lactis. Appl. Microbiol. Biotechnol. 68, 705–717 (2005).
Boldog, T., Li, M. & Hazelbauer, G.L. Using Nanodiscs to create water-soluble transmembrane chemoreceptors inserted in lipid bilayers. Methods Enzymol. 423, 317–335 (2007).
Jeschke, G. & Polyhach, Y. Distance measurements on spin-labelled biomacromolecules by pulsed electron paramagnetic resonance. Phys. Chem. Chem. Phys. 9, 1895–1910 (2007).
Jeschke, G. DEER distance measurements on proteins. Annu. Rev. Phys. Chem. 63, 419–446 (2012).
Pannier, M., Veit, S., Godt, A., Jeschke, G. & Spiess, H.W. Dead-time free measurement of dipole-dipole interactions between electron spins. J. Magn. Reson. 142, 331–340 (2000).
Jeschke, G. et al. DeerAnalysis2006: a comprehensive software package for analyzing pulsed ELDOR data. Appl. Magn. Reson. 30, 473–498 (2006).
Brandon, S., Beth, A.H. & Hustedt, E.J. The global analysis of DEER data. J. Magn. Reson. 218, 93–104 (2012).
Sobott, F., Hernández, H., McCammon, M.G., Tito, M.A. & Robinson, C.V. A tandem mass spectrometer for improved transmission and analysis of large macromolecular assemblies. Anal. Chem. 74, 1402–1407 (2002).
Bligh, E.G. & Dyer, W.J. A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol. 37, 911–917 (1959).
Steed, P.R., Zou, P., Trone, K.E. & Mchaourab, H.S. Structure and pH-induced structural rearrangements of the putative multidrug efflux pump EmrD in liposomes probed by site-directed spin labeling. Biochemistry 52, 7964–7974 (2013).
Acknowledgements
We thank D. Claxton, R. Dastvan and D. Hilger for critically reading the manuscript, V. Debruycker (ULB) for providing the solubilized protein used for the MS measurements and J.M. Ruysschaert for helpful discussions. This work was supported by the Fonds de la Recherche Scientifique F.R.S.–F.N.R.S. (grant F.4523.12 to C.G.) and the NIH (grant R01GM077659 to H.S.M.) C.M. is supported as a Research Fellow of the F.R.I.A. C.G. is supported as a Research Associate of the F.R.S.–F.N.R.S. and as a Welbio Investigator.
Author information
Authors and Affiliations
Contributions
C.M., H.S.M. and C.G. performed experimental design. M.M., S.M., A.R. and C.M. performed mutagenesis, expression, activity, purification, reconstitution and labeling experiments. R.A.S. performed EPR measurements. R.A.S., C.M. and H.S.M. performed DEER data analysis. C.G. and C.M. performed molecular modeling. C.M. and A.R. performed transport assays. A.K. and F.S. performed native MS measurements and data analysis. R.D. performed LC-MS/MS measurements and analysis. C.G. and H.S.M. oversaw all aspects of the experiments and manuscript preparation. All authors participated in interpreting the data and writing the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 Limited ligand-dependent conformational changes within the N or C lobe of LmrP.
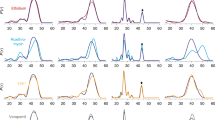
DEER distance distributions for spin-labeled cysteine pairs located on the extracellular (left panels) and cytoplasmic (right panels) at ends of TM helices, within a same lobe, at pH8 (black), pH6 (red), pH8 + Hoechst 33342 (blue) and pH8 + EtBr (orange). Residue numbers and positions are depicted on an LmrP model viewed from the extracellular (top left) or cytoplasmic (top right) side. The N-lobe is blue and the C-lobe is grey.
Supplementary Figure 2 Transport activity assay of double-cysteine mutants.
LmrP-dependent Hoechst 33342 extrusion is monitored in inside-out membrane vesicles by a fluorescence-based assay (see Methods). Values were normalized for fluorescence intensity and total protein amount. Addition of ATP activates the endogenous F0/F1 – ATPase leading to formation of a transmembrane proton gradient, thus triggering LmrP activity. All the double cysteine mutants retained significant activity, compared to the D68C mutant used as a negative control.
Supplementary Figure 3 EtBr binding induces minor conformational changes.
DEER distance distributions for spin-labeled cysteine pairs located on the extracellular (top panels) and cytoplasmic (bottom panels) ends of TM helices. No significant changes are observed at pH8 in the absence (black) and in presence of 1mM EtBr (orange), except for the 256R1-310R1 pair, reporting substrate binding. For this pair, substrate binding leads to narrowing of the distance distribution. Targeted helices are highlighted in orange with TM numbers indicated atop, on an LmrP model viewed from the extracellular (top) or cytoplasmic (bottom) side. The N-lobe is blue and the C-lobe is grey.
Supplementary Figure 4 Uncoupling of the charge-relay network in detergent.
(a) Crystal structure of the LmrP homolog YajR in the outward-open conformation. The charge-relay network of conserved residues is highlighted. TM2 and TM11 are displayed in orange. (b). the single mutations D68N, R72K and D128N were combined with extracellular (160R1-310R1) and (c) cytoplasmic (137R1-349R1) reporters. DEER distance distributions at pH5 (red) pH8 (black) and pH8 + Hoechst 33342 (blue) in the absence (dashed line) and presence (solid line) of each mutation.
Supplementary Figure 5 Phospholipid headgroup modulates the pH-dependent conformational equilibrium.
DEER distance distributions of the 160R1-310R1 (upper half) and the 137R1-349R1 (lower half) pairs, used as extracellular and cytoplasmic distance reporters, respectively, reconstituted in nanodiscs composed of DOPE-DOPG-CL (grey), DOPE(Me)2-DOPG-CL (blue) and DOPC-DOPG-CL (orange), at four pHs values ranging from 5 to 8.
Supplementary Figure 6 Various endogenous cardiolipin species bind to LmrP.
(a) Mass spectrum of detergent-solubilized LmrP sample (native MS). Red rectangle: Cardiolipin molecules ejected from LmrP upon increased voltage. Blue rectangle: LmrP with (red arrows) and without (blue labels) cardiolipin(s) bound. (b) Lipids quantification by liquid chromatography and tandem mass spectrometry (n=3, Error bars: s.e.m). Red: lipids extracted from L.lactis membranes. Blue: lipids co-purified with LmrP.
Supplementary Figure 7 The conformational equilibrium of LmrP is not affected by changes in the ionic strength.
DEER distance distributions of the 160R1-310R1 pair in E.coli polar lipids nanodiscs (left) and detergent micelles (right) at NaCl concentrations ranging from a third to the double of the usual concentration.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–7 (PDF 1271 kb)
Supplementary Data Set 1
EmrD based homology model (TXT 245 kb)
Supplementary Data Set 2
LacY based homology model (TXT 245 kb)
Supplementary Data Set 3
FucP based homology model (TXT 167 kb)
Supplementary Data Set 4
YajR based homology model (TXT 247 kb)
Supplementary Data Set 5
Continuous-wave EPR spectra (TIFF 237 kb)
Supplementary Data Set 6
DEER data analysis (TIFF 423 kb)
Supplementary Data Set 7
DEER data analysis continued (TIFF 467 kb)
Rights and permissions
About this article
Cite this article
Martens, C., Stein, R., Masureel, M. et al. Lipids modulate the conformational dynamics of a secondary multidrug transporter. Nat Struct Mol Biol 23, 744–751 (2016). https://doi.org/10.1038/nsmb.3262
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb.3262