Abstract
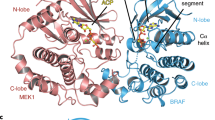
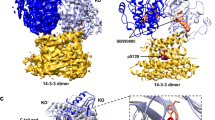
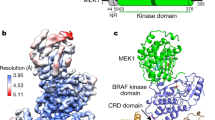
Reported RAF kinase domain structures adopt a side-to-side dimer configuration reflective of an 'on' state that underpins an allosteric mechanism of regulation. Atomic details of the monomer 'off' state have been elusive. Reinspection of the BRAF kinase domain structures revealed that sulfonamide inhibitors induce features of an off state, primarily a laterally displaced helix αC stabilized by the activation segment helix 1 (AS-H1). These features correlated with the ability of sulfonamides to disrupt human BRAF homodimers in cells, in vitro and in crystals yielding a structure of BRAF in a monomer state. The crystal structure revealed exaggerated, nonproductive positions of helix αC and AS-H1, the latter of which is the target of potent BRAF oncogenic mutations. Together, this work provides formal proof of an allosteric link between the RAF dimer interface, the activation segment and the catalytic infrastructure.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Rajakulendran, T., Sahmi, M., Lefrancois, M., Sicheri, F. & Therrien, M. A dimerization-dependent mechanism drives RAF catalytic activation. Nature 461, 542–545 (2009).
Zhang, B.H. & Guan, K.L. Activation of B-Raf kinase requires phosphorylation of the conserved residues Thr598 and Ser601. EMBO J. 19, 5429–5439 (2000).
Chong, H., Lee, J. & Guan, K.L. Positive and negative regulation of Raf kinase activity and function by phosphorylation. EMBO J. 20, 3716–3727 (2001).
Freeman, A.K., Ritt, D.A. & Morrison, D.K. Effects of Raf dimerization and its inhibition on normal and disease-associated Raf signaling. Mol. Cell 49, 751–758 (2013).
Wan, P.T. et al. Mechanism of activation of the RAF-ERK signaling pathway by oncogenic mutations of B-RAF. Cell 116, 855–867 (2004).
Heidorn, S.J. et al. Kinase-dead BRAF and oncogenic RAS cooperate to drive tumor progression through CRAF. Cell 140, 209–221 (2010).
Poulikakos, P.I., Zhang, C., Bollag, G., Shokat, K.M. & Rosen, N. RAF inhibitors transactivate RAF dimers and ERK signalling in cells with wild-type BRAF. Nature 464, 427–430 (2010).
Hatzivassiliou, G. et al. RAF inhibitors prime wild-type RAF to activate the MAPK pathway and enhance growth. Nature 464, 431–435 (2010).
Garnett, M.J. & Marais, R. Guilty as charged: B-RAF is a human oncogene. Cancer Cell 6, 313–319 (2004).
Kandoth, C. et al. Mutational landscape and significance across 12 major cancer types. Nature 502, 333–339 (2013).
Forbes, S.A. et al. COSMIC: mining complete cancer genomes in the Catalogue of Somatic Mutations in Cancer. Nucleic Acids Res. 39, D945–D950 (2011).
Zhang, X., Gureasko, J., Shen, K., Cole, P.A. & Kuriyan, J. An allosteric mechanism for activation of the kinase domain of epidermal growth factor receptor. Cell 125, 1137–1149 (2006).
Shi, F., Telesco, S.E., Liu, Y., Radhakrishnan, R. & Lemmon, M.A. ErbB3/HER3 intracellular domain is competent to bind ATP and catalyze autophosphorylation. Proc. Natl. Acad. Sci. USA 107, 7692–7697 (2010).
Jeffrey, P.D. et al. Mechanism of CDK activation revealed by the structure of a cyclinA-CDK2 complex. Nature 376, 313–320 (1995).
Tsai, J. et al. Discovery of a selective inhibitor of oncogenic B-Raf kinase with potent antimelanoma activity. Proc. Natl. Acad. Sci. USA 105, 3041–3046 (2008).
Bollag, G. et al. Clinical efficacy of a RAF inhibitor needs broad target blockade in BRAF-mutant melanoma. Nature 467, 596–599 (2010).
De Bondt, H.L. et al. Crystal structure of cyclin-dependent kinase 2. Nature 363, 595–602 (1993).
Kornev, A.P., Haste, N.M., Taylor, S.S. & Eyck, L.F. Surface comparison of active and inactive protein kinases identifies a conserved activation mechanism. Proc. Natl. Acad. Sci. USA 103, 17783–17788 (2006).
Lavoie, H. et al. Inhibitors that stabilize a closed RAF kinase domain conformation induce dimerization. Nat. Chem. Biol. 9, 428–436 (2013).
Luo, Z. et al. Oligomerization activates c-Raf-1 through a Ras-dependent mechanism. Nature 383, 181–185 (1996).
Weber, C.K., Slupsky, J.R., Kalmes, H.A. & Rapp, U.R. Active Ras induces heterodimerization of cRaf and BRaf. Cancer Res. 61, 3595–3598 (2001).
Röring, M. et al. Distinct requirement for an intact dimer interface in wild-type, V600E and kinase-dead B-Raf signalling. EMBO J. 31, 2629–2647 (2012).
Poulikakos, P.I. et al. RAF inhibitor resistance is mediated by dimerization of aberrantly spliced BRAF(V600E). Nature 480, 387–390 (2011).
Shan, Y. et al. Oncogenic mutations counteract intrinsic disorder in the EGFR kinase and promote receptor dimerization. Cell 149, 860–870 (2012).
Arkhipov, A. et al. Architecture and membrane interactions of the EGF receptor. Cell 152, 557–569 (2013).
Endres, N.F. et al. Conformational coupling across the plasma membrane in activation of the EGF receptor. Cell 152, 543–556 (2013).
Tran, N.H., Wu, X. & Frost, J.A. B-Raf and Raf-1 are regulated by distinct autoregulatory mechanisms. J. Biol. Chem. 280, 16244–16253 (2005).
Chong, H. & Guan, K.L. Regulation of Raf through phosphorylation and N terminus-C terminus interaction. J. Biol. Chem. 278, 36269–36276 (2003).
Wenglowsky, S. et al. Pyrazolopyridine inhibitors of B-Raf(V600E). Part 1: the development of selective, orally bioavailable, and efficacious inhibitors. ACS Med. Chem. Lett. 2, 342–347 (2011).
Vonrhein, C. et al. Data processing and analysis with the autoPROC toolbox. Acta Crystallogr. D Biol. Crystallogr. 67, 293–302 (2011).
Adams, P.D. et al. PHENIX: a comprehensive Python-based system for macromolecular structure solution. Acta Crystallogr. D Biol. Crystallogr. 66, 213–221 (2010).
Emsley, P., Lohkamp, B., Scott, W.G. & Cowtan, K. Features and development of Coot. Acta Crystallogr. D Biol. Crystallogr. 66, 486–501 (2010).
Douziech, M. et al. Bimodal regulation of RAF by CNK in Drosophila. EMBO J. 22, 5068–5078 (2003).
McKay, M.M. & Morrison, D.K. Proteomic analysis of scaffold proteins in the ERK cascade. Methods Mol. Biol. 661, 323–334 (2010).
Acknowledgements
We thank M. Gao from Bristol-Myers Squibb Molecular Structure & Design group for making BRAF expression constructs. N.T. is supported by an Ontario Graduate Scholarship and a Canadian Institutes for Health Research (CIHR) Canada Graduate Scholarship. H.L. is supported by a CIHR Banting Postdoctoral Fellowship. F.S. is supported as a Canada Research Chair. This work was supported by a grant from the Ministère de l'Économie, de l'Innovation et de l'Exportation du Québec to A.M. and an Impact Grant from the Canadian Cancer Society Research Institute to M.T. (702319) as well as by operating funds from the CIHR to M.T. (MOP119443) and F.S. (MOP36399).
Author information
Authors and Affiliations
Contributions
N.T., H.L., F.S. and M.T. designed the experiments and wrote the manuscript. N.T. performed AUC experiments and in vitro kinase assays. H.L. performed BRET and co-IP experiments. D.A.C. conducted protein production, crystallization and X-ray data collection. Structural analysis of the BRAF monomer was performed by D.A.C., A.T., N.T. and H.L. A.M. provided expert advice for the interpretation of compound binding mode.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 Sulfonamide inhibitors occupy the RAF-selective pocket and retain dimer-breaker ability at nonsaturating concentrations in vitro.
(a) Structure.sdf files of sulfonamide compounds were obtained from the PDB and displayed using ChemBioDraw 12.0. (b) Comparison of the two protomers in crystal structures of BRAF bound to PLX4720 (PDB ID 3C4C15) or PLX4032 (PDB ID 3OG716). Protomers in a helix “αC-out”, off-like state conformation (blue) have the “RAF-selective” pocket occupied. For the protomer displaying a helix “αC-in”, on state conformation (orange), the “RAF-selective” pocket is not occupied due to an alternate binding mode of the inhibitor arising from the indicated rotation of the terminal sulfonamide motif. (c) Sedimentation velocity AUC analysis of a solution of the BRAF kinase domain (25 μM of protein) in a titration series with saturating (40 μM) and non-saturating (2.5 μM and 10 μM) of the sulfonamide inhibitors PLX4720, PLX4032 or B0R.
Supplementary Figure 2 The monomer off-state structure of BRAF closely resembles the forced dimer sulfonamide co-structures.
(a) Hydrophobic spines representation of BRAF in the dimer on state, monomer off state and forced dimer intermediate-state (Top). Hydrophobic spines representation of CDK2 in on and off states (Bottom). (b) Binding mode of PLX4720 in the BRAF monomer structure (PDB ID 4WO5; this study) and forced dimer structure (PDB ID 3C4C15). (c) Dimer interface residues of the 24 on state dimers (orange) and 10 off-like state forced dimers (green).
Supplementary Figure 3 Crystal contacts in the BRAF monomeric structure in complex with PLX4720.
Protomer 1 and 2 and their symmetry related mates are shown in light and dark blue, respectively. The side-to-side dimer configuration of on state structure is shown in orange and yellow for reference.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–3 and Supplementary Table 1 (PDF 5534 kb)
Supplementary Data Set 1
Uncropped western blots and autoradiograms presented in the main figures (PDF 442 kb)
Rights and permissions
About this article
Cite this article
Thevakumaran, N., Lavoie, H., Critton, D. et al. Crystal structure of a BRAF kinase domain monomer explains basis for allosteric regulation. Nat Struct Mol Biol 22, 37–43 (2015). https://doi.org/10.1038/nsmb.2924
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb.2924
This article is cited by
-
Analysis of RAS and drug induced homo- and heterodimerization of RAF and KSR1 proteins in living cells using split Nanoluc luciferase
Cell Communication and Signaling (2023)
-
Live-cell target engagement of allosteric MEKi on MEK–RAF/KSR–14-3-3 complexes
Nature Chemical Biology (2023)
-
Pan-cancer clinical impact of latent drivers from double mutations
Communications Biology (2023)
-
Structural insights into the BRAF monomer-to-dimer transition mediated by RAS binding
Nature Communications (2022)
-
Inhibitors of BRAF dimers using an allosteric site
Nature Communications (2020)