Abstract
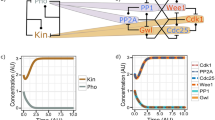
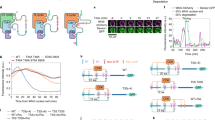
Cks is an evolutionarily conserved protein that regulates cyclin-dependent kinase (CDK) activity. Clarifying the underlying mechanisms and cellular contexts of Cks function is critical because Cks is essential for proper cell growth, and its overexpression has been linked to cancer. We observe that budding-yeast Cks associates with select phosphorylated sequences in cell cycle–regulatory proteins. We characterize the molecular interactions responsible for this specificity and demonstrate that Cks enhances CDK activity in response to specific priming phosphosites. Identification of the binding consensus sequence allows us to identify putative Cks-directed CDK substrates and binding partners. We characterize new Cks-binding sites in the mitotic regulator Wee1 and discover a new role for Cks in regulating CDK activity at mitotic entry. Together, our results portray Cks as a multifunctional phosphoadaptor that serves as a specificity factor for CDK activity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Ubersax, J.A. & Ferrell, J.E. Jr. Mechanisms of specificity in protein phosphorylation. Nat. Rev. Mol. Cell Biol. 8, 530–541 (2007).
Brognard, J. & Hunter, T. Protein kinase signaling networks in cancer. Curr. Opin. Genet. Dev. 21, 4–11 (2011).
Morgan, D.O. The Cell Cycle: Principles of Control (New Science Press, 2007).
Ubersax, J.A. et al. Targets of the cyclin-dependent kinase Cdk1. Nature 425, 859–864 (2003).
Holt, L.J. et al. Global analysis of Cdk1 substrate phosphorylation sites provides insights into evolution. Science 325, 1682–1686 (2009).
Sherr, C.J. Cancer cell cycles. Science 274, 1672–1677 (1996).
Loog, M. & Morgan, D.O. Cyclin specificity in the phosphorylation of cyclin-dependent kinase substrates. Nature 434, 104–108 (2005).
Schulman, B.A., Lindstrom, D.L. & Harlow, E. Substrate recruitment to cyclin-dependent kinase 2 by a multipurpose docking site on cyclin A. Proc. Natl. Acad. Sci. USA 95, 10453–10458 (1998).
Kõivomägi, M. et al. Dynamics of Cdk1 substrate specificity during the cell cycle. Mol. Cell 42, 610–623 (2011).
Koivomagi, M. et al. Multisite phosphorylation networks as signal processors for Cdk1. Nat. Struct. Mol. Biol. 20, 10.1038/nsmb.2706 (3 November 2013).
Hayles, J., Aves, S. & Nurse, P. suc1 is an essential gene involved in both the cell cycle and growth in fission yeast. EMBO J. 5, 3373–3379 (1986).
Tang, Y. & Reed, S.I. The Cdk-associated protein Cks1 functions both in G1 and G2 in Saccharomyces cerevisiae. Genes Dev. 7, 822–832 (1993).
Pines, J. Cell cycle: reaching for a role for the Cks proteins. Curr. Biol. 6, 1399–1402 (1996).
Shapira, M. et al. Alterations in the expression of the cell cycle regulatory protein cyclin kinase subunit 1 in colorectal carcinoma. Cancer 100, 1615–1621 (2004).
Martinsson-Ahlzén, H.S. et al. Cyclin-dependent kinase-associated proteins Cks1 and Cks2 are essential during early embryogenesis and for cell cycle progression in somatic cells. Mol. Cell Biol. 28, 5698–5709 (2008).
Lan, Y. et al. Aberrant expression of Cks1 and Cks2 contributes to prostate tumorigenesis by promoting proliferation and inhibiting programmed cell death. Int. J. Cancer 123, 543–551 (2008).
Westbrook, L. et al. High Cks1 expression in transgenic and carcinogen-initiated mammary tumors is not always accompanied by reduction in p27Kip1. Int. J. Oncol. 34, 1425–1431 (2009).
Ganoth, D. et al. The cell-cycle regulatory protein Cks1 is required for SCFSkp2-mediated ubiquitinylation of p27. Nat. Cell Biol. 3, 321–324 (2001).
Spruck, C. et al. A CDK-independent function of mammalian Cks1: targeting of SCFSkp2 to the CDK inhibitor p27Kip1. Mol. Cell 7, 639–650 (2001).
Morris, M.C. et al. Cks1-dependent proteasome recruitment and activation of CDC20 transcription in budding yeast. Nature 423, 1009–1013 (2003).
Yu, V.P., Baskerville, C., Grunenfelder, B. & Reed, S.I. A kinase-independent function of Cks1 and Cdk1 in regulation of transcription. Mol. Cell 17, 145–151 (2005).
Nash, P. et al. Multisite phosphorylation of a CDK inhibitor sets a threshold for the onset of DNA replication. Nature 414, 514–521 (2001).
Harvey, S.L., Charlet, A., Haas, W., Gygi, S.P. & Kellogg, D.R. Cdk1-dependent regulation of the mitotic inhibitor Wee1. Cell 122, 407–420 (2005).
Kim, S.Y. & Ferrell, J.E. Jr. Substrate competition as a source of ultrasensitivity in the inactivation of Wee1. Cell 128, 1133–1145 (2007).
Kõivomägi, M. et al. Cascades of multisite phosphorylation control Sic1 destruction at the onset of S phase. Nature 480, 128–131 (2011).
Trunnell, N.B., Poon, A.C., Kim, S.Y. & Ferrell, J.E. Jr. Ultrasensitivity in the Regulation of Cdc25C by Cdk1. Mol. Cell 41, 263–274 (2011).
Burke, J.R., Hura, G.L. & Rubin, S.M. Structures of inactive retinoblastoma protein reveal multiple mechanisms for cell cycle control. Genes Dev. 26, 1156–1166 (2012).
Goldbeter, A. & Koshland, D.E. Jr. An amplified sensitivity arising from covalent modification in biological systems. Proc. Natl. Acad. Sci. USA 78, 6840–6844 (1981).
Ferrell, J.E. Jr. Tripping the switch fantastic: how a protein kinase cascade can convert graded inputs into switch-like outputs. Trends Biochem. Sci. 21, 460–466 (1996).
Salazar, C. & Hofer, T. Multisite protein phosphorylation: from molecular mechanisms to kinetic models. FEBS J. 276, 3177–3198 (2009).
Thomson, M. & Gunawardena, J. Unlimited multistability in multisite phosphorylation systems. Nature 460, 274–277 (2009).
Deibler, R.W. & Kirschner, M.W. Quantitative reconstitution of mitotic CDK1 activation in somatic cell extracts. Mol. Cell 37, 753–767 (2010).
Harvey, S.L. et al. A phosphatase threshold sets the level of Cdk1 activity in early mitosis in budding yeast. Mol. Biol. Cell 22, 3595–3608 (2011).
Arvai, A.S., Bourne, Y., Hickey, M.J. & Tainer, J.A. Crystal structure of the human cell cycle protein CksHs1: single domain fold with similarity to kinase N-lobe domain. J. Mol. Biol. 249, 835–842 (1995).
Bourne, Y. et al. Crystal structure of the cell cycle-regulatory protein suc1 reveals a β-hinge conformational switch. Proc. Natl. Acad. Sci. USA 92, 10232–10236 (1995).
Bourne, Y. et al. Crystal structure and mutational analysis of the human CDK2 kinase complex with cell cycle-regulatory protein CksHs1. Cell 84, 863–874 (1996).
Hao, B. et al. Structural basis of the Cks1-dependent recognition of p27Kip1 by the SCFSkp2 ubiquitin ligase. Mol. Cell 20, 9–19 (2005).
Patra, D., Wang, S.X., Kumagai, A. & Dunphy, W.G. The Xenopus Suc1/Cks protein promotes the phosphorylation of G2/M regulators. J. Biol. Chem. 274, 36839–36842 (1999).
Mimura, S., Seki, T., Tanaka, S. & Diffley, J.F. Phosphorylation-dependent binding of mitotic cyclins to Cdc6 contributes to DNA replication control. Nature 431, 1118–1123 (2004).
Odaert, B. et al. Solution NMR study of the monomeric form of p13suc1 protein sheds light on the hinge region determining the affinity for a phosphorylated substrate. J. Biol. Chem. 277, 12375–12381 (2002).
Crowley, P.B. & Golovin, A. Cation-π interactions in protein-protein interfaces. Proteins 59, 231–239 (2005).
Tyanova, S., Cox, J., Olsen, J., Mann, M. & Frishman, D. Phosphorylation variation during the cell cycle scales with structural propensities of proteins. PLOS Comput. Biol. 9, e1002842 (2013).
Ishida, T. & Kinoshita, K. PrDOS: prediction of disordered protein regions from amino acid sequence. Nucleic Acids Res. 35, W460–W464 (2007).
Mueller, P.R., Coleman, T.R. & Dunphy, W.G. Cell cycle regulation of a Xenopus Wee1-like kinase. Mol. Biol. Cell 6, 119–134 (1995).
Tang, Z., Coleman, T.R. & Dunphy, W.G. Two distinct mechanisms for negative regulation of the Wee1 protein kinase. EMBO J. 12, 3427–3436 (1993).
Mukhopadhyay, N.K. et al. An array of insulin-activated, proline-directed serine/threonine protein kinases phosphorylate the p70 S6 kinase. J. Biol. Chem. 267, 3325–3335 (1992).
Burke, J.R., Deshong, A.J., Pelton, J.G. & Rubin, S.M. Phosphorylation-induced conformational changes in the retinoblastoma protein inhibit E2F transactivation domain binding. J. Biol. Chem. 285, 16286–16293 (2010).
Reynard, G.J., Reynolds, W., Verma, R. & Deshaies, R.J. Cks1 is required for G1 cyclin-cyclin-dependent kinase activity in budding yeast. Mol. Cell Biol. 20, 5858–5864 (2000).
Leslie, A.G. The integration of macromolecular diffraction data. Acta Crystallogr. D Biol. Crystallogr. 62, 48–57 (2006).
Howell, P.L. & Smith, G.D. Identification of heavy-atom derivatives by normal probability methods. J. Appl. Cryst. 25, 81–86 (1992).
McCoy, A.J. et al. Phaser crystallographic software. J. Appl. Crystallogr. 40, 658–674 (2007).
Emsley, P. & Cowtan, K. Coot: model-building tools for molecular graphics. Acta Crystallogr. D Biol. Crystallogr. 60, 2126–2132 (2004).
Adams, P.D. et al. PHENIX: a comprehensive Python-based system for macromolecular structure solution. Acta Crystallogr. D Biol. Crystallogr. 66, 213–221 (2010).
Kinoshita, E., Kinoshita-Kikuta, E., Takiyama, K. & Koike, T. Phosphate-binding tag, a new tool to visualize phosphorylated proteins. Mol. Cell Proteomics 5, 749–757 (2006).
Acknowledgements
The authors acknowledge R. Cook of the Massachusetts Institute of Technology Biopolymers group for synthesis of peptide arrays and E. van Veen for providing valuable advice regarding peptide arrays. The authors thank E. Chen for assistance with CD experimental design and analysis. This research was supported by funding to S.M.R. from the American Cancer Society (RSG-12-131-01-CCG) and by a targeted financing scheme and an institutional grant IUT2-21 from the Estonian government to M.L.
Author information
Authors and Affiliations
Contributions
D.A.M., E.R.M.B., M.K., R.L., D.R.K., M.L. and S.M.R. designed the study. D.A.M., E.R.M.B., M.K., R.L., M.V.M. and A.H. performed experiments. All authors analyzed data. D.A.M., E.R.M.B. and S.M.R. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 2 Cks1 alkylation control.
Alkylation of Cks1 does not abrogate binding of phosphopeptides.The wild-type phosCdc62-9 peptide was probed with Cks1 and alkylated Cks1 as described for Figure 2a. Alkylation, which was confirmed by mass spectrometry, occurs on a single cysteine that is distal to the phosphate-binding pocket.
Supplementary Figure 3 Solution behavior of fusion crystallization construct.
Domain swapping and binding properties of the Cks1-Cdc6 fusion construct. (a) ITC data demonstrate that the phosCdc63-9-Cks11-112 fusion protein does not bind phosphorylated Cdc6 peptide in trans (left), while the unphosphorylated fusion binds the Cdc6 peptide similar to wild-type Cks1 (right). These measurements demonstrate that the phosCdc6 sequence in the fusion can bind Cks1 in cis at the appropriate site and exclude the added peptide. (b) In the crystal structure, the fusion undergoes domain swapping in which the phosCdc6 sequence at the C-terminus of one fusion molecule extends to bind the cationic pocket site on a different Cks1 molecule, related by crystallographic symmetry. Four molecules (cyan) are observed in the asymmetric unit. Unlike in several other Cks crystals34,35, there is no evidence that Cks11-112-phosCdc63-9 forms domain-swapped dimers by exchanging strand 4, although the electron density for the loop preceding strand 4 is weak. Instead, dimers are formed between a molecule in the asymmetric unit (cyan) and its crystallographic symmetry mate (yellow) by the swapping of appended phosCdc63-9 sequences. (c) Superdex 75 gel filtration analysis demonstrates that the phosphorylated fusion behaves as a monomer in gel filtration. The unphosphorylated fusion (maroon line) elutes from gel filtration as a double peak, consistent with a monomer-dimer equilibrium in solution and suggesting that the construct undergoes domain swapping similar to Cks1. The phosphorylated Cks11-112-phosCdc63-9 (blue dashes) elutes as a single peak and in the same volume as the monomer-promoting mutant Cks11-112/P93A-Cdc63-9 (green dots)55. We conclude that Cks11-112-phosCdc63-9 fusion protein is a monomer in solution. It is likely that the phosphorylated Cdc6 sequence binds to Cks1 in cis, which disrupts the domain-swapped dimerization that occurs through strand 4 in the wild-type protein in vitro at high concentrations.
Supplementary Figure 4 Cks-mutant structure-function controls.
Mutations to Cks that disrupt consensus sequence binding do not disrupt folding or Cdk binding. (a) Circular dichroism spectra of wild-type and mutant Cks1 proteins at a concentration of ~10 μM were obtained as previously described55. The spectra demonstrate that the mutations do not disrupt proper folding of Cks1. (b) Binding of Cks1 consensus site mutants to recombinant human Cdk2. The Cks-Cdk interface is highly conserved, and budding yeast Cks1 binds Cdk2 with micromolar affinity55,56. We used Cdk2 to facilitate the in vitro binding experiment with recombinant proteins. We mixed 12.5 μM purified His6-Cdk2 with 12.5 μM of the indicated GST-tagged Cks1 protein in a buffer containing 150 mM NaCl, 25mM Tris, and 1 mM DTT (pH 8). The 1 mL binding reaction was mixed with 100 μL GS4B-Sepharose beads and the precipitated beads were washed with binding buffer and eluted with SDS-PAGE sample loading buffer. Input (I), flow-through unbound (U), and bound (B) protein fractions were run on SDS-PAGE and stained with Coomassie Blue. Wild-type and consensus site mutants all bind Cdk2 similarly, which is consistent with the structural observation that the phosphopeptide and Cdk binding interfaces are on opposite faces of Cks (Figure 3a).
Supplementary Figure 5 Kinetic priming assay controls.
Comparison in Cdk kinase reaction of unprimed to primed substrate. Kinase reactions are carried out as described in Figure 4 with wild-type Cks. Both the H1 peptide and Sic1ΔC-T5 construct contain single phosphoacceptor sites. In the Sic1ΔC-T5 construct, all the phosphoacceptor sites except T5 are mutated. The Cdc6 peptide contains phosphorylated T7 and a phosphoacceptor site at T23. The kcat for the reaction with the H1 peptide (marked with *) was previously determined9. Absolute kcat values reported here and in Figure 4 were calculated by comparison of the Vmax value in each experiment to the Vmax determined here for the H1 peptide.
Supplementary Figure 6 Cks1-hsCks1 structure comparison.
Comparison of the phosCdc6-Cks1 structure determined here with the structure of phosp27-hsCks1-Skp2 (PDB code: 2AST)37.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–7 and Supplementary Table 1 (PDF 8203 kb)
Supplementary Table 2
Putative Cks-binding sequences (XLSX 32 kb)
Rights and permissions
About this article
Cite this article
McGrath, D., Balog, E., Kõivomägi, M. et al. Cks confers specificity to phosphorylation-dependent CDK signaling pathways. Nat Struct Mol Biol 20, 1407–1414 (2013). https://doi.org/10.1038/nsmb.2707
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb.2707
This article is cited by
-
Phosphosite Scanning reveals a complex phosphorylation code underlying CDK-dependent activation of Hcm1
Nature Communications (2023)
-
Structural basis of human separase regulation by securin and CDK1–cyclin B1
Nature (2021)
-
A processive phosphorylation circuit with multiple kinase inputs and mutually diversional routes controls G1/S decision
Nature Communications (2020)
-
Multisite phosphorylation code of CDK
Nature Structural & Molecular Biology (2019)
-
An allosteric conduit facilitates dynamic multisite substrate recognition by the SCFCdc4 ubiquitin ligase
Nature Communications (2017)