Abstract
Cell identity is determined by specific gene expression patterns that are conveyed by interactions between transcription factors and DNA in the context of chromatin. In development, epigenetic modifiers are thought to stabilize gene expression and ensure that patterns of DNA methylation and histone modification are reinstated in cells as they divide. Global erasure of epigenetic marks occurs naturally at two stages in the mammalian life cycle, but it can also be artificially engineered using a variety of reprogramming strategies. Here we review some of the recent advances in understanding how epigenetic remodeling contributes to conversion of cell fate in vivo and in vitro. We summarize current models of epigenetic erasure and discuss the various enzymes and mechanisms that may operate in cellular reprogramming.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
ENCODE Project Consortium et al. An integrated encyclopedia of DNA elements in the human genome. Nature 489, 57–74 (2012).
Thurman, R.E. et al. The accessible chromatin landscape of the human genome. Nature 489, 75–82 (2012).
Lengsfeld, B.M., Berry, K.N., Ghosh, S., Takahashi, M. & Francis, N.J. A Polycomb complex remains bound through DNA replication in the absence of other eukaryotic proteins. Sci. Rep. 2, 661 (2012).
Lo, S.M. et al. A bridging model for persistence of a polycomb group protein complex through DNA replication in vitro. Mol. Cell 46, 784–796 (2012).
Petruk, S. et al. TrxG and PcG proteins but not methylated histones remain associated with DNA through replication. Cell 150, 922–933 (2012).
Fisher, A.G. & Brockdorff, N. Epigenetic memory and parliamentary privilege combine to evoke discussions on inheritance. Development 139, 3891–3896 (2012).
Bonasio, R., Tu, S. & Reinberg, D. Molecular signals of epigenetic states. Science 330, 612–616 (2010).
Braun, R.E. Packaging paternal chromosomes with protamine. Nat. Genet. 28, 10–12 (2001).
Adenot, P.G., Mercier, Y., Renard, J.P. & Thompson, E.M. Differential H4 acetylation of paternal and maternal chromatin precedes DNA replication and differential transcriptional activity in pronuclei of 1-cell mouse embryos. Development 124, 4615–4625 (1997).
Santos, F., Peters, A.H., Otte, A.P., Reik, W. & Dean, W. Dynamic chromatin modifications characterise the first cell cycle in mouse embryos. Dev. Biol. 280, 225–236 (2005).
Sobel, R.E., Cook, R.G., Perry, C.A., Annunziato, A.T. & Allis, C.D. Conservation of deposition-related acetylation sites in newly synthesized histones H3 and H4. Proc. Natl. Acad. Sci. USA 92, 1237–1241 (1995).
Erhardt, S. et al. Consequences of the depletion of zygotic and embryonic enhancer of zeste 2 during preimplantation mouse development. Development 130, 4235–4248 (2003).
Liu, H., Kim, J.M. & Aoki, F. Regulation of histone H3 lysine 9 methylation in oocytes and early pre-implantation embryos. Development 131, 2269–2280 (2004).
Arney, K.L., Bao, S., Bannister, A.J., Kouzarides, T. & Surani, M.A. Histone methylation defines epigenetic asymmetry in the mouse zygote. Int. J. Dev. Biol. 46, 317–320 (2002).
Puschendorf, M. et al. PRC1 and Suv39h specify parental asymmetry at constitutive heterochromatin in early mouse embryos. Nat. Genet. 40, 411–420 (2008).
Kourmouli, N. et al. Heterochromatin and tri-methylated lysine 20 of histone H4 in animals. J. Cell Sci. 117, 2491–2501 (2004).
Daujat, S. et al. H3K64 trimethylation marks heterochromatin and is dynamically remodeled during developmental reprogramming. Nat. Struct. Mol. Biol. 16, 777–781 (2009).
Sarmento, O.F. et al. Dynamic alterations of specific histone modifications during early murine development. J. Cell Sci. 117, 4449–4459 (2004).
Lepikhov, K. & Walter, J. Differential dynamics of histone H3 methylation at positions K4 and K9 in the mouse zygote. BMC Dev. Biol. 4, 12 (2004).
Mayer, W., Niveleau, A., Walter, J., Fundele, R. & Haaf, T. Embryogenesis: demethylation of the zygotic paternal genome. Nature 403, 501–502 (2000).
Santos, F., Hendrich, B., Reik, W. & Dean, W. Dynamic reprogramming of DNA methylation in the early mouse embryo. Dev. Biol. 241, 172–182 (2002).
Inoue, A. & Zhang, Y. Replication-dependent loss of 5-hydroxymethylcytosine in mouse preimplantation embryos. Science 334, 194 (2011).
Smith, Z.D. et al. A unique regulatory phase of DNA methylation in the early mammalian embryo. Nature 484, 339–344 (2012).This is the first base-resolution, genome-wide analysis of DNA methylation during preimplantation development.
Gill, M.E., Erkek, S. & Peters, A.H.F.M. Parental epigenetic control of embryogenesis: a balance between inheritance and reprogramming? Curr. Opin. Cell Biol. 24, 387–396 (2012).
Bouniol, C., Nguyen, E. & Debey, P. Endogenous transcription occurs at the 1-cell stage in the mouse embryo. Exp. Cell Res. 218, 57–62 (1995).
Aoki, F., Worrad, D.M. & Schultz, R.M. Regulation of transcriptional activity during the first and second cell cycles in the preimplantation mouse embryo. Dev. Biol. 181, 296–307 (1997).
Henery, C.C., Miranda, M., Wiekowski, M., Wilmut, I. & DePamphilis, M.L. Repression of gene expression at the beginning of mouse development. Dev. Biol. 169, 448–460 (1995).
Latham, K.E. & Schultz, R.M. Embryonic genome activation. Front. Biosci. 6, D748–D759 (2001).
Gu, T.P. et al. The role of Tet3 DNA dioxygenase in epigenetic reprogramming by oocytes. Nature 477, 606–610 (2011).Work in refs. 29 and 36 demonstrates that the global replication-independent loss of 5mC in the paternal genome of the zygote is due to its oxidation into 5hmC by Tet3 enzyme.
Oswald, J. et al. Active demethylation of the paternal genome in the mouse zygote. Curr. Biol. 10, 475–478 (2000).
Wossidlo, M. et al. Dynamic link of DNA demethylation, DNA strand breaks and repair in mouse zygotes. EMBO J. 29, 1877–1888 (2010).
Tahiliani, M. et al. Conversion of 5-methylcytosine to 5-hydroxymethylcytosine in mammalian DNA by MLL partner TET1. Science 324, 930–935 (2009).This work identifies TET proteins as mammalian homologs of trypanosome enzymes that are capable of oxidizing 5mC into 5hmC.
He, Y.F. et al. Tet-mediated formation of 5-carboxylcytosine and its excision by TDG in mammalian DNA. Science 333, 1303–1307 (2011).
Ito, S. et al. Tet proteins can convert 5-methylcytosine to 5-formylcytosine and 5-carboxylcytosine. Science 333, 1300–1303 (2011).
Iqbal, K., Jin, S.G., Pfeifer, G.P. & Szabo, P.E. Reprogramming of the paternal genome upon fertilization involves genome-wide oxidation of 5-methylcytosine. Proc. Natl. Acad. Sci. USA 108, 3642–3647 (2011).
Wossidlo, M. et al. 5-Hydroxymethylcytosine in the mammalian zygote is linked with epigenetic reprogramming. Nat. Commun. 2, 241 (2011).
Inoue, A., Shen, L., Dai, Q., He, C. & Zhang, Y. Generation and replication-dependent dilution of 5fC and 5caC during mouse preimplantation development. Cell Res. 21, 1670–1676 (2011).
Teperek-Tkacz, M., Pasque, V., Gentsch, G. & Ferguson-Smith, A.C. Epigenetic reprogramming: is deamination key to active DNA demethylation? Reproduction 142, 621–632 (2011).
Hajkova, P. et al. Genome-wide reprogramming in the mouse germ line entails the base excision repair pathway. Science 329, 78–82 (2010).
Chan, M.M., Smith, Z.D., Egli, D., Regev, A. & Meissner, A. Mouse ooplasm confers context-specific reprogramming capacity. Nat. Genet. 44, 978–980 (2012).
Booth, M.J. et al. Quantitative sequencing of 5-methylcytosine and 5-hydroxymethylcytosine at single-base resolution. Science 336, 934–937 (2012).
Yu, M. et al. Base-resolution analysis of 5-hydroxymethylcytosine in the mammalian genome. Cell 149, 1368–1380 (2012).
Branco, M.R., Oda, M. & Reik, W. Safeguarding parental identity: Dnmt1 maintains imprints during epigenetic reprogramming in early embryogenesis. Genes Dev. 22, 1567–1571 (2008).
Hirasawa, R. et al. Maternal and zygotic Dnmt1 are necessary and sufficient for the maintenance of DNA methylation imprints during preimplantation development. Genes Dev. 22, 1607–1616 (2008).
Payer, B. et al. Stella is a maternal effect gene required for normal early development in mice. Curr. Biol. 13, 2110–2117 (2003).
Nakamura, T. et al. PGC7/Stella protects against DNA demethylation in early embryogenesis. Nat. Cell Biol. 9, 64–71 (2007).
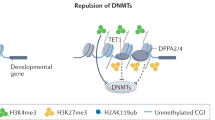
Nakamura, T. et al. PGC7 binds histone H3K9me2 to protect against conversion of 5mC to 5hmC in early embryos. Nature 486, 415–419 (2012).This work shows that Dppa3 protects the maternal genome and genomic imprints from Tet3-mediate oxidation by binding H3K9me2.
Lane, N. et al. Resistance of IAPs to methylation reprogramming may provide a mechanism for epigenetic inheritance in the mouse. Genesis 35, 88–93 (2003).
Olek, A. & Walter, J. The pre-implantation ontogeny of the H19 methylation imprint. Nat. Genet. 17, 275–276 (1997).
Tremblay, K.D., Duran, K.L. & Bartolomei, M.S. A 5′2-kilobase-pair region of the imprinted mouse H19 gene exhibits exclusive paternal methylation throughout development. Mol. Cell Biol. 17, 4322–4329 (1997).
Pittoggi, C. et al. A fraction of mouse sperm chromatin is organized in nucleosomal hypersensitive domains enriched in retroposon DNA. J. Cell Sci. 112, 3537–3548 (1999).
Wykes, S.M. & Krawetz, S.A. The structural organization of sperm chromatin. J. Biol. Chem. 278, 29471–29477 (2003).
van der Heijden, G.W. et al. Sperm-derived histones contribute to zygotic chromatin in humans. BMC Dev. Biol. 8, 34 (2008).
Li, X. et al. A maternal-zygotic effect gene, Zfp57, maintains both maternal and paternal imprints. Dev. Cell 15, 547–557 (2008).Work described in refs. 54–57 demonstrates that sequence-specific DNA-binding factors Zfp57 and Trim28 can cooperate with the histone modification system to maintain DNA methylation marks at imprinted loci, thus preventing their loss during preimplantation development.
Messerschmidt, D.M. et al. Trim28 is required for epigenetic stability during mouse oocyte to embryo transition. Science 335, 1499–1502 (2012).
Quenneville, S. et al. The KRAB-ZFP/KAP1 system contributes to the early embryonic establishment of site-specific DNA methylation patterns maintained during development. Cell Rep. 2, 766–773 (2012).
Quenneville, S. et al. In embryonic stem cells, ZFP57/KAP1 recognize a methylated hexanucleotide to affect chromatin and DNA methylation of imprinting control regions. Mol. Cell 44, 361–372 (2011).
Zuo, X. et al. Zinc finger protein ZFP57 requires its co-factor to recruit DNA methyltransferases and maintains DNA methylation imprint in embryonic stem cells via its transcriptional repression domain. J. Biol. Chem. 287, 2107–2118 (2012).
Schultz, D.C., Ayyanathan, K., Negorev, D., Maul, G.G. & Rauscher, F.J. SETDB1: a novel KAP-1-associated histone H3, lysine 9-specific methyltransferase that contributes to HP1-mediated silencing of euchromatic genes by KRAB zinc-finger proteins. Genes Dev. 16, 919–932 (2002).
Lorthongpanich, C., Doris, T.P., Limviphuvadh, V., Knowles, B.B. & Solter, D. Developmental fate and lineage commitment of singled mouse blastomeres. Development 139, 3722–3731 (2012).
Takaoka, K. & Hamada, H. Cell fate decisions and axis determination in the early mouse embryo. Development 139, 3–14 (2012).
Guo, J.U., Su, Y., Zhong, C., Ming, G.L. & Song, H. Hydroxylation of 5-methylcytosine by TET1 promotes active DNA demethylation in the adult brain. Cell 145, 423–434 (2011).
Johnson, M.H. & Ziomek, C.A. The foundation of two distinct cell lineages within the mouse morula. Cell 24, 71–80 (1981).
Torres-Padilla, M.E., Parfitt, D.E., Kouzarides, T. & Zernicka-Goetz, M. Histone arginine methylation regulates pluripotency in the early mouse embryo. Nature 445, 214–218 (2007).This work shows that increasing the levels of arginine methylation in individual blastomeres from 4-cell-stage embryos results in the dramatic upregulation of Oct4 and Sox2 and directs their progeny to the inner cell mass.
Fisher, C.L. & Fisher, A.G. Chromatin states in pluripotent, differentiated, and reprogrammed cells. Curr. Opin. Genet. Dev. 21, 140–146 (2011).
Azuara, V. et al. Chromatin signatures of pluripotent cell lines. Nat. Cell Biol. 8, 532–538 (2006).
Bernstein, B.E. et al. A bivalent chromatin structure marks key developmental genes in embryonic stem cells. Cell 125, 315–326 (2006).
Voigt, P. et al. Asymmetrically modified nucleosomes. Cell 151, 181–193 (2012).
Margueron, R. & Reinberg, D. The Polycomb complex PRC2 and its mark in life. Nature 469, 343–349 (2011).
Ballaré, C. et al. Phf19 links methylated Lys36 of histone H3 to regulation of Polycomb activity. Nat. Struct. Mol. Biol. 19, 1257–1265 (2012).Work described in refs. 70 and 71 demonstrates that the PRC2 component Phf19 binds H3K36me2 and H3K36me3 and is required both for the deposition of H3K27me3 and the recruitment of H3K36 demethylase enzymes at Polycomb target genes.
Brien, G.L. et al. Polycomb PHF19 binds H3K36me3 and recruits PRC2 and demethylase NO66 to embryonic stem cell genes during differentiation. Nat. Struct. Mol. Biol. 19, 1273–1281 (2012).
Blackledge, N.P. et al. CpG islands recruit a histone H3 lysine 36 demethylase. Mol. Cell 38, 179–190 (2010).
Ku, M. et al. Genomewide analysis of PRC1 and PRC2 occupancy identifies two classes of bivalent domains. PLoS Genet. 4, e1000242 (2008).
Ohinata, Y. et al. A signaling principle for the specification of the germ cell lineage in mice. Cell 137, 571–584 (2009).
Hajkova, P. et al. Chromatin dynamics during epigenetic reprogramming in the mouse germ line. Nature 452, 877–881 (2008).
Seki, Y. et al. Extensive and orderly reprogramming of genome-wide chromatin modifications associated with specification and early development of germ cells in mice. Dev. Biol. 278, 440–458 (2005).
Mansour, A.A. et al. The H3K27 demethylase Utx regulates somatic and germ cell epigenetic reprogramming. Nature 488, 409–413 (2012).
Tachibana, M. et al. Histone methyltransferases G9a and GLP form heteromeric complexes and are both crucial for methylation of euchromatin at H3–K9. Genes Dev. 19, 815–826 (2005).
Dong, K.B. et al. DNA methylation in ES cells requires the lysine methyltransferase G9a but not its catalytic activity. EMBO J. 27, 2691–2701 (2008).
Tachibana, M., Matsumura, Y., Fukuda, M., Kimura, H. & Shinkai, Y. G9a/GLP complexes independently mediate H3K9 and DNA methylation to silence transcription. EMBO J. 27, 2681–2690 (2008).
Kurimoto, K. et al. Complex genome-wide transcription dynamics orchestrated by Blimp1 for the specification of the germ cell lineage in mice. Genes Dev. 22, 1617–1635 (2008).
Yamaji, M. et al. Critical function of Prdm14 for the establishment of the germ cell lineage in mice. Nat. Genet. 40, 1016–1022 (2008).
Lee, J. et al. Erasing genomic imprinting memory in mouse clone embryos produced from day 11.5 primordial germ cells. Development 129, 1807–1817 (2002).
Sato, S., Yoshimizu, T., Sato, E. & Matsui, Y. Erasure of methylation imprinting of Igf2r during mouse primordial germ-cell development. Mol. Reprod. Dev. 65, 41–50 (2003).
Yamazaki, Y. et al. Adult mice cloned from migrating primordial germ cells. Proc. Natl. Acad. Sci. USA 102, 11361–11366 (2005).
Hackett, J.A. et al. Promoter DNA methylation couples genome-defence mechanisms to epigenetic reprogramming in the mouse germline. Development 139, 3623–3632 (2012).
Guibert, S., Forne, T. & Weber, M. Global profiling of DNA methylation erasure in mouse primordial germ cells. Genome Res. 22, 633–641 (2012).
Popp, C. et al. Genome-wide erasure of DNA methylation in mouse primordial germ cells is affected by AID deficiency. Nature 463, 1101–1105 (2010).
Chuva de Sousa Lopes, S.M. et al. X chromosome activity in mouse XX primordial germ cells. PLoS Genet. 4, e30 (2008).
Yamaguchi, S. et al. Tet1 controls meiosis by regulating meiotic gene expression. Nature 492, 443–447 (2012).This work shows that during PGCs development Tet1 is required for the demethylation and the consequent expression of a specific functional class of genes, which are required for meiosis.
Cortellino, S. et al. Thymine DNA glycosylase is essential for active DNA demethylation by linked deamination-base excision repair. Cell 146, 67–79 (2011).
Hanna, J. et al. Direct cell reprogramming is a stochastic process amenable to acceleration. Nature 462, 595–601 (2009).Work described in refs. 92 and 93 demonstrates that reprogramming of somatic cells via the forced expression of a defined set of transcriptional factors is initially triggered by a series of stochastic (epigenetic) events that subsequently lead to the hierarchical activation of the pluripotency network.
Buganim, Y. et al. Single-cell expression analyses during cellular reprogramming reveal an early stochastic and a late hierarchic phase. Cell 150, 1209–1222 (2012).
Bui, H.T. et al. Effect of trichostatin A on chromatin remodeling, histone modifications, DNA replication, and transcriptional activity in cloned mouse embryos. Biol. Reprod. 83, 454–463 (2010).
Esteban, M.A. et al. Vitamin C enhances the generation of mouse and human induced pluripotent stem cells. Cell Stem Cell 6, 71–79 (2010).
Huangfu, D. et al. Induction of pluripotent stem cells by defined factors is greatly improved by small-molecule compounds. Nat. Biotechnol. 26, 795–797 (2008).
Kishigami, S. et al. Significant improvement of mouse cloning technique by treatment with trichostatin A after somatic nuclear transfer. Biochem. Biophys. Res. Commun. 340, 183–189 (2006).
Liang, G., Taranova, O., Xia, K. & Zhang, Y. Butyrate promotes induced pluripotent stem cell generation. J. Biol. Chem. 285, 25516–25521 (2010).
Mali, P. et al. Butyrate greatly enhances derivation of human induced pluripotent stem cells by promoting epigenetic remodeling and the expression of pluripotency–associated genes. Stem Cells 28, 713–720 (2010).
Mikkelsen, T.S. et al. Dissecting direct reprogramming through integrative genomic analysis. Nature 454, 49–55 (2008).
Sridharan, R. et al. Role of the murine reprogramming factors in the induction of pluripotency. Cell 136, 364–377 (2009).
Ang, Y.S. et al. Wdr5 mediates self-renewal and reprogramming via the embryonic stem cell core transcriptional network. Cell 145, 183–197 (2011).
Shi, Y. et al. Induction of pluripotent stem cells from mouse embryonic fibroblasts by Oct4 and Klf4 with small-molecule compounds. Cell Stem Cell 3, 568–574 (2008).
Shi, Y. et al. A combined chemical and genetic approach for the generation of induced pluripotent stem cells. Cell Stem Cell 2, 525–528 (2008).
Epsztejn-Litman, S. et al. De novo DNA methylation promoted by G9a prevents reprogramming of embryonically silenced genes. Nat. Struct. Mol. Biol. 15, 1176–1183 (2008).
Ma, D.K., Chiang, C.H., Ponnusamy, K., Ming, G.L. & Song, H. G9a and Jhdm2a regulate embryonic stem cell fusion-induced reprogramming of adult neural stem cells. Stem Cells 26, 2131–2141 (2008).
Ho, L. & Crabtree, G.R. Chromatin remodelling during development. Nature 463, 474–484 (2010).
Gaspar-Maia, A. et al. Chd1 regulates open chromatin and pluripotency of embryonic stem cells. Nature 460, 863–868 (2009).
Ho, L. et al. An embryonic stem cell chromatin remodeling complex, esBAF, is an essential component of the core pluripotency transcriptional network. Proc. Natl. Acad. Sci. USA 106, 5187–5191 (2009).
Ho, L. et al. esBAF facilitates pluripotency by conditioning the genome for LIF/STAT3 signalling and by regulating polycomb function. Nat. Cell Biol. 13, 903–913 (2011).
Singhal, N. et al. Chromatin-remodeling components of the BAF complex facilitate reprogramming. Cell 141, 943–955 (2010).
Banaszynski, L.A., Allis, C.D. & Lewis, P.W. Histone variants in metazoan development. Dev. Cell 19, 662–674 (2010).
Pasque, V., Gillich, A., Garrett, N. & Gurdon, J.B. Histone variant macroH2A confers resistance to nuclear reprogramming. EMBO J. 30, 2373–2387 (2011).
Koche, R.P. et al. Reprogramming factor expression initiates widespread targeted chromatin remodeling. Cell Stem Cell 8, 96–105 (2011).
Pereira, C.F. et al. Heterokaryon-based reprogramming of human B lymphocytes for pluripotency requires Oct4 but not Sox2. PLoS Genet. 4, e1000170 (2008).
Bhutani, N. et al. Reprogramming towards pluripotency requires AID-dependent DNA demethylation. Nature 463, 1042–1047 (2010).
Simonsson, S. & Gurdon, J. DNA demethylation is necessary for the epigenetic reprogramming of somatic cell nuclei. Nat. Cell Biol. 6, 984–990 (2004).
Piccolo, F.M. et al. Using heterokaryons to understand pluripotency and reprogramming. Phil. Trans. R. Soc. Lond. B 366, 2260–2265 (2011).
Foshay, K.M. et al. Embryonic stem cells induce pluripotency in somatic cell fusion through biphasic reprogramming. Mol. Cell 46, 159–170 (2012).
Stadtfeld, M., Maherali, N., Breault, D.T. & Hochedlinger, K. Defining molecular cornerstones during fibroblast to iPS cell reprogramming in mouse. Cell Stem Cell 2, 230–240 (2008).
Doege, C.A. et al. Early-stage epigenetic modification during somatic cell reprogramming by Parp1 and Tet2. Nature 488, 652–655 (2012).This work shows that Tet2 is required during early phases of iPSC reprogramming for remodeling the chromatin at the promoters of key pluripotency genes in a demethylation-independent manner.
Kim, K. et al. Epigenetic memory in induced pluripotent stem cells. Nature 467, 285–290 (2010).
Hackett, J.A. et al. Germline DNA demethylation dynamics and imprint erasure through 5-hydroxymethylcytosine. Science advance online publication, doi:10.1126/science.1229277 (13 December 2012).
Takagi, N. Requirement of mitoses for the reversal of X-inactivation in cell hybrids between murine embryonal carcinoma cells and normal female thymocytes. Exp. Cell Res. 175, 363–375 (1988).
Gurdon, J.B. The developmental capacity of nuclei taken from intestinal epithelium cells of feeding tadpoles. J. Embryol. Exp. Morphol. 10, 622–640 (1962).
Wakayama, T., Perry, A.C., Zuccotti, M., Johnson, K.R. & Yanagimachi, R. Full-term development of mice from enucleated oocytes injected with cumulus cell nuclei. Nature 394, 369–374 (1998).
Tada, M., Takahama, Y., Abe, K., Nakatsuji, N. & Tada, T. Nuclear reprogramming of somatic cells by in vitro hybridization with ES cells. Curr. Biol. 11, 1553–1558 (2001).
Takahashi, K. & Yamanaka, S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 126, 663–676 (2006).
Acknowledgements
We thank the Human Frontiers Science Programme (I.C.) and the Medical Research Council UK (A.G.F.) for support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Cantone, I., Fisher, A. Epigenetic programming and reprogramming during development. Nat Struct Mol Biol 20, 282–289 (2013). https://doi.org/10.1038/nsmb.2489
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb.2489
This article is cited by
-
Many Paths to Anticipatory Behavior: Anticipatory Model Acquisition Across Phylogenetic and Ontogenetic Timescales
Biological Theory (2023)
-
Aberrant epigenetic and transcriptional events associated with breast cancer risk
Clinical Epigenetics (2022)
-
Genotoxic and epigenetic effects of diuron in the Pacific oyster: in vitro evidence of interaction between DNA damage and DNA methylation
Environmental Science and Pollution Research (2021)
-
Chemerin reverses the malignant phenotype and induces differentiation of human hepatoma SMMC7721 cells
Archives of Pharmacal Research (2021)
-
What impact does oocyte vitrification have on epigenetics and gene expression?
Clinical Epigenetics (2020)