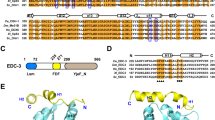
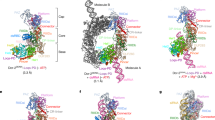
Abstract
The removal of the mRNA 5′ cap structure by the decapping enzyme DCP2 leads to rapid 5′→3′ mRNA degradation by XRN1, suggesting that the two processes are coordinated, but the coupling mechanism is unknown. DCP2 associates with the decapping activators EDC4 and DCP1. Here we show that XRN1 directly interacts with EDC4 and DCP1 in human and Drosophila melanogaster cells, respectively. In D. melanogaster cells, this interaction is mediated by the DCP1 EVH1 domain and a DCP1-binding motif (DBM) in the XRN1 C-terminal region. The NMR structure of the DCP1 EVH1 domain bound to the DBM reveals that the peptide docks at a conserved aromatic cleft, which is used by EVH1 domains to recognize proline-rich ligands. Our findings reveal a role for XRN1 in decapping and provide a molecular basis for the coupling of decapping to 5′→3′ mRNA degradation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Accession codes
References
Chen, C.Y. & Shyu, A.B. Mechanisms of deadenylation-dependent decay. Wiley Interdiscip. Rev. RNA 2, 167–183 (2011).
Houseley, J., LaCava, J. & Tollervey, D. RNA-quality control by the exosome. Nat. Rev. Mol. Cell Biol. 7, 529–539 (2006).
Ling, S.H., Qamra, R. & Song, H. Structural and functional insights into eukaryotic mRNA decapping. Wiley Interdiscip Rev. RNA 2, 193–208 (2011).
Nissan, T., Rajyaguru, P., She, M., Song, H. & Parker, R. Decapping activators in Saccharomyces cerevisiae act by multiple mechanisms. Mol. Cell 39, 773–783 (2010).
Fenger-Grøn, M., Fillman, C., Norrild, B. & Lykke-Andersen, J. Multiple processing body factors and the ARE binding protein TTP activate mRNA decapping. Mol. Cell 20, 905–915 (2005).
Hsu, C.L. & Stevens, A. Yeast cells lacking 5′→3′ exoribonuclease 1 contain mRNA species that are poly(A) deficient and partially lack the 5′ cap structure. Mol. Cell. Biol. 13, 4826–4835 (1993).
Muhlrad, D., Decker, C.J. & Parker, R. Deadenylation of the unstable mRNA encoded by the yeast MFA2 gene leads to decapping followed by 5′→3′ digestion of the transcript. Genes Dev. 8, 855–866 (1994).
Bouveret, E., Rigaut, G., Shevchenko, A., Wilm, M. & Seraphin, B. A Sm-like protein complex that participates in mRNA degradation. EMBO J. 19, 1661–1671 (2000).
Ozgur, S., Chekulaeva, M. & Stoecklin, G. Human Pat1b connects deadenylation with mRNA decapping and controls the assembly of processing bodies. Mol. Cell. Biol. 30, 4308–4323 (2010).
Fromont-Racine, M. et al. Genome-wide protein interaction screens reveal functional networks involving Sm-like proteins. Yeast 17, 95–110 (2000).
Parker, R. & Sheth, U. P bodies and the control of mRNA translation and degradation. Mol. Cell 25, 635–646 (2007).
Ball, L.J., Jarchau, T., Oschkinat, H. & Walter, U. EVH1 domains: structure, function and interactions. FEBS Lett. 513, 45–52 (2002).
Peterson, F.C. & Volkman, B.F. Diversity of polyproline recognition by EVH1 domains. Front. Biosci. 14, 833–846 (2009).
She, M. et al. Crystal structure of Dcp1p and its functional implications in mRNA decapping. Nat. Struct. Mol. Biol. 11, 249–256 (2004).
She, M. et al. Structural basis of Dcp2 recognition and activation by Dcp1. Mol. Cell 29, 337–349 (2008).
Tritschler, F. et al. DCP1 forms asymmetric trimers to assemble into active mRNA decapping complexes in metazoa. Proc. Natl. Acad. Sci. USA 106, 21591–21596 (2009).
Fromm, S.A. et al. The structural basis of Edc3- and Scd6-mediated activation of the Dcp1:Dcp2 mRNA decapping complex. EMBO J. 31, 279–290 (2012).
Jinek, M., Coyle, S.M. & Doudna, J.A. Coupled 5′ nucleotide recognition and processivity in Xrn1-mediated mRNA decay. Mol. Cell 41, 600–608 (2011).
Chang, J.H., Xiang, S., Xiang, K., Manley, J.L. & Tong, L. Structural and biochemical studies of the 5′→3′ exoribonuclease Xrn1. Nat. Struct. Mol. Biol. 18, 270–276 (2011).
Volkman, B.F., Prehoda, K.E., Scott, J.A., Peterson, F.C. & Lim, W.A. Structure of the N-WASP EVH1 domain-WIP complex: insight into the molecular basis of Wiskott-Aldrich Syndrome. Cell 111, 565–576 (2002).
Prehoda, K.E., Lee, D.J. & Lim, W.A. Structure of the enabled/VASP homology 1 domain-peptide complex: a key component in the spatial control of actin assembly. Cell 97, 471–480 (1999).
Fedorov, A.A., Fedorov, E., Gertler, F. & Almo, S.C. Structure of EVH1, a novel proline-rich ligand-binding module involved in cytoskeletal dynamics and neural function. Nat. Struct. Biol. 6, 661–665 (1999).
Carl, U.D. et al. Aromatic and basic residues within the EVH1 domain of VASP specify its interaction with proline-rich ligands. Curr. Biol. 9, 715–718 (1999).
Ball, L.J. et al. Dual epitope recognition by the VASP EVH1 domain modulates polyproline ligand specificity and binding affinity. EMBO J. 19, 4903–4914 (2000).
Beneken, J. et al. Structure of the Homer EVH1 domain-peptide complex reveals a new twist in polyproline recognition. Neuron 26, 143–154 (2000).
Barzik, M. et al. The N-terminal domain of Homer/Vesl is a new class II EVH1 domain. J. Mol. Biol. 309, 155–169 (2001).
Yu, J.H., Yang, W.H., Gulick, T., Bloch, K.D. & Bloch, D.B. Ge-1 is a central component of the mammalian cytoplasmic mRNA processing body. RNA 11, 1795–1802 (2005).
Jinek, M. et al. The C-terminal region of Ge-1 presents conserved structural features required for P-body localization. RNA 14, 1991–1998 (2008).
Bloch, D.B., Nobre, R.A., Bernstein, G.A. & Yang, W.H. Identification and characterization of protein interactions in the mammalian mRNA processing body using a novel two-hybrid assay. Exp. Cell Res. 317, 2183–2199 (2011).
Behm-Ansmant, I. et al. mRNA degradation by miRNAs and GW182 requires both CCR4:NOT deadenylase and DCP1:DCP2 decapping complexes. Genes Dev. 20, 1885–1898 (2006).
Eulalio, A. et al. Target-specific requirements for enhancers of decapping in miRNA-mediated gene silencing. Genes Dev. 21, 2558–2570 (2007).
Chekulaeva, M. et al. miRNA repression involves GW182-mediated recruitment of CCR4-NOT through conserved W-containing motifs. Nat. Struct. Mol. Biol. 18, 1218–1226 (2011).
Xu, J., Yang, J.Y., Niu, Q.W. & Chua, N.H. Arabidopsis DCP2, DCP1, and VARICOSE form a decapping complex required for postembryonic development. Plant Cell 18, 3386–3398 (2006).
Borja, M.S., Piotukh, K., Freund, C. & Gross, J.D. Dcp1 links coactivators of mRNA decapping to Dcp2 by proline recognition. RNA 17, 278–290 (2011).
Davey, N.E. et al. Attributes of short linear motifs. Mol. Biosyst. 8, 268–281 (2012).
Deshmukh, M.V. et al. mRNA decapping is promoted by an RNA-binding channel in Dcp2. Mol. Cell 29, 324–336 (2008).
Floor, S.N., Borja, M.S. & Gross, J.D. Interdomain dynamics and coactivation of the mRNA decapping enzyme Dcp2 are mediated by a gatekeeper tryptophan. Proc. Natl. Acad. Sci. USA 109, 2872–2877 (2012).
Haas, G. et al. HPat provides a link between deadenylation and decapping in metazoa. J. Cell Biol. 189, 289–302 (2010).
Braun, J.E., Huntzinger, E., Fauser, M. & Izaurralde, E. GW182 proteins recruit cytoplasmic deadenylase complexes to miRNA targets. Mol. Cell 44, 120–133 (2011).
Diebold, M.L., Fribourg, S., Koch, M., Metzger, T. & Romier, C. Deciphering correct strategies for multiprotein complex assembly by co-expression: application to complexes as large as the histone octamer. J. Struct. Biol. 175, 178–188 (2011).
Diercks, T., Daniels, M. & Kaptein, R. Extended flip-back schemes for sensitivity enhancement in multidimensional HSQC-type out-and-back experiments. J. Biomol. NMR 33, 243–259 (2005).
Carlomagno, T. et al. PLUSH TACSY: homonuclear planar TACSY with two-band selective shaped pulses applied to C(α),C′ transfer and C (β),C (aromatic) correlations. J. Biomol. NMR 8, 161–170 (1996).
Diercks, T., Coles, M. & Kessler, H. An efficient strategy for assignment of cross-peaks in 3D heteronuclear NOESY experiments. J. Biomol. NMR 15, 177–180 (1999).
Cornilescu, G., Delaglio, F. & Bax, A. Protein backbone angle restraints from searching a database for chemical shift and sequence homology. J. Biomol. NMR 13, 289–302 (1999).
Ginzinger, S.W. & Coles, M. SimShiftDB; local conformational restraints derived from chemical shift similarity searches on a large synthetic database. J. Biomol. NMR 43, 179–185 (2009).
Truffault, V. et al. The solution structure of the N-terminal domain of riboflavin synthase. J. Mol. Biol. 309, 949–960 (2001).
Schwieters, C.D., Kuszewski, J.J. & Clore, G.M. Using Xplor-NIH for NMR molecular structure determination. Prog. Nucl. Magn. Reson. Spectrosc. 48, 47–62 (2006).
Schwieters, C.D., Kuszewski, J.J., Tjandra, N. & Clore, G.M. Xplor-NIH NMR molecular structure determination package. J. Magn. Reson. 160, 65–73 (2003).
Chen, V.B. et al. MolProbity: all-atom structure validation for macromolecular crystallography. Acta Crystallogr. D Biol. Crystallogr. 66, 12–21 (2010).
Davis, I.W. et al. MolProbity: all-atom contacts and structure validation for proteins and nucleic acids. Nucleic Acids Res. 35, W375–W383 (2007).
Acknowledgements
We are grateful to S.F. Newbury (Brighton and Sussex Medical School, University of Sussex, Brighton, UK) for the kind gift of D. melanogaster XRN1 complementary DNA (cDNA) and XRN1 antibodies. This study was supported by the Max Planck Society and grants from the Deutsche Forschungsgemeinschaft (DFG, FOR855 to E.I. and the Gottfried Wilhelm Leibniz Program to E.I.).
Author information
Authors and Affiliations
Contributions
E.I. conceived the project. J.E.B. and E.H. carried out the immunoprecipitations and functional assays in S2 cells. J.E.B. and A.B. purified the complex. C.-T.C. carried out immunoprecipitations in human cells. G.H. was involved in an earlier phase of the project, cloned XRN1 and DCP1 fragments and performed preliminary immunoprecipitations and functional assays in S2 cells. V.T. collected and processed the NMR data. V.T. and M.C. solved the structure and built the model. J.E.B., V.T., A.B., M.C., O.W. and E.I. analyzed the structure and wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–6 (PDF 15006 kb)
Rights and permissions
About this article
Cite this article
Braun, J., Truffault, V., Boland, A. et al. A direct interaction between DCP1 and XRN1 couples mRNA decapping to 5′ exonucleolytic degradation. Nat Struct Mol Biol 19, 1324–1331 (2012). https://doi.org/10.1038/nsmb.2413
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb.2413
This article is cited by
-
Substrate promiscuity of Dicer toward precursors of the let-7 family and their 3′-end modifications
Cellular and Molecular Life Sciences (2024)
-
Ceg1 depletion reveals mechanisms governing degradation of non-capped RNAs in Saccharomyces cerevisiae
Communications Biology (2023)
-
Can microRNA become next-generation tools in molecular diagnostics and therapeutics? A systematic review
Egyptian Journal of Medical Human Genetics (2021)
-
Applications of miRNAs in cardiac development, disease progression and regeneration
Stem Cell Research & Therapy (2019)
-
Regulation of microRNA function in animals
Nature Reviews Molecular Cell Biology (2019)