Abstract
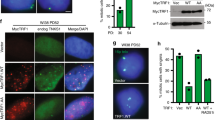
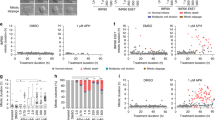
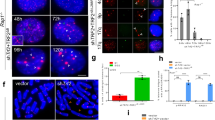
Telomere shortening and disruption of telomeric components are pathways that induce telomere deprotection. Here we describe another pathway, in which prolonged mitotic arrest induces damage signals at telomeres in human cells. Exposure to microtubule drugs, kinesin inhibitors, proteasome inhibitors or the disruption of proper chromosome cohesion resulted in the formation of damage foci at telomeres. Induction of mitotic telomere deprotection coincided with dissociation of TRF2 from telomeres, telomeric 3′-overhang degradation and ATM activation, and deprotection could be suppressed by TRF2 overexpression or inhibition of Aurora B kinase. Normal cells that escaped from prolonged mitotic arrest halted in the following G1 phase, whereas cells lacking p53 continued to cycle and became aneuploid. We propose a telomere-dependent mitotic-duration monitoring system that reacts to improper progression through mitosis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
de Lange, T. How telomeres solve the end-protection problem. Science 326, 948–952 (2009).
van Steensel, B., Smogorzewska, A. & de Lange, T. TRF2 protects human telomeres from end-to-end fusions. Cell 92, 401–413 (1998).
Karlseder, J., Broccoli, D., Dai, Y., Hardy, S. & de Lange, T. p53- and ATM-dependent apoptosis induced by telomeres lacking TRF2. Science 283, 1321–1325 (1999).
Denchi, E.L. & de Lange, T. Protection of telomeres through independent control of ATM and ATR by TRF2 and POT1. Nature 448, 1068–1071 (2007).
Guo, X. et al. Dysfunctional telomeres activate an ATM-ATR-dependent DNA damage response to suppress tumorigenesis. EMBO J. 26, 4709–4719 (2007).
Takai, H., Smogorzewska, A. & de Lange, T. DNA damage foci at dysfunctional telomeres. Curr. Biol. 13, 1549–1556 (2003).
d'Adda di Fagagna, F. et al. A DNA damage checkpoint response in telomere-initiated senescence. Nature 426, 194–198 (2003).
Cesare, A.J. et al. Spontaneous occurrence of telomeric DNA damage response in the absence of chromosome fusions. Nat. Struct. Mol. Biol. 16, 1244–1251 (2009).
Rieder, C.L. & Maiato, H. Stuck in division or passing through: what happens when cells cannot satisfy the spindle assembly checkpoint. Dev. Cell 7, 637–651 (2004).
Brito, D.A. & Rieder, C.L. Mitotic checkpoint slippage in humans occurs via cyclin B destruction in the presence of an active checkpoint. Curr. Biol. 16, 1194–1200 (2006).
Gascoigne, K.E. & Taylor, S.S. Cancer cells display profound intra- and interline variation following prolonged exposure to antimitotic drugs. Cancer Cell 14, 111–122 (2008).
Schvartzman, J.M., Sotillo, R. & Benezra, R. Mitotic chromosomal instability and cancer: mouse modelling of the human disease. Nat. Rev. Cancer 10, 102–115 (2010).
Ganem, N.J., Storchova, Z. & Pellman, D. Tetraploidy, aneuploidy and cancer. Curr. Opin. Genet. Dev. 17, 157–162 (2007).
Ganem, N.J. & Pellman, D. Limiting the proliferation of polyploid cells. Cell 131, 437–440 (2007).
Michaelis, C., Ciosk, R. & Nasmyth, K. Cohesins: chromosomal proteins that prevent premature separation of sister chromatids. Cell 91, 35–45 (1997).
Guacci, V., Koshland, D. & Strunnikov, A. A direct link between sister chromatid cohesion and chromosome condensation revealed through the analysis of MCD1 in S. cerevisiae. Cell 91, 47–57 (1997).
Kitagawa, R., Bakkenist, C.J., McKinnon, P.J. & Kastan, M.B. Phosphorylation of SMC1 is a critical downstream event in the ATM-NBS1-BRCA1 pathway. Genes Dev. 18, 1423–1438 (2004).
Kim, S.T., Xu, B. & Kastan, M.B. Involvement of the cohesin protein, Smc1, in Atm-dependent and independent responses to DNA damage. Genes Dev. 16, 560–570 (2002).
Yazdi, P.T. et al. SMC1 is a downstream effector in the ATM/NBS1 branch of the human S-phase checkpoint. Genes Dev. 16, 571–582 (2002).
Watrin, E. & Peters, J.M. The cohesin complex is required for the DNA damage-induced G2/M checkpoint in mammalian cells. EMBO J. 28, 2625–2635 (2009).
Watrin, E. et al. Human Scc4 is required for cohesin binding to chromatin, sister-chromatid cohesion, and mitotic progression. Curr. Biol. 16, 863–874 (2006).
Schmitz, J., Watrin, E., Lenart, P., Mechtler, K. & Peters, J.M. Sororin is required for stable binding of cohesin to chromatin and for sister chromatid cohesion in interphase. Curr. Biol. 17, 630–636 (2007).
Downing, K.H. Structural basis for the interaction of tubulin with proteins and drugs that affect microtubule dynamics. Annu. Rev. Cell Dev. Biol. 16, 89–111 (2000).
Shen, L. et al. Cell death by bortezomib-induced mitotic catastrophe in natural killer lymphoma cells. Mol. Cancer Ther. 7, 3807–3815 (2008).
Müller, C. et al. Inhibitors of kinesin Eg5: antiproliferative activity of monastrol analogues against human glioblastoma cells. Cancer Chemother. Pharmacol. 59, 157–164 (2007).
Karlseder, J., Smogorzewska, A. & de Lange, T. Senescence induced by altered telomere state, not telomere loss. Science 295, 2446–2449 (2002).
Sfeir, A. et al. Mammalian telomeres resemble fragile sites and require TRF1 for efficient replication. Cell 138, 90–103 (2009).
Giunta, S., Belotserkovskaya, R. & Jackson, S.P. DNA damage signaling in response to double-strand breaks during mitosis. J. Cell Biol. 190, 197–207 (2010).
Andreassen, P.R., Lohez, O.D., Lacroix, F.B. & Margolis, R.L. Tetraploid state induces p53-dependent arrest of nontransformed mammalian cells in G1. Mol. Biol. Cell 12, 1315–1328 (2001).
Celli, G.B. & de Lange, T. DNA processing is not required for ATM-mediated telomere damage response after TRF2 deletion. Nat. Cell Biol. 7, 712–718 (2005).
Musacchio, A. & Salmon, E.D. The spindle-assembly checkpoint in space and time. Nat. Rev. Mol. Cell Biol. 8, 379–393 (2007).
Santaguida, S., Vernieri, C., Villa, F., Ciliberto, A. & Musacchio, A. Evidence that Aurora B is implicated in spindle checkpoint signalling independently of error correction. EMBO J. 30, 1508–1519 (2011).
Kim, H.J., Cho, J.H., Quan, H. & Kim, J.R. Down-regulation of Aurora B kinase induces cellular senescence in human fibroblasts and endothelial cells through a p53-dependent pathway. FEBS Lett. 585, 3569–3576 (2011).
Santaguida, S., Tighe, A., D'Alise, A.M., Taylor, S.S. & Musacchio, A. Dissecting the role of MPS1 in chromosome biorientation and the spindle checkpoint through the small molecule inhibitor reversine. J. Cell Biol. 190, 73–87 (2010).
Dalton, W.B., Nandan, M.O., Moore, R.T. & Yang, V.W. Human cancer cells commonly acquire DNA damage during mitotic arrest. Cancer Res. 67, 11487–11492 (2007).
Quignon, F. et al. Sustained mitotic block elicits DNA breaks: one-step alteration of ploidy and chromosome integrity in mammalian cells. Oncogene 26, 165–172 (2007).
Janssen, A. & Medema, R.H. Mitosis as an anti-cancer target. Oncogene 30, 2799–2809 (2011).
Lanni, J.S. & Jacks, T. Characterization of the p53-dependent postmitotic checkpoint following spindle disruption. Mol. Cell Biol. 18, 1055–1064 (1998).
Halbert, C.L., Demers, G.W. & Galloway, D.A. The E7 gene of human papillomavirus type 16 is sufficient for immortalization of human epithelial cells. J. Virol. 65, 473–478 (1991).
Crabbe, L., Verdun, R.E., Haggblom, C.I. & Karlseder, J. Defective telomere lagging strand synthesis in cells lacking WRN helicase activity. Science 306, 1951–1953 (2004).
Tiscornia, G., Singer, O. & Verma, I.M. Production and purification of lentiviral vectors. Nat. Protoc. 1, 241–245 (2006).
Sarbassov, D.D., Guertin, D.A., Ali, S.M. & Sabatini, D.M. Phosphorylation and regulation of Akt/PKB by the rictor-mTOR complex. Science 307, 1098–1101 (2005).
O'Sullivan, R.J., Kubicek, S., Schreiber, S.L. & Karlseder, J. Reduced histone biosynthesis and chromatin changes arising from a damage signal at telomeres. Nat. Struct. Mol. Biol. 17, 1218–1225 (2010).
Verdun, R.E. & Karlseder, J. The DNA damage machinery and homologous recombination pathway act consecutively to protect human telomeres. Cell 127, 709–720 (2006).
Acknowledgements
We thank J.M. Peters (Institute for Molecular Pathology), T. de Lange (The Rockefeller University) and P. Baumann (Stowers Institute for Medical Research) for antibodies; D. Gibbs (The Salk Institute) for production of lentivirus; and the Karlseder Laboratory for comments. M.T.H. is supported by a Human Frontier Science Program Long Term Fellowship and a Japan Society for the Promotion of Science Postdoctoral Fellowship for Research Abroad. A.J.C. is supported by a training grant from the US National Institutes of Health (NIH) (5T32CA009370-29). This work was supported by the Salk Institute Cancer Center Core Grant P30 CA014195-38 and grants from the NIH to J.K. (AG025837, GM087476).
Author information
Authors and Affiliations
Contributions
M.T.H. designed and carried out experiments and wrote the manuscript, A.J.C. designed and carried out experiments and wrote the manuscript, J.A.J.F. provided imaging expertise, E.L.-D. designed and carried out experiments, and J.K. designed experiments and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–6 (PDF 716 kb)
Rights and permissions
About this article
Cite this article
Hayashi, M., Cesare, A., Fitzpatrick, J. et al. A telomere-dependent DNA damage checkpoint induced by prolonged mitotic arrest. Nat Struct Mol Biol 19, 387–394 (2012). https://doi.org/10.1038/nsmb.2245
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb.2245
This article is cited by
-
A pan-cancer landscape of telomeric content shows that RAD21 and HGF alterations are associated with longer telomeres
Genome Medicine (2022)
-
cGAS guards against chromosome end-to-end fusions during mitosis and facilitates replicative senescence
Protein & Cell (2022)
-
Suppression of genomic instability by replicative senescence and crisis
Genome Instability & Disease (2020)
-
The balance between mitotic death and mitotic slippage in acute leukemia: a new therapeutic window?
Journal of Hematology & Oncology (2019)
-
Replication stress induces mitotic death through parallel pathways regulated by WAPL and telomere deprotection
Nature Communications (2019)