Abstract
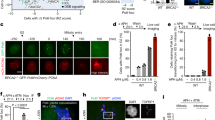
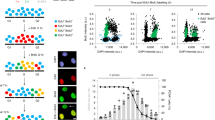
Homologous recombination facilitates accurate repair of DNA double-strand breaks (DSBs) during the S and G2 phases of the cell cycle by using intact sister chromatids as sequence templates. Homologous recombination capacity is maximized in S and G2 by cyclin-dependent kinase (CDK) phosphorylation of CtIP, which subsequently interacts with BRCA1 and the Mre11–Rad50–NBS1 (MRN) complex. Here we show that, in human and mouse, Mre11 controls these events through a direct interaction with CDK2 that is required for CtIP phosphorylation and BRCA1 interaction in normally dividing cells. CDK2 binds the C terminus of Mre11, which is absent in an inherited allele causing ataxia telangiectasia–like disorder. This newly uncovered role for Mre11 does not require ATM activation or nuclease activities. Therefore, functions of MRN are not restricted to DNA damage responses but include regulating homologous recombination capacity during the normal mammalian cell cycle.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Jackson, S.P. & Bartek, J. The DNA-damage response in human biology and disease. Nature 461, 1071–1078 (2009).
Matsuoka, S. et al. ATM and ATR substrate analysis reveals extensive protein networks responsive to DNA damage. Science 316, 1160–1166 (2007).
McKinnon, P.J. & Caldecott, K.W. DNA strand break repair and human genetic disease. Annu. Rev. Genomics Hum. Genet. 8, 37–55 (2007).
San Filippo, J., Sung, P. & Klein, H. Mechanism of eukaryotic homologous recombination. Annu. Rev. Biochem. 77, 229–257 (2008).
Mimitou, E.P. & Symington, L.S. DNA end resection-Unraveling the tail. DNA Repair (Amst.) 10, 344–348 (2011).
Buis, J. et al. Mre11 nuclease activity has essential roles in DNA repair and genomic stability distinct from ATM activation. Cell 135, 85–96 (2008).
Yamaguchi-Iwai, Y. et al. Mre11 is essential for the maintenance of chromosomal DNA in vertebrate cells. EMBO J. 18, 6619–6629 (1999).
Takeda, S., Nakamura, K., Taniguchi, Y. & Paull, T.T. Ctp1/CtIP and the MRN complex collaborate in the initial steps of homologous recombination. Mol. Cell 28, 351–352 (2007).
Sartori, A.A. et al. Human CtIP promotes DNA end resection. Nature 450, 509–514 (2007).
Williams, G.J., Lees-Miller, S.P. & Tainer, J.A. Mre11-Rad50-Nbs1 conformations and the control of sensing, signaling, and effector responses at DNA double-strand breaks. DNA Repair (Amst.) 9, 1299–1306 (2010).
Hopfner, K.P. et al. Structural biochemistry and interaction architecture of the DNA double-strand break repair Mre11 nuclease and Rad50-ATPase. Cell 105, 473–485 (2001).
Williams, R.S. et al. Mre11 dimers coordinate DNA end bridging and nuclease processing in double-strand-break repair. Cell 135, 97–109 (2008).
de Jager, M. et al. Human Rad50/Mre11 is a flexible complex that can tether DNA ends. Mol. Cell 8, 1129–1135 (2001).
Nimonkar, A.V. et al. BLM-DNA2-RPA-MRN and EXO1-BLM-RPA-MRN constitute two DNA end resection machineries for human DNA break repair. Genes Dev. 25, 350–362 (2011).
Lee, J.H. & Paull, T.T. Direct activation of the ATM protein kinase by the Mre11/Rad50/Nbs1 complex. Science 304, 93–96 (2004).
Lee, J.H. & Paull, T.T. ATM activation by DNA double-strand breaks through the Mre11-Rad50-Nbs1 complex. Science 308, 551–554 (2005).
Lloyd, J. et al. A supramodular FHA/BRCT-repeat architecture mediates Nbs1 adaptor function in response to DNA damage. Cell 139, 100–111 (2009).
Williams, R.S. et al. Nbs1 flexibly tethers Ctp1 and Mre11-Rad50 to coordinate DNA double-strand break processing and repair. Cell 139, 87–99 (2009).
Yun, M.H. & Hiom, K. CtIP-BRCA1 modulates the choice of DNA double-strand-break repair pathway throughout the cell cycle. Nature 459, 460–463 (2009).
Chen, L., Nievera, C.J., Lee, A.Y. & Wu, X. Cell cycle-dependent complex formation of BRCA1.CtIP.MRN is important for DNA double-strand break repair. J. Biol. Chem. 283, 7713–7720 (2008).
You, Z. et al. CtIP links DNA double-strand break sensing to resection. Mol. Cell 36, 954–969 (2009).
Huertas, P. & Jackson, S.P. Human CtIP mediates cell cycle control of DNA end resection and double strand break repair. J. Biol. Chem. 284, 9558–9565 (2009).
Germani, A. et al. SIAH-1 interacts with CtIP and promotes its degradation by the proteasome pathway. Oncogene 22, 8845–8851 (2003).
Yu, X. & Chen, J. DNA damage-induced cell cycle checkpoint control requires CtIP, a phosphorylation-dependent binding partner of BRCA1 C-terminal domains. Mol. Cell. Biol. 24, 9478–9486 (2004).
Cerqueira, A. et al. Overall Cdk activity modulates the DNA damage response in mammalian cells. J. Cell Biol. 187, 773–780 (2009).
Satyanarayana, A. & Kaldis, P. Mammalian cell-cycle regulation: several Cdks, numerous cyclins and diverse compensatory mechanisms. Oncogene 28, 2925–2939 (2009).
Huertas, P., Cortes-Ledesma, F., Sartori, A.A., Aguilera, A. & Jackson, S.P. CDK targets Sae2 to control DNA-end resection and homologous recombination. Nature 455, 689–692 (2008).
Müller-Tidow, C. et al. The cyclin A1–CDK2 complex regulates DNA double-strand break repair. Mol. Cell. Biol. 24, 8917–8928 (2004).
Deans, A.J. et al. Cyclin-dependent kinase 2 functions in normal DNA repair and is a therapeutic target in BRCA1-deficient cancers. Cancer Res. 66, 8219–8226 (2006).
Stewart, G.S. et al. The DNA double-strand break repair gene hMRE11 is mutated in individuals with an ataxia-telangiectasia-like disorder. Cell 99, 577–587 (1999).
Theunissen, J.W. et al. Checkpoint failure and chromosomal instability without lymphomagenesis in Mre11(ATLD1/ATLD1) mice. Mol. Cell 12, 1511–1523 (2003).
Frappart, P.O. & McKinnon, P.J. Ataxia-telangiectasia and related diseases. Neuromolecular Med. 8, 495–511 (2006).
Difilippantonio, S. et al. Role of Nbs1 in the activation of the Atm kinase revealed in humanized mouse models. Nat. Cell Biol. 7, 675–685 (2005).
Adelman, C.A., De, S. & Petrini, J.H. Rad50 is dispensable for the maintenance and viability of postmitotic tissues. Mol. Cell. Biol. 29, 483–492 (2009).
Bakkenist, C.J. & Kastan, M.B. DNA damage activates ATM through intermolecular autophosphorylation and dimer dissociation. Nature 421, 499–506 (2003).
Williams, B.R. et al. A murine model of Nijmegen breakage syndrome. Curr. Biol. 12, 648–653 (2002).
Berthet, C., Aleem, E., Coppola, V., Tessarollo, L. & Kaldis, P. Cdk2 knockout mice are viable. Curr. Biol. 13, 1775–1785 (2003).
Santamaría, D. et al. Cdk1 is sufficient to drive the mammalian cell cycle. Nature 448, 811–815 (2007).
Meijer, L. & Raymond, E. Roscovitine and other purines as kinase inhibitors. From starfish oocytes to clinical trials. Acc. Chem. Res. 36, 417–425 (2003).
Chien, C.T., Bartel, P.L., Sternglanz, R. & Fields, S. The two-hybrid system: a method to identify and clone genes for proteins that interact with a protein of interest. Proc. Natl. Acad. Sci. USA 88, 9578–9582 (1991).
Pierce, A.J., Johnson, R.D., Thompson, L.H. & Jasin, M. XRCC3 promotes homology-directed repair of DNA damage in mammalian cells. Genes Dev. 13, 2633–2638 (1999).
Mirzoeva, O.K. & Petrini, J.H. DNA damage-dependent nuclear dynamics of the Mre11 complex. Mol. Cell. Biol. 21, 281–288 (2001).
Lisby, M., Barlow, J.H., Burgess, R.C. & Rothstein, R. Choreography of the DNA damage response: spatiotemporal relationships among checkpoint and repair proteins. Cell 118, 699–713 (2004).
Rupnik, A., Lowndes, N.F. & Grenon, M. MRN and the race to the break. Chromosoma 119, 115–135 (2010).
Morales, M. et al. The Rad50S allele promotes ATM-dependent DNA damage responses and suppresses ATM deficiency: implications for the Mre11 complex as a DNA damage sensor. Genes Dev. 19, 3043–3054 (2005).
Hopfner, K.P. et al. Mre11 and Rad50 from Pyrococcus furiosus: cloning and biochemical characterization reveal an evolutionarily conserved multiprotein machine. J. Bacteriol. 182, 6036–6041 (2000).
Cromie, G.A. & Leach, D.R. Recombinational repair of chromosomal DNA double-strand breaks generated by a restriction endonuclease. Mol. Microbiol. 41, 873–883 (2001).
Dinkelmann, M. et al. Multiple functions of MRN in end-joining pathways during isotype class switching. Nat. Struct. Mol. Biol. 16, 808–813 (2009).
Zha, S., Sekiguchi, J., Brush, J.W., Bassing, C.H. & Alt, F.W. Complementary functions of ATM and H2AX in development and suppression of genomic instability. Proc. Natl. Acad. Sci. USA 105, 9302–9306 (2008).
Helmink, B.A. et al. MRN complex function in the repair of chromosomal Rag-mediated DNA double-strand breaks. J. Exp. Med. 206, 669–679 (2009).
Acknowledgements
The authors thank M. Weitzman (Salk Institute), P. Kaldis (A*STAR, Institute of Molecular and Cell Biology) and B. Sleckman (Washington University) for providing cell lines; J. Sekiguchi (University of Michigan) for providing ATM−/− mice; and M. Jasin (Memorial Sloan-Kettering Cancer Center) for providing DR-GFP plasmid. We thank T. Wilson, X. Yu, J. Sekiguchi, G. Dressler and C. Canman for helpful discussions regarding the manuscript. Support for this work was provided by US National Institutes of Health (NIH) grant R01-HL079118 (to D.O.F.), the Leukemia and Lymphoma Society (to D.O.F.), the University of Michigan Cancer Center Support Grant 5-P30-CA46592 (D.O.F.), NIH F32-GM087073 (J.B.) and NIH T32-AI007413 (E.S.).
Author information
Authors and Affiliations
Contributions
J.B. planned and conducted all experiments except the two-hybrid and B-lymphocyte analyses, analyzed and interpreted data from all experiments and participated in writing the manuscript. T.S. conducted and interpreted the two-hybrid analyses. E.S. carried out the B lymphocyte western blot analysis from spleens of mice from complex breeds. D.O.F. participated in design of all experiments, analyses and interpretation of data and in writing of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–5 (PDF 4353 kb)
Rights and permissions
About this article
Cite this article
Buis, J., Stoneham, T., Spehalski, E. et al. Mre11 regulates CtIP-dependent double-strand break repair by interaction with CDK2. Nat Struct Mol Biol 19, 246–252 (2012). https://doi.org/10.1038/nsmb.2212
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb.2212
This article is cited by
-
Cyclers’ kinases in cell division: from molecules to cancer therapy
Cell Death & Differentiation (2023)
-
CtIP-dependent nascent RNA expression flanking DNA breaks guides the choice of DNA repair pathway
Nature Communications (2022)
-
Cellular functions of the protein kinase ATM and their relevance to human disease
Nature Reviews Molecular Cell Biology (2021)
-
CopyCatchers are versatile active genetic elements that detect and quantify inter-homolog somatic gene conversion
Nature Communications (2021)
-
Suppression of DDX39B sensitizes ovarian cancer cells to DNA-damaging chemotherapeutic agents via destabilizing BRCA1 mRNA
Oncogene (2020)