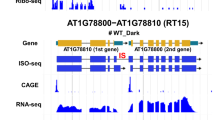
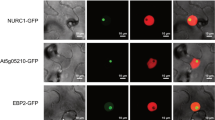
Abstract
Transcription factor IIIA (TFIIIA) is required for eukaryotic synthesis of 5S ribosomal RNA by RNA polymerase III. Here we report the discovery of a structured RNA element with clear resemblance to 5S rRNA that is conserved within TFIIIA precursor mRNAs from diverse plant lineages. TFIIIA protein expression is controlled by alternative splicing of the exon containing the plant 5S rRNA mimic (P5SM). P5SM triggers exon skipping upon binding of ribosomal protein L5, a natural partner of 5S rRNA, which demonstrates the functional adaptation of its structural mimicry. As the exon-skipped splice product encodes full-length TFIIIA protein, these results reveal a ribosomal protein–mRNA interaction that is involved in 5S rRNA synthesis and has implications for cross-coordination of ribosomal components. This study also provides insight into the origin and function of a newfound class of structured RNA that regulates alternative splicing.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Accession codes
References
Matlin, A.J., Clark, F. & Smith, C.W. Understanding alternative splicing: towards a cellular code. Nat. Rev. Mol. Cell Biol. 6, 386–398 (2005).
Wang, B.B. & Brendel, V. Genomewide comparative analysis of alternative splicing in plants. Proc. Natl. Acad. Sci. USA 103, 7175–7180 (2006).
Buratti, E. & Baralle, F.E. Influence of RNA secondary structure on the pre-mRNA splicing process. Mol. Cell. Biol. 24, 10505–10514 (2004).
Hiller, M., Zhang, Z., Backofen, R. & Stamm, S. Pre-mRNA secondary structures influence exon recognition. PLoS Genet. 3, e204 (2007).
Buckanovich, R.J. & Darnell, R.B. The neuronal RNA binding protein Nova-1 recognizes specific RNA targets in vitro and in vivo. Mol. Cell. Biol. 17, 3194–3201 (1997).
Blanchette, M. & Chabot, B. A highly stable duplex structure sequesters the 5′ splice site region of hnRNP A1 alternative exon 7B. RNA 3, 405–419 (1997).
Ehresmann, C. et al. Molecular mimicry in translational regulation: the case of ribosomal protein S15. RNA Biol. 1, 66–73 (2004).
Merianos, H.J., Wang, J. & Moore, P.B. The structure of a ribosomal protein S8/spc operon mRNA complex. RNA 10, 954–964 (2004).
Chao, J.A. & Williamson, J.R. Joint x-ray and NMR refinement of the yeast L30e-mRNA complex. Structure 12, 1165–1176 (2004).
Vilardell, J., Chartrand, P., Singer, R.H. & Warner, J.R. The odyssey of a regulated transcript. RNA 6, 1773–1780 (2000).
Cheah, M.T., Wachter, A., Sudarsan, N. & Breaker, R.R. Control of alternative RNA splicing and gene expression by eukaryotic riboswitches. Nature 447, 497–500 (2007).
Wachter, A. et al. Riboswitch control of gene expression in plants by splicing and alternative 3′ end processing of mRNAs. Plant Cell 19, 3437–3450 (2007).
Kubodera, T. et al. Thiamine-regulated gene expression of Aspergillus oryzae thiA requires splicing of the intron containing a riboswitch-like domain in the 5′-UTR. FEBS Lett. 555, 516–520 (2003).
Sudarsan, N., Barrick, J.E. & Breaker, R.R. Metabolite-binding RNA domains are present in the genes of eukaryotes. RNA 9, 644–647 (2003).
Bocobza, S. et al. Riboswitch-dependent gene regulation and its evolution in the plant kingdom. Genes Dev. 21, 2874–2879 (2007).
Croft, M.T., Moulin, M., Webb, M.E. & Smith, A.G. Thiamine biosynthesis in algae is regulated by riboswitches. Proc. Natl. Acad. Sci. USA 104, 20770–20775 (2007).
Nishihara, H., Smit, A.F. & Okada, N. Functional noncoding sequences derived from SINEs in the mammalian genome. Genome Res. 16, 864–874 (2006).
Kapitonov, V.V. & Jurka, J. A novel class of SINE elements derived from 5S rRNA. Mol. Biol. Evol. 20, 694–702 (2003).
Kalendar, R. et al. Cassandra retrotransposons carry independently transcribed 5S RNA. Proc. Natl. Acad. Sci. USA 105, 5833–5838 (2008).
Wolffe, A.P. The role of transcription factors, chromatin structure and DNA replication in 5S RNA gene regulation. J. Cell Sci. 107, 2055–2063 (1994).
Rogers, S.O. & Bendich, A.J. Ribosomal RNA genes in plants—variability in copy number and in the intergenic spacer. Plant Mol. Biol. 9, 509–520 (1987).
Douet, J. & Tourmente, S. Transcription of the 5S rRNA heterochromatic genes is epigenetically controlled in Arabidopsis thaliana and Xenopus laevis. Heredity 99, 5–13 (2007).
Andrews, M.T. & Brown, D.D. Transient activation of oocyte 5S RNA genes in Xenopus embryos by raising the level of the trans-acting factor TFIIIA. Cell 51, 445–453 (1987).
Yao, Z., Weinberg, Z. & Ruzzo, W.L. CMfinder—a covariance model based RNA motif finding algorithm. Bioinformatics 22, 445–452 (2006).
Soukup, G.A. & Breaker, R.R. Relationship between internucleotide linkage geometry and the stability of RNA. RNA 5, 1308–1325 (1999).
Griffiths-Jones, S. et al. Rfam: annotating non-coding RNAs in complete genomes. Nucleic Acids Res. 33, D121–D124 (2005).
Mathieu, O. et al. Identification and characterization of transcription factor IIIA and ribosomal protein L5 from Arabidopsis thaliana. Nucleic Acids Res. 31, 2424–2433 (2003).
Pieler, T. & Theunissen, O. TFIIIA: nine fingers—three hands? Trends Biochem. Sci. 18, 226–230 (1993).
Yoine, M., Ohto, M.A., Onai, K., Mita, S. & Nakamura, K. The lba1 mutation of UPF1 RNA helicase involved in nonsense-mediated mRNA decay causes pleiotropic phenotypic changes and altered sugar signalling in Arabidopsis. Plant J. 47, 49–62 (2006).
Szymanski, M., Barciszewska, M.Z., Erdmann, V.A. & Barciszewski, J. 5S rRNA: structure and interactions. Biochem. J. 371, 641–651 (2003).
Ban, N., Nissen, P., Hansen, J., Moore, P.B. & Steitz, T.A. The complete atomic structure of the large ribosomal subunit at 2.4 angstrom resolution. Science 289, 905–920 (2000).
Scripture, J.B. & Huber, P.W. Analysis of the binding of Xenopus ribosomal protein L5 to oocyte 5S ribosomal RNA—the major determinants of recognition are located in helix III-loop C. J. Biol. Chem. 270, 27358–27365 (1995).
Zengel, J.M. & Lindahl, L. Diverse mechanisms for regulating ribosomal protein synthesis in Escherichia coli. Prog. Nucleic Acid Res. Mol. Biol. 47, 331–370 (1994).
Nomura, M., Yates, J.L., Dean, D. & Post, L.E. Feedback regulation of ribosomal protein gene expression in Escherichia coli: structural homology of ribosomal RNA and ribosomal protein MRNA. Proc. Natl. Acad. Sci. USA 77, 7084–7088 (1980).
McIntosh, K.B. & Bonham-Smith, P.C. Ribosomal protein gene regulation: what about plants? Can. J. Bot. 84, 342–362 (2006).
Sorek, R., Ast, G. & Graur, D. Alu-containing exons are alternatively spliced. Genome Res. 12, 1060–1067 (2002).
Bejerano, G. et al. A distal enhancer and an ultraconserved exon are derived from a novel retroposon. Nature 441, 87–90 (2006).
Cloix, C., Yukawa, Y., Tutois, S., Sugiura, M. & Tourmente, S. In vitro analysis of the sequences required for transcription of the Arabidopsis thaliana 5S rRNA genes. Plant J. 35, 251–261 (2003).
McBryant, S.J. et al. Interaction of the RNA binding fingers of Xenopus transcription factor IIIA with specific regions of 5S ribosomal RNA. J. Mol. Biol. 248, 44–57 (1995).
Staiger, D., Zecca, L., Wieczorek Kirk, D.A., Apel, K. & Eckstein, L. The circadian clock regulated RNA-binding protein AtGRP7 autoregulates its expression by influencing alternative splicing of its own pre-mRNA. Plant J. 33, 361–371 (2003).
Sureau, A., Gattoni, R., Dooghe, Y., Stevenin, J. & Soret, J. SC35 autoregulates its expression by promoting splicing events that destabilize its mRNAs. EMBO J. 20, 1785–1796 (2001).
Pittman, R.H., Andrews, M.T. & Setzer, D.R. A feedback loop coupling 5 S rRNA synthesis to accumulation of a ribosomal protein. J. Biol. Chem. 274, 33198–33201 (1999).
Li, B., Vilardell, J. & Warner, J.R. An RNA structure involved in feedback regulation of splicing and of translation is critical for biological fitness. Proc. Natl. Acad. Sci. USA 93, 1596–1600 (1996).
House, A.E. & Lynch, K.W. Regulation of alternative splicing: more than just the ABCs. J. Biol. Chem. 283, 1217–1221 (2008).
Egoavil, C., Marton, H.A., Baynton, C.E., McCullough, A.J. & Schuler, M.A. Structural analysis of elements contributing to 5′ splice site selection in plant pre-mRNA transcripts. Plant J. 12, 971–980 (1997).
Reddy, A.S. Alternative splicing of pre-messenger RNAs in plants in the genomic era. Annu. Rev. Plant Biol. 58, 267–294 (2007).
Sudarsan, N. et al. Tandem riboswitch architectures exhibit complex gene control functions. Science 314, 300–304 (2006).
Weinberg, Z. et al. Identification of 22 candidate structured RNAs in bacteria using the CMfinder comparative genomics pipeline. Nucleic Acids Res. 35, 4809–4819 (2007).
Hofgen, R. & Willmitzer, L. Biochemical and genetic analysis of different patatin isoforms expressed in various organs of potato (Solanum tuberosum). Plant Sci. 66, 221–230 (1990).
Acknowledgements
We thank K. Corbino, K. Leppek and A. Westermann for technical assistance, Z. Weinberg for advice on bioinformatics, E. Puerta-Fernandez for advice on gel-shift assays and A. Roth for critical reading of the manuscript. M.C.H. is supported by a Career Award at the Scientific Interface from the Burroughs Wellcome Fund, and A.W. was supported by the German Research Foundation while in the Breaker laboratory. The research was supported by a grant from the US National Institutes of Health (GM068819). RNA science in the Breaker laboratory also is supported by the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Contributions
M.C.H. conceived of and led project, performed bioinformatics analysis, designed and carried out in vitro structural probing and binding experiments, and wrote the manuscript. A.W. designed and carried out RT-PCR and qRT-PCR analyses and in vivo reporter assays, and edited the manuscript. R.R.B. advised on the project and edited the manuscript. All authors contributed to discussions regarding the data and their interpretation.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–11, Supplementary Table 1, Supplementary Methods (PDF 8796 kb)
Rights and permissions
About this article
Cite this article
Hammond, M., Wachter, A. & Breaker, R. A plant 5S ribosomal RNA mimic regulates alternative splicing of transcription factor IIIA pre-mRNAs. Nat Struct Mol Biol 16, 541–549 (2009). https://doi.org/10.1038/nsmb.1588
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb.1588