Abstract
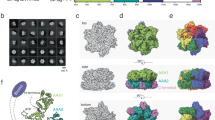
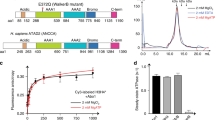
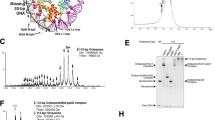
The NMR structure of budding yeast chaperone Chz1 complexed with histones H2A.Z-H2B has been determined. Chz1 forms a long irregular chain capped by two short α-helices, and uses both positively and negatively charged residues to stabilize the histone dimer. A molecular model that docks Chz1 onto the nucleosome has implications for its potential functions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Accession codes
References
De Koning, L., Corpet, A., Haber, J.E. & Almouzni, G. Nat. Struct. Mol. Biol. 14, 997–1007 (2007).
Mizuguchi, G. et al. Science 303, 343–348 (2003).
Luk, E. et al. Mol. Cell 25, 357–368 (2007).
Wu, W.H. et al. Nat. Struct. Mol. Biol. 12, 1064–1071 (2005).
Suto, R.K., Clarkson, M.J., Tremethick, D.J. & Luger, K. Nat. Struct. Biol. 7, 1121–1124 (2000).
English, C.M., Adkins, M.W., Carson, J.J., Churchill, M.E. & Tyler, J.K. Cell 127, 495–508 (2006).
Natsume, R. et al. Nature 446, 338–341 (2007).
Barbera, A.J. et al. Science 311, 856–861 (2006).
Luger, K., Mader, A.W., Richmond, R.K., Sargent, D.F. & Richmond, T.J. Nature 389, 251–260 (1997).
Hansen, D.F. et al. J. Am. Chem. Soc. 129, 11468–11479 (2007).
Acknowledgements
We thank A. Bax, C. Klee and M. Lichten for comments. This work was supported by the intramural program of the US National Cancer Institute (C.W. and Y.B.) and a grant from the Canadian Institutes of Health Research (L.E.K.). D.F.H. is the recipient of a postdoctoral fellowship from the Danish Agency for Science, Technology and Innovation.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–6, Supplementary Table 1 and Supplementary Methods (PDF 2979 kb)
Rights and permissions
About this article
Cite this article
Zhou, Z., Feng, H., Hansen, D. et al. NMR structure of chaperone Chz1 complexed with histones H2A.Z-H2B. Nat Struct Mol Biol 15, 868–869 (2008). https://doi.org/10.1038/nsmb.1465
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb.1465
This article is cited by
-
OsChz1 acts as a histone chaperone in modulating chromatin organization and genome function in rice
Nature Communications (2020)
-
Structure-function relationship of H2A-H2B specific plant histone chaperones
Cell Stress and Chaperones (2020)
-
Dynamic intramolecular regulation of the histone chaperone nucleoplasmin controls histone binding and release
Nature Communications (2017)
-
Histone chaperone networks shaping chromatin function
Nature Reviews Molecular Cell Biology (2017)
-
Redundant Functions for Nap1 and Chz1 in H2A.Z Deposition
Scientific Reports (2017)