Abstract
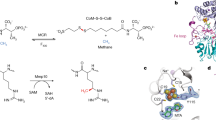
B12-dependent methionine synthase (MetH) from Escherichia coli is a large modular protein that uses bound cobalamin as an intermediate methyl carrier. Major domain rearrangements have been postulated to explain how cobalamin reacts with three different substrates: homocysteine, methyltetrahydrofolate and S-adenosylmethionine (AdoMet). Here we describe the 3.0 Å structure of a 65 kDa C-terminal fragment of MetH that spans the cobalamin- and AdoMet-binding domains, arranged in a conformation suitable for the methyl transfer from AdoMet to cobalamin that occurs during activation. In the conversion to the activation conformation, a helical domain that capped the cofactor moves 26 Å and rotates by 63°, allowing formation of a new interface between cobalamin and the AdoMet-binding (activation) domain. Interactions with the MetH activation domain drive the cobalamin away from its binding domain in a way that requires dissociation of the axial cobalt ligand and, thereby, provide a mechanism for control of the distribution of enzyme conformations.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Matthews, R.G. In Chemistry and biochemistry of B12 (ed. Banerjee, R.) 681–706 (Wiley, New York; 1999).
Taylor, R.T. & Weissbach, H. Arch. Biochem. Biophys. 129, 728–744 (1969).
Fujii, K. & Huennekens, F.M. J. Biol. Chem. 249, 6745–6753 (1974).
Banerjee, R.V., Harder, S.R., Ragsdale, S.W. & Matthews, R.G. Biochemistry 29, 1129–1135 (1990).
LeClerc, D. et al. Proc. Nat. Acad. Sci. USA 95, 3059–3064 (1998).
Banerjee, R.V., Johnston, N.L., Sobeski, J.K., Datta, P. & Matthews, R.G. J. Biol. Chem. 264, 13888–13895 (1989).
Drummond, J.T., Huang, S., Blumenthal, R.M. & Matthews, R.G. Biochemistry 32, 9290–9295 (1993).
Goulding, C.W., Postigo, D. & Matthews, R.G. Biochemistry 36, 8082–8091 (1997).
Drennan, C.L., Huang, S., Drummond, J.T., Matthews, R.G. & Ludwig, M.L. Science 266, 1669–1674 (1994).
Dixon, M., Huang, S., Matthews, R.G. & Ludwig, M.L. Structure 4, 1263–1275 (1996).
Bandarian, V. & Matthews, R.G. Biochemistry 40, 5056–5064 (2001).
Jarrett, J.T. et al. Biochemistry 35, 2464–2475 (1996).
Jarrett, J.T. et al. Bioorg. Med. Chem. 4, 1237–1246 (1996).
Ludwig, M.L. & Matthews, R.G. Annu. Rev. Biochem. 66, 269–313 (1997).
Drennan, C.L. et al. In Vitamin B12 and B12 proteins (eds Krautler, B., Arigoni, D. & Golding, B.T.) 133–156 (Wiley-VCH, Weinheim; 1998).
Matthews, R.G. In Enzymatic mechanisms (eds Frey, P.A. & Northrup, D.B.) 155–161 (IOS Press, Amsterdam; 1999).
Jarrett, J.T., Huang, S. & Matthews, R.G. Biochemistry 37, 5372–5382 (1998).
Jarrett, J.T., Hoover, D.M., Ludwig, M.L. & Matthews, R.G. Biochemistry 37, 12649–12658 (1998).
Hoover, D.M. et al. Biochemistry 36, 127–138 (1997).
Hayward, S. Proteins Struct. Funct. Genet. 36, 425–435 (1999).
Otwinowski, Z. & Minor, W. Methods Enzymol. 276, 307–326 (1997).
Tong, L. & Rossmann, M.G. Methods Enzymol. 276, 594–610 (1997).
Brünger, A.T. et al. Acta Crystallogr. D 54, 905–921 (1998).
Hall, D.A., Jordan-Starck, T.C., Loo, R.O., Ludwig, M.L. & Matthews, R.G. Biochemistry 39, 10711–10719 (2000).
Carson, M. Methods Enzymol. 277, 493–505 (1997).
McRee, D.E. J. Struct. Biol. 125, 156–165 (1999).
Merritt, E.A. & Bacon, D.J. Methods Enzymol. 277, 505–524 (1997).
Acknowledgements
Supported by NIH grants to M.L.L. and to R.G.M., and NRSA postdoctoral fellowships to V.B. and B.W.L. are acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bandarian, V., Pattridge, K., Lennon, B. et al. Domain alternation switches B12-dependent methionine synthase to the activation conformation. Nat Struct Mol Biol 9, 53–56 (2002). https://doi.org/10.1038/nsb738
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsb738
This article is cited by
-
Structure of full-length cobalamin-dependent methionine synthase and cofactor loading captured in crystallo
Nature Communications (2023)
-
Comparative proteome analysis of embryo and endosperm reveals central differential expression proteins involved in wheat seed germination
BMC Plant Biology (2015)
-
Macromolecular juggling by ubiquitylation enzymes
BMC Biology (2013)
-
Visualizing molecular juggling within a B12-dependent methyltransferase complex
Nature (2012)
-
Natural history of S-adenosylmethionine-binding proteins
BMC Structural Biology (2005)