Abstract
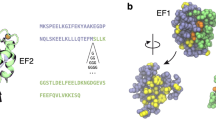
The crystal structures of two circular permutants of the α-spectrin SH3 domain with new termini within the RT loop (S19–P20s) and the distal loop (N47–D48s) have been determined at 2.02 and 1.77 Å resolution respectively. Both fold into the same three-dimensional structure as the wild-type SH3 domain except for the engineered loop that fuses the wild-type termini. The cleaved RT loop in S19–P20s loses nine conserved hydrogen bonds through local hydrogen bond unzipping; no hydrogen bond unzipping occurs in N47–D48s. The structures of the transition states for folding of wild-type α-spectrin SH3 domain and the two circular permutants have been examined by analysis of the folding kinetics of eight strategically distributed point mutants. Unlike the native structures, the transition states of the three proteins are considerably different, suggesting that there is no direct relationship between these two states in a protein.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Jackson, S.E. & Fersht, A.R. Folding of chymotrypsin inhibitor 2.1. Evidence for a two-state transition. Biochemistry 30, 10428–10435 (1991).
Alexander, P., Oman, J. & Bryan, P. Kinetic analysis of folding and unfolding the 56 amino acid IgG-binding domain of streptoccocal protein G. Biochemistry 31, 7243–7248 (1992).
Sosnick, T.R., Mayne, L., Hiller, R. & Englander, S.W. The barriers in protein folding. Nature Struct. Biol. 1, 149–156 (1994).
Villegas, V. et al. Evidence for a two state transition in the folding process of the activation domain of human procarboxipeptidase A2. Biochemistry 34, 15105–15110 (1995).
Kragelund, B.B., Robinson, C.V., Knudesn, J., Dobson, C.M. & Poulsen, F.M. Folding of a four-helix bundle: studies of acyl-coenzyme A binding protein. Biochemistry 34, 7217–7224 (1995).
Viguera, A.R., Martinez, J.C., Filimonov, V.V., Mateo, P.L. & Serrano, L. Thermodynamic and kinetic analysis of the SH3 domain of spectrin shows a two-state transition. Biochemistry 33, 2142–2150 (1994).
Fersht, A.R. Characterizing transition states in protein folding: an essential step in the puzzle. Curr. Opin. Struct. Biol. 5, 79–84 (1995).
Itzhaki, L.S., Otzen, D.E. & Fersht, A.R. The structure of the transition state for folding of chymotrypsin inhibitor 2 analysed by protein engineering methods: Evidence for a nucleation-condensation mechanism for protein folding. J. Mol. Biol. 254, 260–288 (1995).
Levinthal, C. Are there pathways for protein folding? J. Chim. Phys., 65, 44–45 (1968).
Kuwajima, K., Semisotnov, G.V., Finkelstein, A.V., Sugai, S. & Ptitsyn, O.B. Secondary structure of globular proteins at the early and the final stages in protein folding. FEBS. Lett. 3, 265–268 (1993).
Musacchio, A., Noble, M.E.M, Pautit, R., Wierenga, R. & Saraste, M. Crystal structure of a Src-homology 3 (SH3) domain. Nature 359, 851–855 (1992).
Musacchio, A., Wimanns, M. & Saraste, M. Structure and function of the SH3 domains. Progr. Biophys. molec. Biol. 61, 283–297 (1994).
Viguera, A.R., Blanco, F.J. & Serrano, L. The order of secondary structure elements does not determine the structure of a protein but does affect its folding kinetics. J. Mol. Biol. 247, 670–681 (1995).
Luzzati, V. Traitement statistique des erreurs dans la détermination des structures cristallines. Acta Crystallogr. 5, 802–810 (1952).
Musacchio, A., Saraste, M. & Wilmanns, M. High-resolution crystal structures of tyrosine kinase SH3 domains complexed with proline-rich peptides. Nature Struct. Biol. 1, 546–551 (1994).
Otzen, D.E., Itzhaki, L.S., Elmasry, N.F., Jackson, S.E. & Fersht, A.R. Structure of the transition state for the folding/unfolding of the barley chymotrypsin inhibitor 2 and its implication for the mechanism of protein folding Proc. Natl. Acad. Sci. USA 91, 10422–10425 (1994).
Fersht, A.R., Itzhaki, L.S., Elmasry, N.F., Matthews, J.M. & Otzen, D.E. Single versus parallel pathways of protein folding and fractional formation of structure in the transition state. Proc. Natl. Acad. Sci. USA 91, 10426–10429 (1994).
López-Hernández, E. & Serrano, L. Structure of the transition state for folding of the 129 aa protein CheY resembles that of a smaller protein, CI2. Folding & Design 1, 43–55 (1996).
Fersht, A.R. Optimization of rates of protein folding: the nucleation-condensation mechanism and its implications. Proc. Natl. Acad. Sci. USA 92, 10869–10873 (1995).
Viguera, A.R., Jiménez, M.A., Rico, M. & Serrano, L. Conformational analysis of peptides corresponding to β-hairpins and a β-sheet that represent the entire sequence of the α-spectrin SH3 domain. J. Mol. Biol. 255, 507–521 (1996).
Shakhnovich, E., Abkevich, V. & Ptitsyn, O. Conserved residues and the mechanism of protein folding. Nature 379, 96–98 (1996).
Harrison, S.C. & Durbin, R. Is there a single pathway for the folding of a polypeptide chain?. Proc. Natl. Acad. Sci. USA 82, 4028–4030 (1985).
Navaza, J. AMoRe: An automated package for molecular replacement. Acta Crystallogr. A50, 157–163 (1994).
Jones, T.A., Zou, J.-Y., Cowan, S.W. & Kjeldgaard, M. Improved methods for building protein models in electron density maps and the location of errors in these models. Acta Crystallogr. A47, 110–119 (1991).
Brünger, A.T. X-PLOR Version 3.1. Yale University, New Haven, CT, USA (1993).
Lamzin, V.S. & Wilson, K.S. Automated refinement of protein models. Acta Crystallogr. D49, 129–147 (1993).
Kunkel, T.A. Rapid and efficient site-directed mutagenesis without phenotypic selection. Proc. natn. Acad. Sci. U.S.A. 82, 488–492 (1985).
Peränen, J., Rikkonen, M., Hyvönen, M. & Kääriäinen, L. T7 vectors with a modified T7 lac promoter for expression of proteins in Escherichia coli. Anal. Biochem. 236, 371–373 (1996).
Matthews, J.M.&., Fersht, A.R. Exploring the energy surface of protein folding by structure reactivity relationships and engineered proteins: observation of Hammond behavior for the gross structure of the transition state and anti-Hammond behavior for structural elements for unfolding of barnase. Biochemistry 34, 6805–6814 (1995).
Vriend, G. WHAT IF - a molecular modeling and drug design program. J. Mol Graph. 8, 52–56 (1990).
Carson, M. Ribbons 2.0. J. Appl. Cryst. 24, 958–961 (1991).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Viguera, A., Serrano, L. & Wilmanns, M. Different folding transition states may result in the same native structure. Nat Struct Mol Biol 3, 874–880 (1996). https://doi.org/10.1038/nsb1096-874
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nsb1096-874
This article is cited by
-
Circular permutation at azurin’s active site slows down its folding
JBIC Journal of Biological Inorganic Chemistry (2023)
-
Versatile format of minichaperone-based protein fusion system
Scientific Reports (2019)
-
Adaptive local learning in sampling based motion planning for protein folding
BMC Systems Biology (2016)
-
Topological Quantities Determining the Folding/Unfolding Rate of Two-state Folding Proteins
Journal of Solution Chemistry (2010)
-
Predicting protein folding pathways at the mesoscopic level based on native interactions between secondary structure elements
BMC Bioinformatics (2008)