Abstract
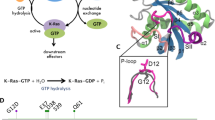
Ras p21 plays a major role in the control of cell growth, and oncogenic mutations of this protein have been found in human cancers. Unfortunately, the detailed mode of action of Ras p21 is still unclear, in spite of the great interest in this protein and the availability of its X-ray crystal structure. In particular, mutagenesis studies of different active site residues could not identify the general base for GTP hydrolysis. Here we tackle this question using a computer simulation approach with clear and reliable energy considerations and conclude that the most likely general base is the bound GTP itself. Obviously, the identification of such a general base cannot be easily accomplished by mutagenesis experiments.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Temeles, G.L., Gibbs, J.B., D'Alonzo, J.S., Sigal, I.S. & Scolnick, E.M. Yeast and mammalian Ras proteins have conserved biochemical properties. Nature 313, 700–703 (1985).
Gideou, P. et al. Mutational and kinetic analysis of the GTPase-activating protein (GAP)-p21 interaction. Molec. Cell. Biol., 12, 2050–2054 (1992).
De Vos, A.M. et al. 3-Dimensional structure of an oncogene protein - catalytic domain of human c-H-Ras. Science 239, 888–893 (1988).
Krengel, U. et al. 3-Dimensional structures of H-Ras p21 mutants -molecular basis for their inability to function as signal transduction switch molecules. Cell 62, 539–548 (1990).
Milburn, M. et al. Molecular switch for signal transduction -structural differences between active and inactive forms of protooncogenic Ras mutants. Science 247, 939–945 (1990).
Pai, E.F. et al. Structure of the guanine-nucleotide-binding domain of the Ha-Ras oncogene product p21 in the triphosphate conformation. Nature 341, 209–214 (1989).
Pai, E.F. et al. Refined crystal-structure of the triphosphate conformation of H-Ras p21 at 1.35Å resolution—implications for the mechanism of GTP hydrolysis. EMBO J. 9, 2351–2359 (1990).
Langen, R., Schweins, T. & Warshel, A. On the mechanism of guanosine triphosphate hydrolysis in Ras p21 proteins. Biochemistry 31, 8691–8696 (1992).
Prive, G.G. et al. X-Ray crystal-structures of transforming p21 Ras mutants suggests a transition-state stabilization mechanism for GTP hydrolysis. Proc. natn. Acad. Sci. U.S.A. 89, 3649–3653 (1992).
Chung, H.-H., Benson, D.R. & Schultz, P.G. Probing the structure and mechanism of Ras protein with an expanded genetic code. Science 259, 806–809 (1993).
Berchtold, H. et al. Crystal-structure of active elongation-factor Tu reveals major domain rearrangements. Nature 365, 368–368 (1993).
Noel, J.P., Hamm, H. & Sigler, P.B. The 2.2Å crystal structure of transducin complexed with GTPγs. Nature 366, 654–663 (1993).
Warshel, A. Computer Modeling of Chemical Reactions in Enzymes and Solutions (John Wiley, New York, 1991).
Aqvist, J. & Warshel, A. Calculations of the free energy profiles for the staphylococcal nuclease catalyzed reactions. Biochemistry 28, 4680–4689 (1989).
Lee, F.S., Chu, Z.T. & Warshel, A. Microscopic and semimicroscopic calculations of electrostatic energies in proteins by the POLARIS and ENZYMIX programs. J. comp. Chem. 14, 161–185 (1993).
Aqvist, J. & Warshel, A. Computer simulation of the initial proton-transfer step in human carbonic anhydrase. J. molec. Biol. 224, 7–14 (1992).
Silverman, D.N. & Lindskog, S. The catalytic mechanism of carbonic anhydrase- Implications of a rate-limiting protolysis of water. Accts. chem. Res. 21, 30 (1988).
Eigen, M. & de Mayer, L. Kinetics of neutralization Z. Elektrochem. 59, 986–993 (1955).
Guthrie, J.P. Hydration and dehydration of phosphoric acid derivatives: free energies of formation of the pentacoordinate intermediates for phosphate ester hydrolysis and of monomeric metaphosphate. J. Am. chem. Soc. 99, 3991–4001 (1977).
Warshel, A. & Russell, S. Calculations of electrostatic interactions in biological systems and in solution. Q. Rev. Biophys. 17, 283–422 (1984).
Warshel, A. Calculations of enzymatic reactions: calculations of pKa, proton transfer reactions, and general acid catalysis reactions in enzymes. Biochemistry 20, 3167 (1981).
Feuerstein, J., Goody, R.S. & Webb, M.R. The mechanism of guanosine nucleotide hydrolysis by p21 c-Ha-Ras. J. biol. Chem. 264, 6188–6190 (1989).
Harmark, K., Anborgh, P.H., Merola, M., Clark-B.F.C & Parmeggiani, A. Substitution of aspartic acid-80, a residue involved in coordination of magnesium, weakens the GTP binding and strongly enhances the GTPase of the G-domain of elongation factor-Tu. Biochemistry 31, 7367–7372 (1992).
Mistou, M.Y., Cool, R.H. & Parmeggiani, A. Effects of ions on the intrinsic activities of C-H-Ras protein p21 - a comparison with elongation factor-Tu. Eur. J. Biochem. 204, 179 (1992).
Williams, A. Concerted mechanisms of acyl group transfer reactions in solution. Accts. chem. Res. 22, 387–392 (1989).
Warshel, A., Sussman, F. & Hwang, J.-K. Evaluation of catalytic free energies in genetically modified proteins. J. molec. Biol. 201, 139–159 (1988).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Schweins, T., Langen, R. & Warshel, A. Why have mutagenesis studies not located the general base in ras p21. Nat Struct Mol Biol 1, 476–484 (1994). https://doi.org/10.1038/nsb0794-476
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nsb0794-476
This article is cited by
-
GTPases of the Translation Apparatus
Molecular Biology (2005)
-
How do the GTPases really work?
Nature Structural Biology (1995)
-
Substrate-assisted catalysis as a mechanism for GTP hydrolysis of p21ras and other GTP-binding proteins
Nature Structural Biology (1995)
-
GTPase mechanism of Gproteins from the 1.7-Å crystal structure of transducin α - GDP AIF−4
Nature (1994)
-
How G proteins turn off
Nature (1994)