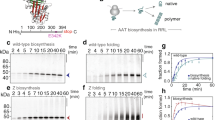
Abstract
Emphysema is often associated with the Z type mutation of α1,-antitrypsin, which causes aggregation of the molecule in the liver and consequent plasma deficiency. The aggregation appears to be due to loop-sheet polymerization, although why the mutant protein polymerizes in vivo is unclear. Here we show that, unlike wild type antitrypsin, which folds in minutes, the folding of Z type α1,-antitrypsin is extremely slow. Once folded, however, the native Z protein shows substantial stability towards urea and incubation at 37 °C. The folding defect in Z antitrypsin leads to accumulation of an intermediate and it is the intermediate rather than the native protein which has a high tendency to aggregate.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Huber, R. & Carrell, R.W. Implications of the three-dimensional structure of α1-antitrypsin for structure and function of serpins. Biochemistry 28, 8951–8966 (1989).
Loebermann, H., Tokuoka, R., Deisenhofer, J. & Huber, R. Human α-proteinase inhibitor: crystal structure analysis of two crystal modifications, molecular model and preliminary analysis of the implications for function. J. molec. Biol. 177, 531–556(1984).
Crystal, R.G. The α1-antitrypsin gene and its deficiency states. Trends Genet. 5, 411–417 (1989).
Stein, P.E. & Carrell, R.W. What do dysfunctional serpins tell us about molecular mobility and disease?. Nature struct. Biol. 2, 96–113 (1995).
Lomas, D.A., Evans, D.L., Finch, J.T. & Carrell, R.W. The mechanism of Z α1-antitrypsin accumulation in the liver. Nature 357, 605–607 (1992).
Lomas, D.A., Evans, D.L., Stone, S.R., Chang, W.-S.W. & Carrell, R.W. Effect of the Z mutation on the physical and inhibitory properties of α1-antitrypsin. Biochemistry 32, 500–508.(1993).
Carrell, R.W., Evans, D.L. & Stein, P.E. Mobile reactive centre of serpins and the control of thrombosis. Nature 353, 576–578 (1991).
Carrell, R.W. & Evans, D.L.I. Serpins: mobile conformations in a family of proteinase inhibitors. Curr. Opin. struct. Biol. 2, 438–446 (1992).
Verbanac, K.M. & Heath, E.C. Biosynthesis, processing, and secretion of M and Z variant human α1-antitrypsin. J. biol. Chem. 261, 9979–9989 (1986).
Le, A., Ferrell, D.A., Dishon, D.S., Le, Q.-Q.A. & Sifers, R.N. Soluble aggregates of the human PiZ α1-antitrypsin variant are degraded within the endoplasmic reticulum by a mechanism sensitive to inhibitors of protein synthesis. J. biol. Chem. 267, 1072–1080 (1992).
Creighton, T.E. Electrophoretic analysis of the unfolding of proteins by urea. J. molec. Biol. 129, 235–264 (1979).
Creighton, T.E. Kinetic study of protein unfolding and refolding using urea gradient electrophoresis. J. molec. Biol. 137, 61–80 (1980).
Craig, S., Hollecker, M., Creighton, T.E. & Pain, R.H. Single amino acid mutations block a late step in the folding of β-lactamase from Staphylococcusaureus. J. molec. Biol. 185, 681–687 (1985).
Bathurst, I.C., Travis, J, George, P.M. & Carrell, R.W. Structural and functional characterization of the abnormal Zα1-antitrypsin isolated from human liver. FEBS Lett. 177, 179–183 (1984).
Ogushi,F, Fells, G.A., Hubbard, R.C., Straus, S.D., & Crystal, R.G. Z-type α1-antitrypsin is less competent than M1-type α1-antitrypsinas an inhibitor of neutrophil elastase. J. clin. Invest. 80, 1366–1374 (1987).
Carrell, R.W., Stein, P.E., Fermi, G. & Wardell, M.R. Biological implications of a 3 Å structure of dimeric antithrombin. Structure 2, 257–270 (1994).
Schreuder, H.A. et al. The intact and cleaved human antithrombin III complex as a model for serpin-protease interactions. Nature Struct. Biol. 1, 48–54 (1994).
Ou, W.-J., Cameron, P.H., Thomas, D.Y. & Bergeron, J.J.M. Association of folding intermediates of glycoproteins with calnexin during protein maturation. Nature 364, 771–776 (1993).
Le, A., Steiner, J.L., Ferrell, G.A., Shaker, J.C. & Sifers, R.N. Association between calnexin and a secretion-incompetent variant of human α1-antitrypsin. J. biol. Chem. 269, 7514–7519 (1994).
Hammond, C., Braakman, I. & Helenius, A. Role of N-linked oligosaccharide recognition, glucose trimming, and calnexin in glycoprotein folding and quality control. Proc. natn. Acad. Sci. U.S.A. 91, 913–917 (1994).
Wu, Y. et al A lag in intracellular degradation of mutant α1-antitrypsin correlates with the liver disease phenotype in homozygous PiZZ α1-antitrypsin deficiency. Proc natn. Acad. Sd. U.S.A. 91, 9014–9018 (1994).
Thomas, P.J., Ko, Y.H. & Pedersen, P.L. Altered protein folding may be the molecular basis of most cases of cystic fibrosis. FEBS Lett. 312, 7–9 (1992).
Welsh, M.J. & Smith, A.E. Molecular mechanisms of CFTR chloride channel dysfunction in cystic fibrosis. Cell 73, 1251–1254 (1993).
Thomas, P.J. & Pedersen, P.L. Effects of the ΔF508 mutation on the structure, function, and folding of the first nucleotide-binding domain of CFTR. J. Bioenergetics Biomembr. 25, 11–19 (1993).
Kocisko, D.A. et al. Cell-free formation of protease-resistant prion protein. Nature 370, 471–474 (1994).
Cohen, F.E. et al. Structural clues to prion replication. Science 264, 530–531 (1994).
Goldenberg, D.P. Analysis of protein conformation by gel electrophoresis. in Protein Structure: A practical Approach. (ed. Creighton, T.E.) 225–250 (IRL press, New York; 1989).
Kwon, K.-S., Kim, J., Shin, H.S. & Yu, M.-H. Single amino acid substitutions of α1-antitrypsin that confer enhancement in thermal stability. J. biol. Chem. 269, 9627–9631 (1994).
Kunkel, T. A. Rapid and efficient site-specific mutagenesis without phenotypic selection. Proc. natn. Acad. Sci. U.S.A. 82, 488–492 (1985).
Edelhoch, H. Spectroscopic determination of tryptophan and tyrosine in proteins. Biochemistry 6, 1948–1954 (1967).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Yu, MH., Lee, K. & Kim, J. The Z type variation of human α1-antitrypsin causes a protein folding defect. Nat Struct Mol Biol 2, 363–367 (1995). https://doi.org/10.1038/nsb0595-363
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nsb0595-363
This article is cited by
-
Nascent chains can form co-translational folding intermediates that promote post-translational folding outcomes in a disease-causing protein
Nature Communications (2021)
-
Artificial DnaJ Protein for protein production and conformational diseases
Scientific Reports (2017)
-
Alpha-1 Antitrypsin Deficiency: New Developments in Augmentation and Other Therapies
BioDrugs (2013)
-
Dynamic local unfolding in the serpin α-1 antitrypsin provides a mechanism for loop insertion and polymerization
Nature Structural & Molecular Biology (2011)
-
Molecular basis of α1‐antitrypsin deficiency revealed by the structure of a domain‐swapped trimer
EMBO reports (2011)