Abstract
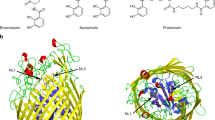
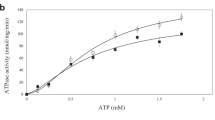
Siderophore binding proteins play a key role in the uptake of iron in many gram-positive and gram-negative bacteria. FhuD is a soluble periplasmic binding protein that transports ferrichrome and other hydroxamate siderophores. The crystal structure of FhuD from Escherichia coli in complex with the ferrichrome homolog gallichrome has been determined at 1.9 Å resolution, the first structure of a periplasmic binding protein involved in the uptake of siderophores. Gallichrome is held in a shallow pocket lined with aromatic groups; Arg and Tyr side chains interact directly with the hydroxamate moieties of the siderophore. FhuD possesses a novel fold, suggesting that its mechanisms of ligand binding and release are different from other structurally characterized periplasmic ligand binding proteins.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Accession codes
References
Weinberg, E.D. Perspect. Biol. Med. 40, 578–583 (1997).
Braun, V. & Killmann, H. Trends Biol. Sci. 24, 104–109 (1999).
Neilands, J.B. J. Biol. Chem. 270, 26723–26726 (1995).
Guerinot, M.-L. Annu. Rev. Microbiol. 48, 743–772 (1994).
Mietzner, T.A. et al. Curr. Topics Microbiol. Immun. 225, 113–135 (1998).
Köster, W. Biol. Metals 4, 23–32 (1991).
Pierce, J.R. & Earhart, C.F. J. Bacteriol. 166, 930–936 (1986).
Ozenberger, B.A., Nahlik, M.S. & McIntosh, M.A. J. Bacteriol. 169, 3638–3646 (1987).
Shea, C.M. & McIntosh, M.A. Mol. Microbiol. 5, 1415–1428 (1991).
Staudenmaier, H. et al. J. Bacteriol . 171, 2626–2633 (1989).
Braun, V., Gunther, K., Hantke, K. & Zimmermann, L. J. Bacteriol. 156, 308–315 (1983).
Miller, M.J. & Malouin, F. Acc. Chem. Res. 26, 241–249 (1993).
Ghosh, A. & Miller, M. J. Bioorg. Med. Chem. 4, 43–48 (1996).
Ferguson, A.D. et al. Science 282, 2215–2220 (1998).
Locher, K.P. et al. Cell 95, 771–778 (1998).
Buchanan, S.K. et al. Nature Struct. Biol. 6, 56–63 (1999).
Van der Helm, D. et al. Acta Crystallogr. B 37, 323–330 (1981).
Llinás, M. & Neilands, J.B. Biophys. Struct. Mech . 2, 105–117 (1976).
Llinás, M., Wilson, D.M. & Klein, M.P. J. Am. Chem. Soc. 99, 6846–6850 (1977).
Llinás, M. & Wüthrich, K. Biochim. Biophys. Acta. 532, 29–40 (1978).
DeMarco, A. & Llinás, M. Biochemistry 18, 3846–3854 (1979).
Aramini, J., McIntyre, D.D. & Vogel, H.J. J. Am. Chem. Soc. 116, 11506–11511 (1994).
Quiocho, F.A. & Ledvina, P.S. Mol. Microbiol. 20, 17–25 (1996).
Holm, L. & Sander, C. J. Mol. Biol. 233, 123–138 (1993).
Holm, L. & Sander, C. Science 273, 595–602 (1996).
Lee, Y.-H. et al. Nature Struct. Biology 6, 628–633 (1999).
Kim, J. & Rees, D.C. Nature 360, 553–560 (1992).
Lawrence, M.C. et al. Structure 6, 1553–1561 (1999).
Tam, R. & Saier, M.H. Microbiol. Rev. 57, 320–346 (1993).
Rohrbach, M.R., Braun, V. & Köster, W. J. Bacteriol. 177, 7186–7193 (1995).
Gerstein, M., Lesk, A.M. & Chothia, C. Biochemistry 33, 6739–6749 (1994).
Rohrbach, M.R., Paul, S. & Köster, W. Mol. Gen. Genet. 248, 33–42 (1995).
Otwinowski, Z. & Minor, W. Methods Enzymol. 276, 307–326 (1997).
Brünger, A.T. et al. Acta Crystallogr. 54, 905–921 (1998).
Roussel, A. & Cambillau, C. TURBO-FRODO. In Silicon Graphics Geometry Partner Directory, 77–88 (Silicon Graphics, Mountain View, California; 1989).
Evans, S.V. J. Mol. Graphics 11, 134–138 (1993).
Nicholls, A., Sharp, K. & Honig, B. GRASP Manual (Columbia University, New York, New York; 1992).
Acknowledgements
We would like to thank V. Braun (Universität Tübingen, Germany) and W. Köster (Zurich, Switzerland) for providing strains used in this study. We would also like to thank L. Howell and J. Berensden for generously providing access to beam time and L. Flaks at the X8C beamline at BNL for assistance with data collection. This work was supported by operating grants from the Alberta Heritage Foundation for Medical Research (AHFMR) and the University of Calgary to L.W.T. and a grant from the Medical Research Council of Canada to H.J.V. T.E.C. is a holder of an MRC Doctoral Research Award. S.-Y.K. was supported by a Natural Sciences and Engineering Research Council of Canada summer studentship. L.W.T. and H.J.V. hold Medical Scholar and Scientist Awards, respectively, from AHFMR.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Clarke, T., Ku, SY., Dougan, D. et al. The structure of the ferric siderophore binding protein FhuD complexed with gallichrome. Nat Struct Mol Biol 7, 287–291 (2000). https://doi.org/10.1038/74048
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/74048
This article is cited by
-
Cryo-EM reveals unique structural features of the FhuCDB Escherichia coli ferrichrome importer
Communications Biology (2021)
-
Structure and dynamics of Type III periplasmic proteins VcFhuD and VcHutB reveal molecular basis of their distinctive ligand binding properties
Scientific Reports (2017)
-
Conformational Change of a Tryptophan Residue in BtuF Facilitates Binding and Transport of Cobinamide by the Vitamin B12 Transporter BtuCD-F
Scientific Reports (2017)
-
Fate of ferrisiderophores after import across bacterial outer membranes: different iron release strategies are observed in the cytoplasm or periplasm depending on the siderophore pathways
Amino Acids (2013)
-
Siderophore uptake in bacteria and the battle for iron with the host; a bird’s eye view
BioMetals (2010)