Abstract
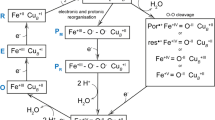
Folding of cytochrome c from its low pH guanidine hydrochloride (Gdn-HCl) denatured state revealed a new intermediate, a five-coordinate high spin species with a water molecule coordinated to the heme. Incorporation of this five-coordinated intermediate into the previously reported ligand exchange model can quantitatively account for the observed folding kinetics. In this new model, unfolded cytochrome c is converted to its native structure through an obligatory folding intermediate, the histidine-water coordination state, whereas the five-coordinate state and a bis-histidine state are off-pathway intermediates. When the concentration of Gdn-HCl in the refolding solution was increased, an acceleration of the conversion from the bis-histidine coordinated state to the histidine-water coordinated state was observed, demonstrating that the reaction requires unfolding of the mis-organized polypeptide structure associated with the bis-histidine state.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Kim, P.S. & Baldwin, R.L. Intermediates in the folding reactions of small molecules. Annu. Rev. Biochem. 59, 631–660 (1990).
Chan, H.S. & Dill, K.A. The protein folding problem. Phys. Today February 24–32 (1993).
Dill, K.A. & Chan, H.S. From Levinthal to pathways to funnels. Nature Struct. Biol. 4, 10–19 (1997).
Jones, C.M. et al. Fast events in protein folding initiated by nanosecond laser photolysis. Proc. Nat. Acad. Sci. USA 90, 11860–11864 (1993).
Phillips, C.M., Mizutani, Y. & Hochstrasser, R.M. Ultrafast thermally induced unfolding of Rnase A. Proc. Nat. Acad. Sci. USA 92, 7292–7296 (1995).
Nolting, B., Golbik, P. & Fersht, A.R. Submillisecond events in protein folding. Proc. Nat. Acad. Sci. USA 92, 10668–10672 (1995).
Williams, S. et al. Fast events in protein folding: helix melting and formation in a small peptide. Biochemistry 35, 691–697 (1995).
Ballew, R.M., Sabelko, J. & Gruebele, M. Direct observation of fast protein folding: the initial collapse of apomyoglobin. Proc. Natl. Acad. Sci. USA 93, 5759–5764 (1996).
Pascher, T., Chesick, J.P., Winkler, J.R. & Gray, H.B. Protein folding triggered by electron transfer. Science 271, 1558–1560 (1996).
Takahashi, S. et al. Folding of cytochrome c initiated by submillisecond mixing. Nature Struct. Biol. 4, 44–50 (1997).
Yeh, S.-R., Takahashi, S., Fan, B. & Rousseau, D.L. Ligand exchange during cytochrome c folding. Nature Struct. Biol. 4, 51–56 (1997).
Chan, C.-K. et al. Submillisecond protein folding kinetics studied by ultrarapid mixing. Proc. Natl. Acad. Sci. USA 94, 1779–1784 (1997).
Evans, P.A. & Radford, S.E. Probing the Structure of Folding Intermediates. Curr. Opin. Struct. Biol. 4, 100–106 (1994).
Englander, S.W. & Mayne, L. Protein Folding Studies Using Hydrogen Exchange Labeling and Two-Dimensional NMR. Annu. Rev. Biophys. Biomol. Struct. 21, 243–265 (1992).
and Protein Folding. Curr. Opin. Struct. Biol. 4, 112–116 (1994).
Johnson, W.C., Jr. Structure of Proteins Through Circular Dichroism Spectroscopy. Annu. Rev. Biophys. Biophys. Chem. 17, 145–166 (1988).
Elove, G.A., Chaffotte, A.F., Roder, H. & Goldgerg, M.E. Early Steps in the Cytochrome c Folding Probed by Time Resolved Circular Dichroism and Fluorescence Spectrscopy. Biochemistry 31, 6876–6883 (1992).
Elove, G.A., Bhuyan, A.K. & Roder, H. Kinetic mechanism of cytochrome c folding: involvement of the heme and its ligands. Biochemistry 33, 6925–6935 (1994).
Sosnick, T.R., Mayne, L., Hiller, R. & Englander, S.W. The barriers in protein folding. Nature Struct. Biol. 1, 149–156 (1994).
Pierce, M.M. & Nall, B.T. Fast folding of cytochrome c. Prot. Sci. 6, 618–627 (1997).
Hu, S., Morris, I.K., Singh, J.P., Smith, K.M. & Spiro, T.G. Complete assignment of cytochrome c resonance Raman spectra through enzymatic reconstitution with isotopically labeled hemes. J. Am. Chem. Soc. 115, 12446–12458 (1993).
Jordan, T., Eads, J.C. & Spiro, T.G. Secondary and tertiary structure of the A-state of cytochrome c from resonance Raman spectroscopy. Prot. Sci. 4, 716–728 (1995).
Wang, J.-S., Tsai, A.-L., Held, J., Palmer, G. & Van Wart, H.E. Temperature- and pH-dependent changes in the coordination sphere of the heme c group in the model peroxidase Nα-acetyl microperoxidase-8. J. Biol. Chem. 267, 15310–15318 (1992).
Sosnick, T.R., Mayne, L. & Englander, S.W. Molecular collapse: the rate-limiting step in two-state cytochrome c folding. Proteins Struct. Func. Genet. 24, 413–426 (1996).
Myer, Y.P. & Saturno, A.F. Horse heart ferricytochrome c: conformation and heme configuration of low ionic strength acidic forms. J. Prot. Chem. 9, 379–387 (1990).
Adams, P.A., Baldwin, D.A. & Marques, H.M. The hemepeptides from cytochrome c: preparation, physical and chemical properties, and their use as model compounds for the hemoproteins. In Cytochrome c A Multidisciplinary Approach (eds Scott, R. A. & Mauk, A. G.) 635–692 (University Science Books, Sausilito; 1996).
Bushnell, G.W., Louie, G.V. & Brayer, G.D. High resolution three dimensional structure of horse heart cytochrome c. J. Mol. Biol. 214, 585–595 (1990).
Levinthal, C. Are there pathways for protein folding? J. Chim. Phys. 65, 44–45 (1968).
Eaton, E.A., Munoz, V., Thompson, P.A., Chan, C.-K. & Hofrichter, J. Submillisecond kinetics of protein folding. Curr. Opin. Struct. Biol. 7, 10–14 (1997).
Roder, H. & Colon, W. Kinetic role of early intermediates in protein folding. Curr. Opin. Struct. Biol. 7, 15–28 (1997).
Sosnick, T.R., Shtilerman, M.D., Mayne, L. & Englander, S.W. Ultrafast signals in protein folding and the polypeptide contracted state. Proc. Natl. Acad. Sci. USA 94, 8545–8550 (1997).
Takahashi, S., Ching, Y.-c., Wang, J. & Rousseau, D.L. Microsecond generation of oxygen-bound cytochrome c oxidase by rapid solution mixing. J. Biol. Chem. 270, 8405–8407 (1995).
Colon, W., Elove, G.A., Wakem, L.P., Sherman, F. & Roder, H. Side chain packing of the N- and C-terminal helices plays a critical role in the kinetics of cytochrome c folding. Biochemistry 35, 5538–5549 (1996).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yeh, SR., Rousseau, D. Folding intermediates in cytochrome c. Nat Struct Mol Biol 5, 222–228 (1998). https://doi.org/10.1038/nsb0398-222
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nsb0398-222
This article is cited by
-
Expression and Characterization of Recombinant Sucrose Phosphorylase
The Protein Journal (2018)
-
The role of key residues in structure, function, and stability of cytochrome-c
Cellular and Molecular Life Sciences (2014)
-
The impact of urea-induced unfolding on the redox process of immobilised cytochrome c
JBIC Journal of Biological Inorganic Chemistry (2010)
-
Redox thermodynamics of cytochromes c subjected to urea induced unfolding
Journal of Applied Electrochemistry (2009)
-
Characterization of N-terminal amino group–heme ligation emerging upon guanidine hydrochloric acid induced unfolding of Hydrogenobacter thermophilus ferricytochrome c 552
JBIC Journal of Biological Inorganic Chemistry (2007)