Abstract
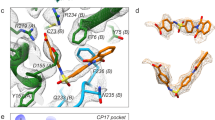
We have determined the crystal structure of a bovine enterovirus, revealing that the topologies of the major capsid proteins and the overall architecture of the virion are similar to those of related picornaviruses. The external loops joining β-strands are truncated and the canyon region is partially filled by an extension of the VP3 G–H loop giving the viral capsid a relatively smooth appearance. These changes may have implications for cell attachment. In spite of these differences the virus maintains a hydrophobic pocket within VP1, occupied by a specific ‘pocket factor’ which appears to be myristic acid. These observations support the proposal that a kinetic equilibrium exists between occupied and unoccupied pocket states, with occupation inhibiting uncoating.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Palmenberg, A.C. & Ehrenfeld, E. (ed.Semler,B.A.) 211–241 (American Society for Microbiology, Washington D.C; 1989).
Rossmann, M.G. et al. Structure of a human common cold virus and functional relationship to other picornaviruses. Nature 317, 145–153 (1985).
Hogle, J.M., Chow, M. & Filman, D.J. Three-dimensional structure of poliovirus at 2.9 Å resolution. Science 229, 1358–1365 (1985).
Olson, N.H. et al. Structure of a human rhinovirus complexed with its receptor molecule. Proc. natn. Acad. Sci. U.S.A. 90, 507–511 (1993).
Fillman, D.J. et al. Structural factors that control conformational transitions and serotype specificity in type 3 poliovirus. EMBO J. 8, 1567–1579 (1989).
Oliveira, M.A. et al. The structure of human rhinovirus 16. Structure 1, 51–68 (1993).
Moll, T. & Davis, A.D. Isolation and characterisation of cytopathogenic enteroviruses from cattle with respiratory disease.Am. J. vet. Sci. 20, 27–32 (1959).
Martin, S.J., Johnson, M.D. & Clements, J.B. Purification and characterization of bovine enterovirus. J. gen. Virol. 7, 103–113 (1970).
Kalter, S.S. Animal orphan enteroviruses. Bull. W. H. O. 22, 319–337 (1960).
Acharya, R. et al. The three-dimensional structure of foot-and-mouth disease virus at 2.9 Å resolution. Nature 337, 709–716 (1989).
Smyth, M., Hoey, E.M., Trudgett, A., Martin, S.J. & Brown, F. Chemically synthesised peptides elicit neutralizing antibody to bovine enterovirus. J. gen. Virol. 71, 231–234 (1990).
Sherry, B., Mosser, A.G., Colonno, R.J. & Rueckert, R.R. Use of monoclonal antibodies to identify four neutralisation epiptopes on a common cold picornavirus, human rhinovirus 14. J. Virol. 57, 246–257 (1986).
Minor, P.D., Ferguson, M., Evans, D.M.A., Almond, J.W. & Icenogle, J.P. Antigenic structure of poliovirus serotypes 1, 2 and 3. J. gen. Virol. 67, 1283–1291 (1986).
Arnold, E. & Rossmann, M.G. The use of molecular replacement phases for the refinement of the human rhinovirus 14 structure. Acta crystallogr. A44, 270–282 (1988).
Kim, S. et al. Crystal structure of human rhinovirus serotype 1A (HRV1A). J. molec. Biol. 210, 91–111 (1989).
Chow, M. et al. Myristylation of picornavirus capsid protein VP4 and its structural significance. Nature 327, 482–486 (1987).
Lea, S. et al. The structure and antigenicity of a type C foot-and-mouth disease virus. Structure 2, 123–139 (1994).
Colonno, R.J. et al. Evidence for the direct involvement of the rhinovirus canyon in receptor binding. Proc. natn. Acad. Sci. U.S.A. 85, 5449–5453 (1988).
Kim, S. et al. Conformational variability of a picornavirus capsid: pH-dependent structural changes of mengo virus related to its host receptor attachment site and disassembly. Virology 175, 176–190 (1990).
Logan, D. et al. Structure of a major immunogenic site on foot-and-mouth disease virus. Nature 362, 566–568 (1993).
Smith, T.J. et al. The site of attachment in human rhinovirus 14 for antiviral agents that inhibit uncoating. Science 233, 1286–1293 (1986).
Diana, G.D. et al. Anti-viral activity of beta-diketones. 1. Aryl alkyl diketones. In vitro activiy against both RNA and DMA viruses. J. med. Chem. 20, 750–756 (1977).
Grant, R.A. et al. Structures of poliovirus complexes with antiviral drugs: implications for viral stability and drug design. Curr. Biol. 4, 784–797 (1994).
Ismail-Cassim, N., Chezzi, C. & Newman, J.F.E. Inhibition of the uncoating of bovine enterovirus by short chain fatty acids. J. gen. Virol. 71, 2283–2289 (1990).
Dorval, B.L., Chow, M. & Klibanov, A.M. Stabilization of poliovirus against heat inactivation. Biochem. biophys. res. commun. 159, 1177–1183 (1989).
Fry, E. et al. in Protein Engineering (ed. Goodenough, P.) (CPL Press. 1992).
Symth, M. et al. Preliminary crystallographic analysis of bovine enterovirus. J. molec. Biol. 231, 930–932 (1993).
Arnold, E. et al. Structure determination of a common cold virus, human rhinovirus 14. Acta crystallogr. A43, 346–361 (1987).
Brunger, A.T. X-PLOR Version 3.0. (Yale University, New Haven, C.T. 1992).
Jones, T.A. Interactive computer graphics: FRODO. Meths Enzymol. 115, 157–171 (1985).
Brunger, A.T., Krutowski, A. & Erickson, J. Slow cooling protocols for crystallographic refinement by simulated annealing. Acta crystallogr. A46, 585–593 (1990).
Stuart, D.I., Levine, M., Muirhead, H. & Stammers, D.K. The crystal structure of cat private kinase at a resolution of 2.6 Å. J. molec Biol. 134, 109–142 (1979).
Jones, T.A., Zou, Y.-J., Cowan, S.W. & Kjeldgaard, M. Improved methods for building protein models in electron density maps and the location of errors in these models. Acta crystallogr. A47, 110–119 (1991).
Kraulis, P.J. MOLSCRIPT: a program to produce both detailed and schematic plots of protein structures. J. app. Crystallogr. 24, 946–950 (1991).
Merritt, E.A. & Murphy, M.E.P. Raster 3D version 2.0. A program for photorealistic molecular graphics. Acta crystallogr. D50, 869–873 (1994).
Kabsch, W. & Sander, C. Dictionary of protein secondary structure: pattern recognition of hydrogen-bonded and geometrical features. Biopolymers 22, 2577–2637 (1983).
Kleywegt, G.J. & Jones, T.A. Detection, delineation, measurement and display of cavities in macromolecular structures. Acta crystallogr. D50, 178–185 (1994).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Smyth, M., Tate, J., Hoey, E. et al. Implications for viral uncoating from the structure of bovine enterovirus. Nat Struct Mol Biol 2, 224–231 (1995). https://doi.org/10.1038/nsb0395-224
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nsb0395-224
This article is cited by
-
High-speed fixed-target serial virus crystallography
Nature Methods (2017)
-
Multiple capsid-stabilizing interactions revealed in a high-resolution structure of an emerging picornavirus causing neonatal sepsis
Nature Communications (2016)
-
A sensor-adaptor mechanism for enterovirus uncoating from structures of EV71
Nature Structural & Molecular Biology (2012)
-
A 3D framework for understanding enterovirus 71
Nature Structural & Molecular Biology (2012)
-
Characterizing sequence variation in the VP1 capsid proteins of foot and mouth disease virus (serotype 0) with respect to virion structure
Journal of Molecular Evolution (1998)