Abstract
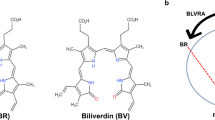
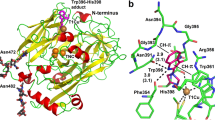
Biliverdin IXβ reductase (BVR-B) catalyzes the pyridine nucleotide-dependent production of bilirubin-IXβ, the major heme catabolite during early fetal development. BVR-B displays a preference for biliverdin isomers without propionates straddling the C10 position, in contrast to biliverdin IXα reductase (BVR-A), the major form of BVR in adult human liver. In addition to its tetrapyrrole clearance role in the fetus, BVR-B has flavin and ferric reductase activities in the adult. We have solved the structure of human BVR-B in complex with NADP+ at 1.15 Å resolution. Human BVR-B is a monomer displaying an α/β dinucleotide binding fold. The structures of ternary complexes with mesobiliverdin IVα, biliverdin IXα, FMN and lumichrome show that human BVR-B has a single substrate binding site, to which substrates and inhibitors bind primarily through hydrophobic interactions, explaining its broad specificity. The reducible atom of both biliverdin and flavin substrates lies above the reactive C4 of the cofactor, an appropriate position for direct hydride transfer. BVR-B discriminates against the biliverdin IXα isomer through steric hindrance at the bilatriene side chain binding pockets. The structure also explains the enzyme's preference for NADP(H) and its B-face stereospecificity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others

References
Blumenthal, S.G. et al. Biochem. J. 186, 693–700 (1980).
Yamaguchi, T. & Nakajima, H. Eur. J. Biochem. 233, 467–472 (1995).
Yamaguchi, T., Komoda, Y. & Nakajima, H. J. Biol. Chem. 269, 24343–24348 (1994).
Shalloe, F., Elliott, G., Ennis, O. & Mantle, T.J. Biochem. J. 316, 385–387 (1996).
Mack, C.P., Hultquist, D.E. & Shlafer, M. Biochem. Biophys. Res. Commun. 212, 35–40 (1995).
Cunningham, O., Dunne, A., Sabido, P., Lightner, D. & Mantle, T.J. J. Biol. Chem. 275, 19009–19017 (2000).
Cunningham, O., Gore, M.G. & Mantle, T.J. Biochem. J. 345, 393–399 (2000).
Xu, F. et al. Biochem. Biophys. Res. Commun. 193, 434–439 (1993).
Thoden, J.B., Frey, P.A. & Holden, H.M. Biochemistry 35, 5137–5144 (1996).
Woods, C.M., Zhu, J., Coleman, T., Bloom, S.E. & Lazarides, E. J. Cell Sci. 108, 699–710 (1995).
Zhu, J., Bloom, S.E., Lazarides, E. & Woods, C. J. Cell Sci. 108, 685–698 (1995).
Levy, H.R., Vought, V.E., Yin, X. & Adams, M.J. Arch. Biochem. Biophys. 326, 145–151 (1996).
Didierjean, C. et al. J. Mol. Biol. 268, 739–759 (1997).
Adams, M.J., Ellis, G.H., Gover, S., Naylor, C.E. & Phillips, C. Structure 2, 651–668 (1994).
Deacon, A.M., Ni, Y.S., Coleman, W.G. Jr. & Ealick, S.E. Structure 8, 453–462 (2000).
Yaoi, T., Miyazaki, K. & Oshima, T. FEBS Lett. 355, 171–172 (1994).
Chen, R., Greer, A. & Dean, A.M. Proc. Natl. Acad. Sci. USA 92, 11666–11670 (1995).
Frydman, R.B., Bari, S., Tomaro, M.L. & Frydman, B. Biochem. Biophys. Res. Commun. 171, 465–473 (1990).
Sun, D. et al. Acta Crystallogr. D 56, 1180–1182 (2000).
Cunningham, O. PhD thesis, University of Dublin (1998).
Leslie, A. In Crystallographic computing V (eds., Moras, D., Podjarny, A.D. & Thierry, J.C.) 27–38 (Oxford University Press, Oxford, UK; 1991).
Collaborative Computational Project No. 4. Acta Crystallogr. D 50, 760–763 (1994).
de La Fortelle, E. & Bricogne, G. Methods Enzymol. 276, 472–494 (1997).
Brünger, A.T. et al. Acta Crystallogr. D 54, 905–921 (1998).
Roussel, A. & Cambillau, C. TurboFRODO in Sillicon Graphics geometry (Sillicon Graphics, Mountain View, California; 1989).
Sheldrick, G.M. & Schneider, T.R. Methods Enzymol. 277, 319–343 (1997).
Ennis, O., Maytum, R. & Mantle, T.J. Biochem. J. 328, 33–36 (1997).
Yamaguchi, T., Komuro, A., Nakano, Y., Tomita, M. & Nakajima, H. Biochem. Biophys. Res. Commun. 197, 1518–1523 (1993).
Quandt, K.S. & Hultquist, D.E. Proc. Natl. Acad. Sci. USA 91, 9322–9326 (1994).
Acknowledgements
This work was supported by grants from the Ministerio de Educación y Cultura and the Generalitat de Catalunya to M.C. P.J.B.P. was supported in part by a FEBS Long Term fellowship. P.J.B.P. and S.M.R. acknowledge postdoctoral fellowships from Programa Praxis XXI (FCT, Portugal). O.C. was supported by the Health Research Board, Ireland. We thank D.A. Lightner for supplying the various verdins used in this work. Synchrotron data collection was supported by EU grants and the ESRF.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pereira, P., Macedo-Ribeiro, S., Párraga, A. et al. Structure of human biliverdin IXβ reductase, an early fetal bilirubin IXβ producing enzyme. Nat Struct Mol Biol 8, 215–220 (2001). https://doi.org/10.1038/84948
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/84948
This article is cited by
-
Cloning and characterization of enoate reductase with high β-ionone to dihydro-β-ionone bioconversion productivity
BMC Biotechnology (2018)
-
dissectHMMER: a HMMER-based score dissection framework that statistically evaluates fold-critical sequence segments for domain fold similarity
Biology Direct (2015)
-
Medium- and short-chain dehydrogenase/reductase gene and protein families
Cellular and Molecular Life Sciences (2008)
-
Phototherapy: From Ancient Egypt to the New Millennium
Journal of Perinatology (2001)