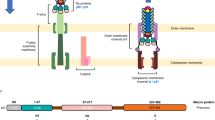
Abstract
The structure of the two N-terminal domains of the gene 3 protein of filamentous phages (residues 1–217) has been solved by multiwavelength anomalous diffraction and refined at 1.46 A resolution. Each domain consists of either five or eight β-strands and a single α-helix. Despite missing sequence homology, their cores superimposed with a root-mean-square deviation of 2 ?. The domains are engaged in extensive interactions, resulting in a horseshoe shape with aliphatic amino acids and threonines lining the inside, delineating the likely binding site for the F-pilus. The glycine-rich linker connecting the domains is invisible in the otherwise highly ordered structure and may confer flexibility between the domains required during the infection process.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Smith, G.P. Filamentous fusion phage: novel expression vectors that display cloned antigens on the virion surface. Science 228, 1315–1317 (1985).
Phizicky, E.M. & Fields, S. Protein–protein interactions: methods for detection and analysis. Microbiol. Rev. 59, 94–123 (1995).
Cortese, R. et al Selection of biologically active peptides by phage display of random peptide libraries. Curr. Opin. Biotechnol. 7, 616–621 (1996).
Dunn, I.S. Phage display of proteins. Curr. Opin. Biotechnol. 7, 547–553 (1996).
Spada, S., Krebber, C. & Plückthun, A. Selectively infective phages (SIP). Biol. Chem. 378, 445–456 (1997).
Gray, C.W., Brown, R.S. & Marvin, D.A. Adsorption complex of filamentous fd virus. J. Mol. Biol. 146, 621–627 (1981).
Jacobson, A. Role of F pili in the penetration of bacteriophage fl. J. Virol. 10, 835–843 (1972).
Sun, T.P. & Webster, R.E. fii, a bacterial locus required for filamentous phage infection and its relation to colicin-tolerant tolA and tolB. J. Bacterial. 165, 107–115 (1986).
Sun, T.P. & Webster, R.E. Nucleotide sequence of a gene cluster involved in entry of E colicins and single-stranded DNA of infecting filamentous bacteriophages into Escherichia coli. J. Bacteriol. 169, 2667–2674 (1987).
Riechmann, L. & Holliger, P. The C-terminal domain of TolA is the coreceptorfor filamentous phage infection of E. coli. Cell 90, 351–360 (1997).
Nelson, F.K., Friedman, S.M. & Smith, G.P. Filamentous phage DNA cloning vectors: a noninfective mutant with a nonpolar deletion in gene III. Virology 108, 338–350 (1981).
Crissman, J.W. & Smith, G.P. Gene-Ill protein of filamentous phages: evidence for a carboxyl-terminal domain with a role in morphogenesis. Virology 132, 445–455 (1984).
Endemann, H., Gailus, V. & Rasched, I. Interchangeability of the adsorption proteins of bacteriophages Ff and IKe. J. Virol. 67, 3332–3337 (1993).
Hutchinson, E.G. & Thornton, J.M. PROMOTIF - a program to identify and analyze structural motifs in proteins. Protein Sci. 5, 212–220 (1996).
Holm, L. & Sander, C Protein structure comparison by alignment of distance matrices. J. Mol. Biol. 233, 123–138 (1993).
Cabral, J.H, et al. Crystal structure of a PDZ domain. Nature 382, 649–652 (1996).
Faber, H.R. et al. 1.8 ? crystal structure of the C-terminal domain of rabbit serum haemopexin. Structure 3, 551–559 (1995).
Viguera, A.R., Blanco, F.J. & Serrano, L. The order of secondary structure elements does not determine the structure of a protein but does affect its folding kinetics. J. Mol. Biol. 247, 670–681 (1995).
Holliger, P. & Riechmann, L. A conserved infection pathway for filamentous bacteriophages is suggested by the structure of the membrane penetration domain of the minor coat protein g3p from phage Fd. Structure 5, 265–275 (1997).
Koradi, R., Billeter, M. & Wüthrich, K. MOLMOL: a program for display and analysis of macromolecular structures. J. Mol. Graph. 14, 51–5, 29–32 (1996).
Baldwin, E.T. et al. Crystal structure of interleukin-8: symbiosis of NMR and crystallography. Proc. Natl. Acad. Sci. USA 88, 502–506 (1991).
Lubkowski, J. et al. The structure of MCP-1 in two crystal forms provides a rare example of variable quarternary interactions. Nature Struct. Biol. 4, 64–69 (1997).
Stengele, I., Bross, P., Garces, X., Giray, J. & Rasched, I. Dissection of functional domains in phage fd adsorption protein. Discrimination between attachment and penetration sites. J. Mol. Biol. 212, 143–149 (1990).
Krebber, C. et al. Selectively-infective phage (SIP): a mechanistic dissection of a novel in vivo selection for protein-ligand interactions. J. Mol. Biol. 268, 607–618 (1997).
Peters, B.P.H., Peters, R.M., Schoenmakers, J.G.G. & Konings, R.N.H. Nucleotide sequence and genetic organizaton of the genome of the N-specific filamentous bacteriophage IKe. Comparison with the genome of the F-specific filamentous phages M13, fd and fl. J. Mol. Biol. 181, 27–39 (1985).
Frost, L.S., Ippen-lhler, K. & Skurray, R.A. Analysis of the sequence and gene products of the transfer region of the F sex factor. Microbiol. Rev. 58, 162–210 (1994).
Marvin, D.A. & Folkhard, W. Structure of F-pili: reassessment of the symmetry. J. Mol. Biol. 191, 299–300 (1986).
Glucksman, M.J., Bhattacharjee, S. & Makowski, L. Three-dimensional structure of a cloning vector. X-ray diffraction studies of filamentous bacteriophage M13 at 7? resolution. J. Mol. Biol. 226, 455–470 (1992).
Lin, T.C., Webster, R.E. & Konigsberg, W. Isolation and characterization of the C and D proteins coded by gene IX and gene VI in the filamentous bacteriophage fl and fd. J. Biol. Chem. 255, 10331–10337 (1980).
Grant, R.A., Lin, T.C., Webster, R.E. & Konigsberg, W. Structure of filamentous bacteriophage: isolation, characterization, and localization of the minor coat proteins and orientation of the DNA. Prog. Clin. Biol. Res. 64: 413–428 (1981).
Tzagoloff, H. & Pratt, D. The initial steps in infection with coliphage M13. Virology 24, 372–380 (1964).
Goldsmith, M.E. & Konigsberg, W.H. Adsorption protein of the bacteriophage fd: isolation, molecular properties, and location in the virus. Biochemistry 16, 2686–2694 (1977).
Beck, E. & Zink, B. Nucleotide sequence and genome organisation of filamentous bacteriophages fl and fd. Gene 16, 35–58 (1981).
Freund, C., Ross, A., Guth, B., Plückthun, A. & Holak, T.A. Characterization of the linker peptide of the single-chain Fv fragment of an antibody by NMR spectroscopy. FEBS Lett. 320, 97–100 (1993).
S tudier, F.W. & Moffatt, B.A. Use of bacteriophage T7 RNA polymerase to direct selective high-level expression of cloned genes. J. Mol. Biol. 189, 113–130 (1986).
Qoronfleh, M.W. et al. Production of selenomethionine-labeled recombinant human neutrophil collagenase in Escherichia coli. J. Biotechnol. 39, 119–128 (1995).
Plückthun, A., et al. Producing antibodies in Escherichia coli: from PCR to fermentation. In Antibody engineering: a practical approach (eds McCafferty, J. & Hoogenboom, H.R., Chriswell, D.J.) 203–252 (IRL Press, Oxford; 1996).
Hendrickson, W.A., Horton, J.R. & LeMaster, D.M. Selenomethionyl proteins produced for analysis by multiwavelength anomalous diffraction (MAD): a vehicle for direct determination of three dimensional structure. EMBO J. 9, 1665–1672 (1990).
Matthews, B.W. Solvent content of protein crystals. J. Mol. Biol. 33, 491–497 (1968).
Otwinowski, Z. An oscillation data processing suite for macromolecular crystallography (Yale University, New Haven, Connecticut, USA; 1992).
Jones, T.A. & Kjeldgaard, M.O — the manual (Uppsala University, Uppsala, 1994).
Brünger, A. X-plor version 3.1: a system for X-ray crystallography and NMR (Yale University Press, New Haven, Connecticut, USA; 1992).
Furey, W. & Swaminathan, S. PHASES - A program package for the processing and analysis of diffraction data for macromolecules. Acta Crystallogr. 18, 73 (1990).
Sheldrick, G.M. Patterson superposition and ab initio phasing. Meth. Enz. 276, 628–641 (1997).
Wang, B.C. Resolution of phase ambiguity in macromolecular crystallography. Meth. Enz. 115, 90- 112 (1985).
CCP4:SERC Collaborative computing project no.4 (Warrington, UK; 1979).
Lundblad, R.L. & Noyes, C.M. Chemical modification of tryptophan. In Chemical reagents for protein modification (eds. Lundblad, R.L. & Noyes, C.M.) 47–71 (CRC Press, Boca Raton, Florida; 1984 ).
Kraulis, P.J. MOLSCRIPT: a program to produce both detailed and schematic plots of protein structures. J. Appl. Crystallogr. 24, 946–950 (1991).
Carson, M. RIBBONS 4.0. J. Appl.Crystallogr. 24, 958–961 (1991).
Laskowski, R.A., MacArthur, M.W., Moss, D.S. & Thornton, J.M. PROCHECK: A program to check the stereochemical quality of protein structures. J. Appl. Crystallogr. 26, 283–291 (1993).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lubkowski, J., Hennecke, F., Plückthun, A. et al. The structural basis of phage display elucidated by the crystal structure of the N-terminal domains of g3p. Nat Struct Mol Biol 5, 140–147 (1998). https://doi.org/10.1038/nsb0298-140
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsb0298-140
This article is cited by
-
Cryo-EM structure of a bacteriophage M13 mini variant
Nature Communications (2023)
-
M13 phage: a versatile building block for a highly specific analysis platform
Analytical and Bioanalytical Chemistry (2023)
-
M13 Bacteriophage-Based Bio-nano Systems for Bioapplication
BioChip Journal (2022)
-
Bee Venom Immunotherapy: Current Status and Future Directions
Clinical Reviews in Allergy & Immunology (2020)
-
Continuous directed evolution of aminoacyl-tRNA synthetases
Nature Chemical Biology (2017)