Abstract
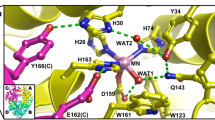
Human ferrochelatase (E.C. 4.99.1.1) is a homodimeric (86 kDa) mitochondrial membrane-associated enzyme that catalyzes the insertion of ferrous iron into protoporphyrin to form heme. We have determined the 2.0 Å structure from the single wavelength iron anomalous scattering signal. The enzyme contains two NO-sensitive and uniquely coordinated [2Fe-2S] clusters. Its membrane association is mediated in part by a 12-residue hydrophobic lip that also forms the entrance to the active site pocket. The positioning of highly conserved residues in the active site in conjunction with previous biochemical studies support a catalytic model that may have significance in explaining the enzymatic defects that lead to the human inherited disease erythropoietic protoporphyria.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Accession codes
References
Dailey, H.A. In Mechanism of metallocenter assembly (ed. Dailey, H.A.) 77–89 (VCH, New York; 1996).
Hansson, M. & Hederstedt, L. Eur J Biochem 220, 201–208 (1994).
Al-Karadaghi, S., Hansson, M., Nikonov, S., Jonsson, B. & Hederstedt, L. Structure 5, 1501–1510 (1997).
Dailey, H.A., Sellers, V.M. & Dailey, T.A. J. Biol. Chem. 269, 390–395 (1994).
Dailey, H.A. & Fleming, J.E. J. Biol. Chem. 258, 11453–11459 (1983).
Lavallee, D.K. In Mechanistic principles of enzyme activity (eds Liebman, J.F. & Greenberg, A.) 279–311 (VCH, New York; 1988).
Cochran, A.G. & Schultz, P.G. Science 249, 781–783 (1990).
Blackwood, M.E., Rush, T.S., Medlock, A., Dailey, H.A. & Spiro, T.G. J. Am. Chem. Soc. 119, 12170–12174 (1997).
Dailey, H.A. In Biosynthesis of heme and chlorophylls (ed. Dailey, H.A.) 123–161 (McGraw-Hill, New York; 1990).
Bonkowsky, H.L., Bloomer, J.R., Ebert, P.S. & Mahoney, M.J. J. Clin. Invest. 56, 1139–1148 (1975).
Nordmann, Y. & Deybach, J.C. In Biosynthesis of heme and chlorophylls (ed. Dailey, H.A.) 491–542 (McGraw-Hill, New York; 1990).
Crouse, B.R., Sellers, V.M., Finnegan, M.G., Dailey, H.A. & Johnson, M.K. Biochemistry 35, 16222–16229 (1996).
Sellers, V.M., Wang, K.F., Johnson, M.K. & Dailey, H.A. J. Biol. Chem. 273, 22311–22316 (1998).
Gora, M., Rytka, J. & Labbe-Bois, R. Arch. Biochem. Biophys. 361, 231–240 (1999).
Gora, M., Grzybowska, E., Rytka, J. & Labbe-Bois, R. J. Biol. Chem. 271, 11810–11816 (1996).
Lecerof, D., Fodje, M., Hansson, A., Hansson, M. & Al-Karadaghi, S. J. Mol. Biol. 297, 221–232 (2000).
Bain-Ackerman, M.J. & Laevellee, D.K. Inorg. Chem. 18, 3358–3364 (1979).
Burden, A.E. et al. Biochim. Biophys. Acta 1435, 191–197 (1999).
Otwinowski, Z. & Minor, W. Methods Enzymol. 276, 307–326 (1997).
Furey, W. & Swaminathan, S. Methods Enzymol. 277, 590–620 (1997).
Wang, B.C. Methods Enzymol. 115, 90–112 (1985).
Read, R.J. Acta Crystallogr. D 42, 140–149 (1986).
Jones, T.A., Zou, J.Y., Cowan, S.W. & Kjeldgaard. Acta Crystallogr A 47, 110–119 (1991).
Perrakis, A., Morris R. & Lamzin V.S., Nature Struct. Biol. 6, 458–463 (1999).
Brunger, A.T. et al. Acta Crystallogr D 54, 905–921 (1998).
Ramakrishnan, C. & Ramachandran, G.N. Biophys. J. 5, 909–933 (1965).
Berman, H.M. . et al. Nucleic Acids Res. 28, 235–242 (2000).
Kraulis, P.J. J. Appl. Crystallogr. 24, 946–950 (1991).
Barton, G.J. Methods Enzymol. 183, 403–428 (1990).
Barton, G.J. Protein Eng. 6, 37–40 (1993).
Nicholls A., Sharp K.A. & Honig B. Proteins 11, 281–296 (1991).
Acknowledgements
We thank J. Ferrara and Molecular Structure Corporation, The Woodlands, Texas, for collecting the R-AXIS data, and M.K. Johnson for discussions concerning the cluster. This work was supported in part by a grant from the NIH to H.A.D., and funds from University of Georgia Research Foundation, and the Georgia Research Alliance to B.-C.W.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wu, CK., Dailey, H., Rose, J. et al. The 2.0 Å structure of human ferrochelatase, the terminal enzyme of heme biosynthesis. Nat Struct Mol Biol 8, 156–160 (2001). https://doi.org/10.1038/84152
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/84152
This article is cited by
-
Harnessing large language models (LLMs) for candidate gene prioritization and selection
Journal of Translational Medicine (2023)
-
Heme biosynthesis depends on previously unrecognized acquisition of iron-sulfur cofactors in human amino-levulinic acid dehydratase
Nature Communications (2020)
-
The active site of magnesium chelatase
Nature Plants (2020)
-
Investigation by MD simulation of the key residues related to substrate-binding and heme-release in human ferrochelatase
Journal of Molecular Modeling (2013)
-
Is it possible for Fe2+ to approach protoporphyrin IX from the side of Tyr-13 in Bacillus subtilis ferrochelatase? An answer from QM/MM study
Journal of Molecular Modeling (2013)