Abstract
Over the past few years, MRI has become an indispensable tool for diagnosing multiple sclerosis (MS). However, the current MRI criteria for MS diagnosis have imperfect sensitivity and specificity. The central vein sign (CVS) has recently been proposed as a novel MRI biomarker to improve the accuracy and speed of MS diagnosis. Evidence indicates that the presence of the CVS in individual lesions can accurately differentiate MS from other diseases that mimic this condition. However, the predictive value of the CVS for the development of clinical MS in patients with suspected demyelinating disease is still unknown. Moreover, the lack of standardization for the definition and imaging of the CVS currently limits its clinical implementation and validation. On the basis of a thorough review of the existing literature on the CVS and the consensus opinion of the members of the North American Imaging in Multiple Sclerosis (NAIMS) Cooperative, this article provides statements and recommendations aimed at helping radiologists and neurologists to better understand, refine, standardize and evaluate the CVS in the diagnosis of MS.
Similar content being viewed by others
Main
At present, the diagnosis of multiple sclerosis (MS) relies heavily on the use of MRI, which can demonstrate disease dissemination in space and time1,2,3,4. The current 2010 McDonald criteria have enabled earlier diagnosis5,6 and initiation of disease-modifying treatment, with substantial benefits for disease outcome7,8, but they still have imperfect sensitivity and specificity9,10. The limited accuracy of the criteria results in challenging cases and misdiagnosis, which are prevalent problems in MS11,12. Therefore, more-accurate and pathologically specific MRI criteria are still needed to exclude other disorders that can mimic MS13,14.
The MRI-detectable central vein inside white matter lesions has recently been proposed as a biomarker of inflammatory demyelination and, thus, may aid the diagnosis of MS15. The 'central vein sign' (CVS) has been investigated in various neurological conditions by several groups, and evidence has accumulated that the CVS may have the ability to accurately differentiate MS from its mimics15,16,17,18,19,20,21. As a consequence, recent guidelines from the Magnetic Resonance Imaging in MS (MAGNIMS) group1,4 and the Consortium of MS Centers (CMSC) task force22 have acknowledged the potential of the CVS and its dedicated MRI acquisitions for the differential diagnosis of MS, while calling for further research before considering a possible modification of the diagnostic criteria. However, the lack of standardization for the definition and imaging of the CVS, as well as a dearth of large-scale prospective studies evaluating the CVS for MS diagnosis, are currently preventing the clinical validation of this potential biomarker1,23.
This Consensus Statement aims to provide recommendations for the definition, standardization and clinical evaluation of the CVS in the diagnosis of MS. These statements are based on a thorough review of the existing literature on the CVS and the consensus opinion of the members of the North American Imaging in Multiple Sclerosis (NAIMS) Cooperative (Box 1).
Methods
In November 2015, a panel of experts on the use of MRI in the management of MS convened at the University of Southern California, Los Angeles, USA. This meeting was organized by the NAIMS Cooperative — an independent network of clinical research groups that utilize MRI to better understand, diagnose and treat MS (Box 1). The panel was composed of neurologists, neuroradiologists, MRI scientists, and statisticians from different NAIMS-affiliated institutions in North America, as well as international experts on the topic of CVS.
During the meeting, recently published literature on the CVS in neurological diseases, and associated MRI techniques, was discussed. The following five topics were addressed in detail: the central vein in MS; the central vein in other neurological diseases; radiological definition of the central vein and the CVS; imaging of central veins with MRI; and clinical evaluation of the CVS for MS diagnosis. After open discussion and debate, the group reached a consensus on statements and recommendations on each of these five topics. After the meeting, a draft of the Consensus Statement was written by the first author on the basis of contributions from the panellists. This draft was then circulated to all NAIMS members, who modified the document until a final consensus agreement was reached.
The central vein in MS
Discussion
Central vessels (predominantly veins and venules) in MS plaques were reported by pathological studies as early as the 1820s24. The perivascular space surrounding these veins is thought to be a privileged site for immune cells to interact with antigen-presenting cells, which can then trigger an inflammatory cascade leading to the formation of lesions around the veins25,26. With the development of susceptibility-based magnetic resonance venography in the late 1990s27, it became possible to observe these central veins in MS plaques in vivo, as reported by Tan et al.28. This first in vivo demonstration of the perivenous distribution of MS plaques was further confirmed in 2008 using ultra-high-field MRI29,30. Follow-up imaging studies confirmed this finding in relapsing–remitting MS (RRMS), secondary progressive MS (SPMS) and primary progressive MS (PPMS)31,32. This perivenous distribution in different MS subtypes is illustrated in Fig. 1.
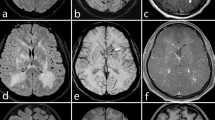
3 T FLAIR* (combined T2*-weighted MRI and fluid-attenuated inversion recovery) images from four individuals with a variety of neurological conditions, who were scanned at different sites. In the patients with relapsing–remitting or primary progressive multiple sclerosis (MS), a central vessel is visible in most hyperintense lesions (data from the NIH cohort). The dark veins are located centrally in the lesion and can be visualized in at least two perpendicular planes (arrows in magnified boxes). On the other hand, a central vein is absent from most of the lesions (arrowheads in magnified boxes) in the patient with migraine (University of Vermont cohort) and the patient with ischaemic small vessel disease (University of Nottingham cohort).
In an imaging study, Kilsdonk et al. examined 1,004 brain lesions in 33 patients with MS (19 with RRMS, nine with PPMS and five with SPMS), and found that 78% of the lesions were located around a central vessel31. The proportion of total lesions with a central vein was not related to the clinical phenotype — a finding that was also supported by another study32. However, when lesions were classified according to their location, the authors reported that central veins were most prevalent in periventricular lesions (94%). This finding was consistent across studies28,30, and might be explained by a higher density of parenchymal veins in periventricular regions. The proportion of CVS-positive lesions decreased with proximity to the neocortex (deep white matter lesions: 84%; juxtacortical lesions: 66%; mixed grey and white matter lesions: 52%; and intracortical lesions: 25%). However, a postmortem study has shown that the sites and characteristics of cortical lesions are strongly influenced by venous topography33 and, therefore, the association between cortical lesions and central veins should be further investigated with dedicated imaging techniques.
Although most studies imaged the supratentorial brain only, central veins have also been demonstrated in lesions located in the thalamus, cerebellum and pons of patients with MS31,34. To our knowledge, no in vivo reports are available on central veins in MS lesions located in the spinal cord, although pathological evidence of this phenomenon exists35. Another finding from the Kilsdonk et al. study was a significantly lower percentage of perivascular deep white matter lesions (73%) in MS patients aged ≥40 years compared with younger patients (92%). One possible explanation for this discrepancy is the presence of age-related vascular lesions without central veins. This finding, which remains to be confirmed, supports the contribution of comorbidities to the brain lesion load in patients with MS36,37. To date, no imaging studies have been performed on the venocentric distribution of brain lesions in paediatric MS.
Statements and recommendations
-
The presence of central veins inside MS lesions is a well-established finding in both ex vivo pathological studies and in vivo imaging studies
-
The venocentric distribution of lesions exists in all MS clinical phenotypes (RRMS, SPMS and PPMS)
-
When imaging is used to examine the proportion of MS lesions with a central vein, the location of the lesion should be taken into account. Current evidence suggests that the prevalence of central veins is highest in periventricular and deep white matter lesions
-
The proportion of MS lesions with a central vein in the cortical, infratentorial and spinal cord regions remains underinvestigated, and additional imaging studies in these areas are recommended
-
The effects of comorbidities (such as vascular conditions) on the proportion of lesions with a central vein in patients with MS should not be neglected. Additional imaging studies on this issue would be useful
-
The perivenous distribution of lesions in paediatric MS has yet not been demonstrated, and future imaging studies in this population are recommended
The central vein in other diseases
Discussion
Over the past few years, various research groups have used MRI to evaluate the presence of central veins inside white matter lesions associated with various neurological diseases, including neuromyelitis optica spectrum disorder (NMOSD), systemic autoimmune diseases (SAD), cerebral small vessel disease (CSVD), Susac syndrome, and migraine.
Neuromyelitis optica spectrum disorder. NMOSD is a CNS autoimmune disease that predominantly affects the optic nerves and spinal cord. NMOSD shares common radiological and clinical features with MS, and the differentiation between NMOSD and MS at early disease stages remains challenging. Sinnecker et al. reported that in ten patients with NMOSD who tested positive for aquaporin-4 autoantibodies (AQP4-IgG), only 35% of the 140 detected white matter lesions were located in the vicinity of — though rarely centred on — blood vessels19. In a different AQP4-IgG-seropositive NMOSD cohort (n = 10), Kister et al. reported that only eight of 92 lesions (9%) were traversed by a central vessel17, further supporting the idea that vein-sensitive MRI could prove useful for differentiating NMOSD from MS. To date, no equivalent imaging studies have been performed in AQP4-IgG-seronegative negative patients with NMOSD.
Systemic autoimmune diseases. White matter lesions are commonly detected in SAD, especially when patients present with neurological symptoms. A recent pilot study38 recruited 38 patients: 24 with MS, and 14 with SAD, including Behçet syndrome, systemic lupus erythematosus and antiphospholipid syndrome. The SAD group had a significantly lower percentage of lesions with central veins (median 15%, range 0–50%) than did the MS group (median 89%, range 68–100%). Patients with Behçet syndrome presented with the highest percentage of perivenous lesions (median 40%, range 16–50%).
Cerebral small vessel disease. CSVD refers to pathological changes in small brain vessels (small arteries, arterioles, capillaries and small veins) related to various aetiologies. CSVD is commonly associated with ageing, and is observed in populations with significant vascular risk factors. CSVD usually causes white matter lesions in the brain, which can mimic MS lesions. Although an early study by Lummel et al. reported no differences between patients with MS and CSVD in terms of the percentage of white matter lesions containing central veins39, multiple recent studies have consistently reported a significantly lower proportion (45% at most) of venocentric white matter lesions in CSVD15,16,18,40,41 — a finding that is illustrated by a representative example in Fig. 1.
Susac syndrome. Susac syndrome is believed to be an autoimmune vasculopathy that causes occlusion of small vessels in the brain, retina and inner ear. A study of five patients with this very rare disease found that a blood vessel was detectable in 54% of 148 white matter lesions21. Interestingly, the identified blood vessels were most commonly located at the lesion periphery.
Migraine. Radiographic findings in migraine can be mistaken for MS. A recent study found that the percentage of lesions with a central vein was significantly lower in migraine than in MS, with a median percentage of 22 (quartiles: 15, 54) in a cohort of ten patients with migraine20. This finding is illustrated by a representative example in Fig. 1.
Other diseases. The presence of central veins within white matter lesions in disorders with highly overlapping pathological findings to MS, such as acute disseminated encephalomyelitis (ADEM)42,43, has not yet been investigated.
Statements and recommendations
-
The available evidence from MRI studies indicates that in comparison to patients with MS, individuals with AQP4-IgG-positive NMOSD, SAD (Behçet syndrome, systemic lupus erythematosus and antiphospholipid syndrome), CSVD, Susac syndrome and migraine have a significantly lower proportion of brain lesions with a central vein. These early results need to be validated by future studies. Other MRI mimics of MS, such as neurosarcoidosis and Sjögren syndrome13, should also be investigated
-
Pathological mimics of MS, such as ADEM, require further investigation to assess the presence of the central veins on MRI
-
Because the differential diagnosis of MS is broad, pooling of data from multiple centres would be a realistic strategy to perform a systematic, well-powered evaluation of the central vein in a variety of diseases
Radiological definitions
Discussion
An accepted standard radiological definition of the central vein would be useful to enable uniform imaging practices among clinicians. Interestingly, existing studies demonstrate good agreement when defining the radiological characteristics of a central vein15,16,17,20,28,30,31,32,38,44,45,46,47: first, the vein should appear as a thin line or dot; second, when technically possible, the vein should be visualized in at least two perpendicular planes; and third, the vein can run partially or entirely through the lesion, but must be located centrally regardless of the lesion's shape. In Box 2, we suggest a standard radiological definition based on these characteristics. Examples of lesions with and without central veins are provided in Figs 1,2.
The lesions are classified as harbouring a central vein (parts a–c), not harbouring a central vein (parts d–f), or excluded from analysis (parts g–i). FLAIR* (combined T2*-weighted MRI and fluid-attenuated inversion recovery) images were collected at 3 T from the NIH multiple sclerosis cohort, and were reformatted in all three planes (axial, coronal and sagittal). The relevant lesions are indicated by arrowheads in each case. a | Dawson's finger-shaped lesion with a central vein running perpendicular to the sagittal plane. b | Periventricular lesion with a hypointense rim located next to the atrium of the lateral ventricle. c | Small finger-like lesion with a diameter slightly >3 mm in its short axis. The central vein is not as conspicuous as in previous examples, but it is still visible. d | Small deep white matter lesion with diameter >3 mm and no visible central vein in any plane. e | Small subcortical lesion with diameter >3 mm and no visible central vein in any plane. f | Juxtacortical lesion with no visible central vein in any plane. g | Small hyperintense area with diameter <3 mm located around a parenchymal vein. h | Periventricular lesion with branching veins. i | Confluent lesions with multiple veins.
A more challenging task is to establish a standard radiological definition of the CVS to improve diagnosis of MS. One proposed definition is the '40% rule', first introduced by Evangelou and colleagues15, which assesses the percentage of lesions with a central vein and uses a cut-off value of 40% to radiologically distinguish MS from non-MS disease states. This simple threshold approach was successfully confirmed (100% positive and negative predictive value for MS) by the same group in a prospective study involving 29 patients who presented with possible MS, that is, typical clinically isolated syndrome (CIS) with insufficient MRI findings, or an atypical CIS presentation with MRI findings suggestive of MS46. The 40% rule was further confirmed by an independent group in a cohort of 17 patients with RRMS44. A recent study, in which patients with MS were compared with healthy volunteers and non-MS patients presenting with neurological syndromes, confirmed that diagnostic certainty could be increased by combining the published MRI criteria with visual assessment of the 40% rule48. However, this rule has some limitations, as counting the number of lesions would be time-consuming in patients with high lesion load and, as highlighted above, >40% of brain lesions can be CVS-positive in some patients without MS.
Another approach, first tested by Kilsdonk et al., combines the number and location of lesions with the percentage of lesions with a central vein16. In a cohort of 16 patients with MS and 16 individuals with risk factors for vascular disease, the authors reported that MS could be diagnosed with a sensitivity of 100% (95% CI 83–100%) and a specificity of 88% (95% CI 62–98%) when all lesions in the brain were considered. When the analysis was restricted to deep white matter lesions, the authors still found a relatively high sensitivity of 81% (95% CI 54–96%), and the specificity increased to 94% (95% CI 70–100%). The capacity of the central vein to discriminate between MS and non-MS white matter lesions was confirmed in another study with five MS and nine non-MS patients, which reported a sensitivity of 84%, a specificity of 89%, a positive predictive value of 94%, a negative predictive value of 73%, and a diagnostic accuracy of 86%45. However, this approach still requires the total number of lesions to be counted in patients' brains.
To overcome this issue, one group recently proposed that assessment of ten lesions per patient might be sufficient. By use of this approach, a diagnosis of MS could be predicted with 90% accuracy in 44 of 45 patients15. More recently, an even simpler set of diagnostic rules for CVS was introduced, consisting of the following three criteria18: if there are six or more morphologically characteristic lesions, the diagnosis is inflammatory demyelination; if there are fewer than six morphologically characteristic lesions, but morphologically characteristic lesions outnumber non-perivenous lesions, the diagnosis is inflammatory demyelination; if neither of these conditions are met, inflammatory demyelination should not be diagnosed. The morphologically characteristic lesions considered here had a 'coffee bean' or 'Dawson's finger' appearance when the MRI slice was along the vein's axis, and a 'ring' or 'doughnut' appearance when the MRI slice was approximately perpendicular to the vein. By applying these rules in a cohort of 13 patients with MS and seven patients diagnosed with small vessel ischaemia, all patients were correctly classified, and the classification process took <2 min per case.
Standardized lesion selection, based on existing MRI criteria2,3,49,50, would enable the proposed CVS rules to be compared across different raters and sites. Given the potential confounding factors of small perivascular spaces surrounding veins (Virchow–Robin space), confluent lesions, lesions with multiple distinct veins, and lesions that are poorly visible owing to image artefacts, we introduce a set of exclusion criteria for lesions (Box 2). Examples of excluded lesions are provided in Fig. 2.
Statements and recommendations
-
A standard radiological definition of a central vein should rely on the characteristics outlined in Box 2
-
No standard radiological definition of the CVS has yet been established
-
To define the CVS, we need a set of simple rules that are practical for clinicians to use while providing the highest possible accuracy and confidence
-
Because patients without MS can display a central vein in >40% of their total lesions, the proposed '40% rule' might not be applicable for all diseases
-
When defining the CVS, several exclusion criteria should be applied to lesions (Box 2)
Imaging central veins with MRI
Discussion
Structural imaging of small cerebral veins is best done using the T2*-based contrast mechanism, which exploits the magnetic properties of blood27. The paramagnetic deoxyhaemoglobin inside venous blood perturbs the local magnetic field and generates reduced signal intensity in voxels containing a vein, causing veins to appear hypointense on T2*-weighted images27,51. Since the first in vivo observation of central veins in the brains of patients with MS28, a variety of T2*-based acquisitions have been employed at different magnetic field strengths to image veins inside MS plaques30,34,45,46,52,53.
Several studies have employed a conventional 2D gradient-echo (GRE) sequence, which allows exquisite submillimetre in-plane resolution, especially at 7 T (Refs 17,19,21,29,32,52). However, 2D GRE acquisitions are typically slow (>10 min), only partially cover the supratentorial brain, and provide poor image resolution in the inferior–superior plane owing to thick slices and/or slice gaps.
Some studies have utilized a 3D T2*-weighted GRE sequence to overcome the slice gap issue, and have applied parallel imaging to shorten scan time while maintaining high image resolution (typically 0.5 × 0.5 × 1–3 mm)39,45,54,55. These T2*-weighted images can be post-processed using the susceptibility-weighted imaging (SWI) technique to further enhance venous conspicuity56. The 3D GRE sequence can also be set up to have a multi-echo read-out57. The multi-echo acquisition can then provide quantitative (and/or multi-contrast) imaging through the use of advanced post-processing techniques58,59.
Another variation of the 3D GRE sequence uses a segmented echo planar imaging (3D EPI) read-out to speed up the acquisition while providing more-efficient brain coverage and isotropic voxel size60. Isotropic resolution — a feature that is available on many radiology viewing platforms — is particularly useful to reformat images in any desired plane, and enables veins to be well visualized irrespective of their orientation. Moreover, the use of small isotropic voxel dimensions increases the sensitivity to small parenchymal veins within lesions61, while reducing the sensitivity to artefacts due to background field inhomogeneities. A shorter scan is also beneficial for limiting the head motion that can occur during the acquisition. Recently, the 3D EPI approach was demonstrated to image perivenous MS lesions throughout the brain at submillimetre resolution (0.55 mm isotropic) in <4 min using a 3 T MRI scanner53. Moreover, this 3D EPI acquisition was shown to be more sensitive than conventional 3D T2* GRE41, probably owing to its smaller voxel dimensions.
Although T2*-based imaging with a 7 T scanner provides the highest sensitivity for central vein detection47, 1.5 T (Ref. 38) and 3 T (Ref. 61) scanners can still provide high rates (>80%) of vein detection if optimized T2* protocols are used. Another way to increase vein conspicuity on T2*-weighted images is to perform SWI28,62 and/or inject an intravascular contrast agent (a chelate of gadolinium, which is paramagnetic) during the MRI acquisition28,53,63. The latter solution is straightforward to implement, as MRI protocols for MS often involve the injection of contrast agent, which can be accomplished via a power injector while the scan is ongoing. Compared with manual contrast injection by a technician and the recommended 5 min wait before post-contrast imaging4,22, such a procedure would not prolong the MRI examination.
Unlike T2-weighted fluid-attenuated inversion recovery (FLAIR) images, T2*-weighted and SWI images lack cerebrospinal fluid suppression and are, therefore, less able to demonstrate contrast between lesions and surrounding tissues, making the detection of lesions more difficult. To overcome these issues, two research groups have recently proposed that FLAIR and T2* images should be combined in a single image34,55. Grabner et al.55 introduced a method that transforms FLAIR images using SWI phase masks from T2*-weighted images, thereby creating a FLAIR–SWI contrast. The other approach, proposed by Sati et al.34 and known as FLAIR*, uses 1 mm isotropic 3D FLAIR (for lesion detection) and 0.55 mm isotropic 3D EPI (for vein detection) sequences — both acquired in <10 min — and provides high-resolution isotropic images of the whole brain. Recent studies reported on the utility of FLAIR* at various field strengths for differentiating MS from other diseases16,20,64, and for improving diagnostic accuracy31,44,48.
T2*-based imaging of the spinal cord is much more challenging, owing to factors such as the small physical dimensions of the spinal cord, strong magnetic field inhomogeneity caused by surrounding tissues (bones, soft tissues and air), and physiological motion (pulsation of cerebrospinal fluid flow, and cardiac and respiratory movement). Nonetheless, recent studies have demonstrated that high-quality, high-resolution T2*-weighted imaging of the cervical65,66 and thoracic67 cord is possible in patients with MS. However, no central vein findings have yet been reported.
Standardization of the optimized MRI acquisitions across centres will be important for the widespread dissemination of central vein imaging. Similarly, standardized image reading and interpretation guidelines will be required to train radiologists and neuroradiologists from non-specialist centres. This process could, in principle, be facilitated by future development of automated image analysis tools for the detection of central veins.
Statements and recommendations
-
Imaging of veins in the brain can be performed using T2*-based MRI sequences at any magnetic field strength (1.5 T, 3 T or 7 T). Although T2* imaging is most sensitive at 7 T, a high detection rate can still be achieved at clinical field strengths (1.5 T and 3 T) with optimized sequences
-
Owing to the small dimensions of the central veins, images should be acquired at the highest resolution possible. The use of submillimetre voxel dimensions can be particularly helpful
-
Images should be acquired using isotropic voxels to enable multiplanar visualization of central veins regardless of their orientation in the brain
-
Specific acquisition protocols (with SWI and/or gadolinium injection) aimed at improving central vein detection require further evaluation, especially at lower field strength (1.5 T)
-
High-resolution isotropic T2*with 3D EPI is currently the most promising acquisition to adequately detect central veins while preserving a clinically compatible scan time. However, its use will be limited to expert academic centres until the sequence is made routinely available by MRI scanner manufacturers
-
Combined FLAIR and T2* images have the potential to become a standard clinical protocol, but manufacturer-provided software for direct, automatic image post-processing on the scanner is necessary for widespread dissemination
-
The intra-rater, inter-rater, scan–rescan and inter-scanner reliability of central vein detection on the optimized MRI sequences should be investigated
-
High-quality, high-resolution T2*-weighted imaging of the spinal cord to detect central veins in MS lesions needs further development
Evaluating the CVS for MS diagnosis
Discussion
Most studies on the use of the CVS for MS diagnosis have included small cohorts of patients in whom the diagnosis (MS or one of its mimics) was already known. As discussed above, the results from these single-centre studies support a beneficial role for the CVS in specifically identifying MS lesions. However, larger-scale studies are still required to confirm these early results.
To formally establish the clinical value of the CVS for the differential diagnosis at disease onset, a large, prospective, multicentre study including patients at first presentation of possible MS is necessary. In any study design, important factors such as disease duration, disease severity and lesion load would need to be taken into account, as they might affect the frequency of the central vein in MS mimics. For example, larger lesions are more likely to incidentally harbour a blood vessel, although the vessel location in lesions caused by MS mimics is more likely to be eccentric than central.
Although differential diagnosis is the most obvious diagnostic application of the CVS, one study has prospectively investigated the predictive diagnostic value of the CVS in patients in whom the question of inflammatory demyelination had been raised at first presentation, that is, patients showing typical CIS or with atypical neurological presentations46. In this longitudinal study, 29 undiagnosed individuals were recruited and underwent a T2*-weighted scan. On the basis of the CVS only, a provisional diagnosis of MS was predicted using the 40% rule. Of the 22 patients who eventually received a clinical diagnosis within a median follow-up period of 26 months, 13 patients diagnosed as having MS had central veins in >40% of brain lesions at baseline. All nine patients whose condition was not diagnosed as MS had central veins in <40% of lesions. According to these data, the CVS had 100% positive and negative predictive value for the diagnosis of MS, although the conclusions were limited by the small number of participants.
To further validate these early results, the same group is currently conducting a prospective longitudinal clinical trial68 aiming to recruit 60 patients suspected — but not proven — to have MS. Recruited patients will undergo a single research MRI brain scan at 3 T, which will be evaluated by blinded investigators to make a diagnosis of MS or non-MS on the basis of the CVS criterion only. No other research tests will be performed, and the patients will be followed up over time until a final diagnosis is made. Although this prospective study should provide strong evidence to support or refute the predictive value of the CVS for MS diagnosis, it remains a single-centre trial and is, therefore, limited in terms of cohort size and external validity. The clinical validation would benefit from a multicentre trial using a similar design but including a larger number of participants and centres, as well as standardized MRI protocols and methodology for CVS identification.
An alternative study design could be used to investigate whether the CVS improves the accuracy of the 2010 McDonald criteria. Modifications of existing MRI criteria to incorporate the CVS could be tested initially in a single-centre prospective study recruiting patients with CIS (or even with radiologically isolated syndrome), followed by a larger-scale multicentre study if the preliminary results are positive.
Statements and recommendations
-
The clinical value of the CVS should be evaluated in the context of the differential diagnosis of suspected MS, the diagnostic predictive value in patients with possible or early MS, and the potentially improved accuracy of the 2010 McDonald criteria
-
Currently available evidence from a small prospective study supports the high predictive value of the CVS in the diagnosis of MS in patients with typical CIS or atypical neurological presentations
-
Large, prospective multicentre trials including patients at first presentation of neurological signs are needed to evaluate the clinical value of the CVS for MS diagnosis
-
Care should be taken when using the CVS in routine clinical practice until its diagnostic value has been formally established
Conclusions
The NAIMS Cooperative has developed this Consensus Statement to better define and evaluate the CVS, as detected by MRI, for the diagnosis of MS. More precisely, our recommendations underscore the need for further investigation of the central vein in MS and its mimics. We have proposed a standard radiological definition of the central vein (Box 2), but we strongly recommend additional investigation to define the optimal CVS criteria. Our recommendations also promote standardization of MRI protocols and lesion selection criteria to assess central veins. Finally, we recommend investigation of the clinical value of the CVS through large multicentre studies involving patients with established diagnoses of MS and its mimics, as well as undiagnosed patients suspected of having MS.
Taken together, our recommendations provide a roadmap to help establish a high-impact role for the CVS in improving the diagnosis of MS. This Consensus Statement is in line with recent guidelines from the MAGNIMS group1,4 and CMSC task force22, which both highlighted the potential of the CVS and its associated MRI acquisitions while calling for further research before considering an update of the diagnostic criteria. Overall, the NAIMS Cooperative is optimistic that the CVS will eventually find substantial clinical utility in daily practice, thus adding another layer of success to a technology that has changed the field of neurology over the past few decades.
References
Filippi, M. et al. MRI criteria for the diagnosis of multiple sclerosis: MAGNIMS consensus guidelines. Lancet Neurol. 15, 292–303 (2016).
McDonald, W. I. et al. Recommended diagnostic criteria for multiple sclerosis: guidelines from the International Panel on the diagnosis of multiple sclerosis. Ann. Neurol. 50, 121–127 (2001).
Polman, C. H. et al. Diagnostic criteria for multiple sclerosis: 2010 revisions to the McDonald criteria. Ann. Neurol. 69, 292–302 (2011).
Rovira, À. et al. Evidence-based guidelines: MAGNIMS consensus guidelines on the use of MRI in multiple sclerosis — clinical implementation in the diagnostic process. Nat. Rev. Neurol. 11, 471–482 (2015).
Brownlee, W. J., Swanton, J. K., Altmann, D. R., Ciccarelli, O. & Miller, D. H. Earlier and more frequent diagnosis of multiple sclerosis using the McDonald criteria. J. Neurol. Neurosurg. Psychiatry 86, 584–585 (2015).
Runia, T. F., Jafari, N. & Hintzen, R. Q. Application of the 2010 revised criteria for the diagnosis of multiple sclerosis to patients with clinically isolated syndromes. Eur. J. Neurol. 20, 1510–1516 (2013).
Bates, D. Treatment effects of immunomodulatory therapies at different stages of multiple sclerosis in short-term trials. Neurology 76 (1 Suppl. 1), S14–S25 (2011).
Freedman, M. S. Long-term follow-up of clinical trials of multiple sclerosis therapies. Neurology 76 (1 Suppl. 1), S26–S34 (2011).
Gómez-Moreno, M., Díaz-Sánchez, M. & Ramos-González, A. Application of the 2010 McDonald criteria for the diagnosis of multiple sclerosis in a Spanish cohort of patients with clinically isolated syndromes. Mult. Scler. 18, 39–44 (2012).
Swanton, J. K. et al. MRI criteria for multiple sclerosis in patients presenting with clinically isolated syndromes: a multicentre retrospective study. Lancet Neurol. 6, 677–686 (2007).
Solomon, A. J. et al. The contemporary spectrum of multiple sclerosis misdiagnosis: a multicenter study. Neurology 87, 1393–1399 (2016).
Solomon, A. J., Klein, E. P. & Bourdette, D. “Undiagnosing” multiple sclerosis: the challenge of misdiagnosis in MS. Neurology 78, 1986–1991 (2012).
Aliaga, E. S. & Barkhof, F. MRI mimics of multiple sclerosis. Handb. Clin. Neurol. 122, 291–316 (2014).
Miller, D. H. et al. Differential diagnosis of suspected multiple sclerosis: a consensus approach. Mult. Scler. 14, 1157–1174 (2008).
Tallantyre, E. C. et al. Ultra-high-field imaging distinguishes MS lesions from asymptomatic white matter lesions. Neurology 76, 534–539 (2011).
Kilsdonk, I. D. et al. Improved differentiation between MS and vascular brain lesions using FLAIR* at 7 Tesla. Eur. Radiol. 24, 841–849 (2014).
Kister, I., Herbert, J., Zhou, Y. & Ge, Y. Ultrahigh-field MR (7 T) imaging of brain lesions in neuromyelitis optica. Mult. Scler. Int. 2013, 398259 (2013).
Mistry, N. et al. Imaging central veins in brain lesions with 3-T T2*-weighted magnetic resonance imaging differentiates multiple sclerosis from microangiopathic brain lesions. Mult. Scler. 22, 1289–1296 (2016).
Sinnecker, T. et al. Distinct lesion morphology at 7-T MRI differentiates neuromyelitis optica from multiple sclerosis. Neurology 79, 708–714 (2012).
Solomon, A. J. et al. “Central vessel sign” on 3T FLAIR* MRI for the differentiation of multiple sclerosis from migraine. Ann. Clin. Transl. Neurol. 3, 82–87 (2015).
Wuerfel, J. et al. Lesion morphology at 7 Tesla MRI differentiates Susac syndrome from multiple sclerosis. Mult. Scler. 18, 1592–1599 (2012).
Traboulsee, A. et al. Revised recommendations of the Consortium of MS Centers task force for a standardized MRI protocol and clinical guidelines for the diagnosis and follow-up of multiple sclerosis. AJNR Am. J. Neuroradiol. 37, 394–401 (2016).
Quinn, M. P., Kremenchutzky, M. & Menon, R. S. Venocentric lesions: an MRI marker of MS? Front. Neurol. 4, 98 (2013).
Rae-Grant, A. D., Wong, C., Bernatowicz, R. & Fox, R. J. Observations on the brain vasculature in multiple sclerosis: a historical perspective. Mult. Scler. Relat. Disord. 3, 156–162 (2014).
Adams, C. W. The onset and progression of the lesion in multiple sclerosis. J. Neurol. Sci. 25, 165–182 (1975).
Barnett, M. H. & Prineas, J. W. Relapsing and remitting multiple sclerosis: pathology of the newly forming lesion. Ann. Neurol. 55, 458–468 (2004).
Reichenbach, J. R., Venkatesan, R., Schillinger, D. J., Kido, D. K. & Haacke, E. M. Small vessels in the human brain: MR venography with deoxyhemoglobin as an intrinsic contrast agent. Radiology 204, 272–277 (1997).
Tan, I. L. et al. MR venography of multiple sclerosis. AJNR Am. J. Neuroradiol. 21, 1039–1042 (2000).
Hammond, K. E. et al. Quantitative in vivo magnetic resonance imaging of multiple sclerosis at 7 Tesla with sensitivity to iron. Ann. Neurol. 64, 707–713 (2008).
Tallantyre, E. C. et al. Demonstrating the perivascular distribution of MS lesions in vivo with 7-Tesla MRI. Neurology 70, 2076–2078 (2008).
Kilsdonk, I. D. et al. Morphological features of MS lesions on FLAIR* at 7 T and their relation to patient characteristics. J. Neurol. 261, 1356–1364 (2014).
Kuchling, J. et al. Identical lesion morphology in primary progressive and relapsing–remitting MS — an ultrahigh field MRI study. Mult. Scler. 20, 1866–1871 (2014).
Kidd, D. et al. Cortical lesions in multiple sclerosis. Brain 122, 17–26 (1999).
Sati, P., George, I. C., Shea, C. D., Gaitan, M. I. & Reich, D. S. FLAIR*: a combined MR contrast technique for visualizing white matter lesions and parenchymal veins. Radiology 265, 926–932 (2012).
Dawson, J. W. The histology of disseminated sclerosis. Trans. R. Soc. Edinb. 50, 517–740 (1916).
Weinstock-Guttman, B. et al. Lipid profiles are associated with lesion formation over 24 months in interferon-β treated patients following the first demyelinating event. J. Neurol. Neurosurg. Psychiatry 84, 1186–1191 (2013).
Zivadinov, R. et al. Smoking is associated with increased lesion volumes and brain atrophy in multiple sclerosis. Neurology 73, 504–510 (2009).
Massacesi, L. Evaluation by brain MRI of white matter perivenular lesions in inflammatory microangiopathic ischemia and in demyelinating mutiple sclerosis lesions [abstract O2217]. Eur. J. Neurol. 23 (Suppl. 2), 86 (2016).
Lummel, N. et al. Presence of a central vein within white matter lesions on susceptibility weighted imaging: a specific finding for multiple sclerosis? Neuroradiology 53, 311–317 (2011).
Lane, J. I., Bolster, B., Campeau, N. G., Welker, K. M. & Gilbertson, J. R. Characterization of multiple sclerosis plaques using susceptibility-weighted imaging at 1.5 T: can perivenular localization improve specificity of imaging criteria? J. Comput. Assist. Tomogr. 39, 317–320 (2015).
Samaraweera, A. P. et al. The central vein sign in multiple sclerosis lesions is present irrespective of the T2* sequence at 3 T. J. Neuroimaging http://dx.doi.org/10.1111/jon.12367 (2016).
Van Bogaert, L. Post-infectious encephalomyelitis and multiple sclerosis; the significance of perivenous encephalomyelitis. J. Neuropathol. Exp. Neurol. 9, 219–249 (1950).
Young, N. P. et al. Perivenous demyelination: association with clinically defined acute disseminated encephalomyelitis and comparison with pathologically confirmed multiple sclerosis. Brain 133, 333–348 (2010).
Campion, T. et al. FLAIR* for the non-invasive histological diagnosis of multiple sclerosis [abstract S29.003]. Neurology 84 (Suppl.), 14 (2015).
Kau, T. et al. The “central vein sign”: is there a place for susceptibility weighted imaging in possible multiple sclerosis? Eur. Radiol. 23, 1956–1962 (2013).
Mistry, N. et al. Central veins in brain lesions visualized with high-field magnetic resonance imaging: a pathologically specific diagnostic biomarker for inflammatory demyelination in the brain. JAMA Neurol. 70, 623–628 (2013).
Tallantyre, E. C. et al. A comparison of 3T and 7T in the detection of small parenchymal veins within MS lesions. Invest. Radiol. 44, 491–494 (2009).
George, I. C. et al. Clinical 3-tesla FLAIR* MRI improves diagnostic accuracy in multiple sclerosis. Mult. Scler. http://dx.doi.org/10.1177/1352458515624975 (2016).
Barkhof, F. et al. Comparison of MRI criteria at first presentation to predict conversion to clinically definite multiple sclerosis. Brain 120, 2059–2069 (1997).
Tintore, M. et al. Isolated demyelinating syndromes: comparison of different MR imaging criteria to predict conversion to clinically definite multiple sclerosis. AJNR Am. J. Neuroradiol. 21, 702–706 (2000).
Chavhan, G. B., Babyn, P. S., Thomas, B., Shroff, M. M. & Haacke, E. M. Principles, techniques, and applications of T2*-based MR imaging and its special applications. Radiographics 29, 1433–1449 (2009).
Ge, Y., Zohrabian, V. M. & Grossman, R. I. Seven-Tesla magnetic resonance imaging: new vision of microvascular abnormalities in multiple sclerosis. Arch. Neurol. 65, 812–816 (2008).
Sati, P. et al. Rapid, high-resolution, whole-brain, susceptibility-based MRI of multiple sclerosis. Mult. Scler. 20, 1464–1470 (2014).
Dal-Bianco, A. et al. Veins in plaques of multiple sclerosis patients — a longitudinal magnetic resonance imaging study at 7 Tesla. Eur. Radiol. 25, 2913–2920 (2015).
Grabner, G. et al. Analysis of multiple sclerosis lesions using a fusion of 3.0 T FLAIR and 7.0 T SWI phase: FLAIR SWI. J. Magn. Reson. Imaging 33, 543–549 (2011).
Haacke, E. M., Xu, Y., Cheng, Y. C. & Reichenbach, J. R. Susceptibility weighted imaging (SWI). Magn. Reson. Med. 52, 612–618 (2004).
Denk, C. & Rauscher, A. Susceptibility weighted imaging with multiple echoes. J. Magn. Reson. Imaging 31, 185–191 (2010).
Luo, J., Yablonskiy, D. A., Hildebolt, C. F., Lancia, S. & Cross, A. H. Gradient echo magnetic resonance imaging correlates with clinical measures and allows visualization of veins within multiple sclerosis lesions. Mult. Scler. 20, 349–355 (2014).
Sati, P., Cross, A. H., Luo, J., Hildebolt, C. F. & Yablonskiy, D. A. in vivo quantitative evaluation of brain tissue damage in multiple sclerosis using gradient echo plural contrast imaging technique. Neuroimage 51, 1089–1097 (2010).
Zwanenburg, J. J., Versluis, M. J., Luijten, P. R. & Petridou, N. Fast high resolution whole brain T2* weighted imaging using echo planar imaging at 7T. Neuroimage 56, 1902–1907 (2011).
Dixon, J. E., Simpson, A., Mistry, N., Evangelou, N. & Morris, P. G. Optimisation of T2*-weighted MRI for the detection of small veins in multiple sclerosis at 3 T and 7 T. Eur. J. Radiol. 82, 719–727 (2013).
Reichenbach, J. R. & Haacke, E. M. High-resolution BOLD venographic imaging: a window into brain function. NMR Biomed. 14, 453–467 (2001).
Maggi, P. et al. SWI enhances vein detection using gadolinium in multiple sclerosis. Acta Radiol. Open 4, 2047981614560938 (2015).
Vuolo, L., Dewey, B. E., Sati, P., Massacesi, L. & Reich, D. S. Efficiency of FLAIR* AT 1.5T, 3T and 7T for detecting perivenular lesions in multiple sclerosis [abstract P479]. Presented at the 31st Congress of the European Committee for Treatment and Research in Multiple Sclerosis (2015).]
Dula, A. N. et al. Magnetic resonance imaging of the cervical spinal cord in multiple sclerosis at 7T. Mult. Scler. 22, 320–328 (2016).
Ozturk, A. et al. Axial 3D gradient-echo imaging for improved multiple sclerosis lesion detection in the cervical spinal cord at 3T. Neuroradiology 55, 431–439 (2013).
Lefeuvre, J. et al. MRI of the thoracic spinal cord in multiple sclerosis at 7T [abstract 4397]. Presented at the 24th Annual Meeting of the International Society for Magnetic Resonance in Medicine (2016).
US National Library of Medicine. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT02485223?term (2015).
Acknowledgements
The authors would like to thank Christina J. Azevedo, Michael G. Dwyer, Léorah Freeman, Christoph Juchem, Shannon Kolind, Naila Makhani, Govind Nair, Nico Papinutto, Haochang Shou, Daniel Schwartz, Ferdinand Schweser, Elizabeth Sweeney, Ian Tagge, Shahamat Tauhid and Subhash Tummala for their participation in, and contribution to, the group workshop at the NAIMS meeting in Los Angeles. Matthew Schindler is acknowledged for helpful suggestions. The authors also acknowledge Rohit Bakshi, Peter Calabresi, Ciprian Crainiceanu and Jack Simon for their contribution as NAIMS members. The authors, on behalf of the NAIMS Cooperative, would also like to thank the Race to Erase Multiple Sclerosis for financial support, and Joel Arnold, Aracely Delgadillo, and Liz Seares for helping with organization of the NAIMS meeting. This research was supported in part by the Intramural Research Program of the National Institute of Neurological Disorders and Stroke.
Author information
Authors and Affiliations
Consortia
Contributions
P.S. researched the data for the article. P.S., D.P. and D.S.R. wrote the text. All authors made substantial contributions to discussions of the content, and reviewed and/or edited the manuscript before submission. D.P. and D.S.R. contributed equally to the work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
PowerPoint slides
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article's Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Sati, P., Oh, J., Constable, R. et al. The central vein sign and its clinical evaluation for the diagnosis of multiple sclerosis: a consensus statement from the North American Imaging in Multiple Sclerosis Cooperative. Nat Rev Neurol 12, 714–722 (2016). https://doi.org/10.1038/nrneurol.2016.166
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrneurol.2016.166
This article is cited by
-
Central intra-lesional iron deposits as a possible novel imaging marker at 7 Tesla MRI in Susac Syndrome - an exploratory study
BMC Medical Imaging (2024)
-
Emerging imaging markers in radiologically isolated syndrome: implications for earlier treatment initiation
Neurological Sciences (2024)
-
The rising role of magnetic resonance imaging biomarkers in diagnosing multiple sclerosis
European Radiology (2023)
-
Glia Connect Inflammation and Neurodegeneration in Multiple Sclerosis
Neuroscience Bulletin (2023)
-
Present and future of the diagnostic work-up of multiple sclerosis: the imaging perspective
Journal of Neurology (2023)