Key Points
-
Initiation of statin use in late life does not seem to prevent cognitive decline and dementia over the subsequent few years
-
The current literature does not address whether mid-life or long-term statin use has beneficial effects on subsequent cognition
-
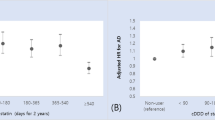
Many studies that assessed time-updated statin use suggest a protective effect of statin use on cognitive decline, but these findings are probably attributable to reverse causation
-
Ethical and feasibility considerations limit randomized controlled trials; carefully designed observational studies that use appropriate analytical methods are our best hope for determining the effects of statin use on cognition
-
Future observational work must incorporate study designs and analytical techniques that minimize the potential for bias, especially that which is due to reverse causation and confounding
Abstract
Firm conclusions about whether mid-life or long-term statin use has an impact on cognitive decline and dementia remain elusive. Here, our objective was to systematically review, synthesize and critique the epidemiological literature that examines the relationship between statin use and cognition, so as to assess the current state of knowledge, identify gaps in our understanding, and make recommendations for future research. We summarize the findings of randomized controlled trials (RCTs) and observational studies, grouped according to study design. We discuss the methods for each, and consider likely sources of bias, such as reverse causation and confounding. Although observational studies that considered statin use at or near the time of dementia diagnosis suggest a protective effect of statins, these findings could be attributable to reverse causation. RCTs and well-conducted observational studies of baseline statin use and subsequent cognition over several years of follow-up do not support a causal preventative effect of late-life statin use on cognitive decline or dementia. Given that much of the human research on statins and cognition in the future will be observational, careful study design and analysis will be essential.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Stone, N. J. et al. 2013 ACC/AHA Guideline on the treatment of blood cholesterol to reduce atherosclerotic cardiovascular risk in adults: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Circulation 129 (25 Suppl. 2), S1–S45 (2014).
Pencina, M. J. et al. Application of new cholesterol guidelines to a population-based sample. New Engl. J. Med. 370, 1422–1431 (2014).
National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III). Third report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III) final report. Circulation 106, 3143–3421 (2002).
Grundy, S. M. et al. Implications of recent clinical trials for the National Cholesterol Education Program Adult Treatment Panel III Guidelines. J. Am. Coll. Cardiol. 44, 720–732 (2004).
Naci, H. et al. Comparative benefits of statins in the primary and secondary prevention of major coronary events and all-cause mortality: a network meta-analysis of placebo-controlled and active-comparator trials. Eur. J. Prev. Cardiol. 20, 641–657 (2013).
Udell, J. A. & Ray, J. G. Primary and secondary prevention of heart failure with statins. Expert Rev. Cardiovasc. Ther. 4, 917–926 (2006).
Taylor, F. et al. Statins for the primary prevention of cardiovascular disease. Cochrane Database of Systematic Reviews, Issue 1. Art. No.: CD004816. http://dx.doi.org/10.1002/14651858.CD004816.pub4.
Rojas-Fernandez, C. H. & Cameron, J. C. Is statin-associated cognitive impairment clinically relevant? A narrative review and clinical recommendations. Ann. Pharmacother. 46, 549–557 (2012).
Evans, M. A. & Golomb, B. A. Statin-associated adverse cognitive effects: survey results from 171 patients. Pharmacotherapy 29, 800–811 (2009).
FDA drug safety communication: important safety label changes to cholesterol-lowering statin drugs. U.S. Food and Drug Administration [online] (2012).
Anstey, K. J., Lipnicki, D. M. & Low, L. F. Cholesterol as a risk factor for dementia and cognitive decline: a systematic review of prospective studies with meta-analysis. Am. J. Geriatr. Psychiatry 16, 343–354 (2008).
Stewart, R., White, L. R., Xue, Q. L. & Launer, L. J. Twenty-six-year change in total cholesterol levels and incident dementia: the Honolulu–Asia Aging Study. Arch. Neurol. 64, 103–107 (2007).
Panza, F. et al. Lipid metabolism in cognitive decline and dementia. Brain Res. Rev. 51, 275–292 (2006).
Gorelick, P. B. et al. Vascular contributions to cognitive impairment and dementia: a statement for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 42, 2672–2713 (2011).
Jain, M. K. & Ridker, P. M. Anti-inflammatory effects of statins: clinical evidence and basic mechanisms. Nat. Rev. Drug Discov. 4, 977–987 (2005).
Miida, T., Takahashi, A. & Ikeuchi, T. Prevention of stroke and dementia by statin therapy: experimental and clinical evidence of their pleiotropic effects. Pharmacol. Ther. 113, 378–393 (2007).
Bifulco, M., Malfitano, A. M. & Marasco, G. Potential therapeutic role of statins in neurological disorders. Expert Rev. Neurother. 8, 827–837 (2008).
Sierra, S. et al. Statins as neuroprotectants: a comparative in vitro study of lipophilicity, blood–brain-barrier penetration, lowering of brain cholesterol, and decrease of neuron cell death. J. Alzheimers Dis. 23, 307–318 (2011).
Carlsson, C. M. et al. Effects of atorvastatin on cerebral blood flow in middle-aged adults at risk for Alzheimer's disease: a pilot study. Curr. Alzheimer Res. 9, 990–997 (2012).
Locatelli, S. et al. Reduction of plasma 24S-hydroxycholesterol (cerebrosterol) levels using high-dosage simvastatin in patients with hypercholesterolemia: evidence that simvastatin affects cholesterol metabolism in the human brain. Arch. Neurol. 59, 213–216 (2002).
Lutjohann, D. & von Bergmann, K. 24S-Hydroxycholesterol: a marker of brain cholesterol metabolism. Pharmacopsychiatry 36 (Suppl. 2), S102–S106 (2003).
Serrano-Pozo, A. et al. Effects of simvastatin on cholesterol metabolism and Alzheimer disease biomarkers. Alzheimer Dis. Assoc. Disord. 24, 220–226 (2010).
Vega, G. L. et al. Reduction in levels of 24S-hydroxycholesterol by statin treatment in patients with Alzheimer disease. Arch. Neurol. 60, 510–515 (2003).
Höglund, K. & Blennow, K. Effect of HMG-CoA reductase inhibitors on β-amyloid peptide levels: implications for Alzheimer's disease. CNS Drugs 21, 449–462 (2007).
Fassbender, K. et al. Effects of statins on human cerebral cholesterol metabolism and secretion of Alzheimer amyloid peptide. Neurology 59, 1257–1258 (2002).
Höglund, K. et al. Statin treatment and a disease-specific pattern of β-amyloid peptides in Alzheimer's disease. Exp. Brain Res. 164, 205–214 (2005).
Riekse, R. G. et al. Effect of statins on Alzheimer's disease biomarkers in cerebrospinal fluid. J. Alzheimers Dis. 10, 399–406 (2006).
Arvanitakis, Z. et al. Statins, incident Alzheimer disease, change in cognitive function, and neuropathology. Neurology 70, 1795–1802 (2008).
Refolo, L. M. et al. Hypercholesterolemia accelerates the Alzheimer's amyloid pathology in a transgenic mouse model. Neurobiol. Dis. 7, 321–331 (2000).
Pollen, D. A. et al. Prevention of Alzheimer's disease in high risk groups: statin therapy in subjects with PSEN1 mutations or heterozygosity for apolipoprotein Eε4. Alzheimers Res. Ther. 2, 31 (2010).
Li, G. et al. Statin therapy is associated with reduced neuropathologic changes of Alzheimer disease. Neurology 69, 878–885 (2007).
McGuinness, B., Craig, D., Bullock, R. & Passmore, P. Statins for the prevention of dementia. Cochrane Database of Systematic Reviews, Issue 2. Art. No.: CD003160 http://dx.doi.org/10.1002/14651858.CD003160.pub2.
Swiger, K. J., Manalac, R. J., Blumenthal, R. S., Blaha, M. J. & Martin, S. S. Statins and cognition: a systematic review and meta-analysis of short- and long-term cognitive effects. Mayo Clin. Proc. 88, 1213–1221 (2013).
Muangpaisan, W., Brayne, C. & Alzheimer's Society Vascular Dementia Systematic Review Group. Systematic review of statins for the prevention of vascular dementia or dementia. Geriatr. Gerontol. Int. 10, 199–208 (2010).
Zhou, B., Teramukai, S. & Fukushima, M. Prevention and treatment of dementia or Alzheimer's disease by statins: a meta-analysis. Dement. Geriatr. Cogn. Disord. 23, 194–201 (2007).
Wong, W. B., Lin, V. W., Boudreau, D. & Devine, E. B. Statins in the prevention of dementia and Alzheimer's disease: a meta-analysis of observational studies and an assessment of confounding. Pharmacoepidemiol. Drug Saf. 22, 345–358 (2013).
Richardson, K. et al. Statins and cognitive function: a systematic review. Ann. Intern. Med. 159, 688–697 (2013).
AlzRisk methods. Alzforum [online] (2015).
Glymour, M. M., Weuve, J., Berkman, L. F., Kawachi, I. & Robins, J. M. When is baseline adjustment useful in analyses of change? An example with education and cognitive change. Am. J. Epidemiol. 162, 267–278 (2005).
Carlsson, C. M. et al. Effects of simvastatin on cerebrospinal fluid biomarkers and cognition in middle-aged adults at risk for Alzheimer's disease. J. Alzheimers Dis. 13, 187–197 (2008).
Carlsson, C. M. et al. Effects of simvastatin on cerebrospinal fluid biomarkers and cognition in middle-aged adults at risk for Alzheimer's disease. J. Alzheimers Dis. 13, 187–197 (2008).
Gibellato, M. G., Moore, J. L., Selby, K. & Bower, E. A. Effects of lovastatin and pravastatin on cognitive function in military aircrew. Aviat. Space Environ. Med. 72, 805–812 (2001).
Kostis, J. B., Rosen, R. C. & Wilson, A. C. Central nervous system effects of HMG CoA reductase inhibitors: lovastatin and pravastatin on sleep and cognitive performance in patients with hypercholesterolemia. J. Clin. Pharmacol. 34, 989–996 (1994).
Muldoon, M. F. et al. Effects of lovastatin on cognitive function and psychological well-being. Am. J. Med. 108, 538–546 (2000).
Muldoon, M. F., Ryan, C. M., Sereika, S. M., Flory, J. D. & Manuck, S. B. Randomized trial of the effects of simvastatin on cognitive functioning in hypercholesterolemic adults. Am. J. Med. 117, 823–829 (2004).
Summers, M. J., Oliver, K. R., Coombes, J. S. & Fassett, R. G. Effect of atorvastatin on cognitive function in patients from the Lipid Lowering and Onset of Renal Disease (LORD) trial. Pharmacotherapy 27, 183–190 (2007).
Trompet, S. et al. Pravastatin and cognitive function in the elderly. Results of the PROSPER study. J. Neurol. 257, 85–90 (2010).
Harrison, R. W. & Ashton, C. H. Do cholesterol-lowering agents affect brain activity? A comparison of simvastatin, pravastatin, and placebo in healthy volunteers. Br. J. Clin. Pharmacol. 37, 231–236 (1994).
Santanello, N. C. et al. Effect of pharmacologic lipid lowering on health-related quality of life in older persons: results from the Cholesterol Reduction in Seniors Program (CRISP) Pilot Study. J. Am. Geriatr. Soc. 45, 8–14 (1997).
Gengo, F. et al. Effects of treatment with lovastatin and pravastatin on daytime cognitive performance. Clin. Cardiol. 18, 209–214 (1995).
Roth, T. et al. Comparative effects of pravastatin and lovastatin on nighttime sleep and daytime performance. Clin. Cardiol. 15, 426–432 (1992).
Heart Protection Study Collaborative Group. MRC/BHF Heart Protection Study of cholesterol lowering with simvastatin in 20,536 high-risk individuals: a randomised placebo-controlled trial. Lancet 360, 7–22 (2002).
Shepherd, J. et al. Pravastatin in elderly individuals at risk of vascular disease (PROSPER): a randomised controlled trial. Lancet 360, 1623–1630 (2002).
Cutler, N. et al. Effects of treatment with simvastatin and pravastatin on cognitive function in patients with hypercholesterolaemia. Br. J. Clin. Pharmacol. 39, 333–336 (1995).
Li, G. et al. Statin therapy and risk of dementia in the elderly: a community-based prospective cohort study. Neurology 63, 1624–1628 (2004).
Li, G. et al. Age-varying association between statin use and incident Alzheimer's disease. J. Am. Geriatr. Soc. 58, 1311–1317 (2010).
Sparks, D. L. et al. Reduced risk of incident AD with elective statin use in a clinical trial cohort. Curr. Alzheimer Res. 5, 416–421 (2008).
Beydoun, M. A. et al. Statins and serum cholesterol's associations with incident dementia and mild cognitive impairment. J. Epidemiol. Community Health 65, 949–957 (2011).
Rea, T. D. et al. Statin use and the risk of incident dementia: the Cardiovascular Health Study. Arch. Neurol. 62, 1047–1051 (2005).
Bernick, C. et al. Statins and cognitive function in the elderly: the Cardiovascular Health Study. Neurology 65, 1388–1394 (2005).
Bettermann, K. et al. Statins, risk of dementia, and cognitive function: secondary analysis of the ginkgo evaluation of memory study. J. Stroke Cerebrovasc. Dis. 21, 436–444 (2012).
Szwast, S. J. et al. Association of statin use with cognitive decline in elderly African Americans. Neurology 69, 1873–1880 (2007).
Steenland, K., Zhao, L., Goldstein, F. C. & Levey, A. I. Statins and cognitive decline in older adults with normal cognition or mild cognitive impairment. J. Am. Geriatr. Soc. 61, 1449–1455 (2013).
Hippisley-Cox, J. & Coupland, C. Unintended effects of statins in men and women in England and Wales: population based cohort study using the QResearch database. BMJ 340, c2197 (2010).
Cramer, C., Haan, M. N., Galea, S., Langa, K. M. & Kalbfleisch, J. D. Use of statins and incidence of dementia and cognitive impairment without dementia in a cohort study. Neurology 71, 344–350 (2008).
Zandi, P. P. et al. Do statins reduce risk of incident dementia and Alzheimer disease? The Cache County Study. Arch. Gen. Psychiatry 62, 217–224 (2005).
Smeeth, L., Douglas, I., Hall, A. J., Hubbard, R. & Evans, S. Effect of statins on a wide range of health outcomes: a cohort study validated by comparison with randomized trials. Br. J. Clin. Pharmacol. 67, 99–109 (2009).
Haag, M. D., Hofman, A., Koudstaal, P. J., Stricker, B. H. & Breteler, M. M. Statins are associated with a reduced risk of Alzheimer disease regardless of lipophilicity. The Rotterdam Study. J. Neurol. Neurosurg. Psychiatry 80, 13–17 (2009).
Starr, J. M. et al. Life long changes in cognitive ability are associated with prescribed medications in old age. Int. J. Geriatr. Psychiatry 19, 327–332 (2004).
Ancelin, M. L. et al. Lipid lowering agents, cognitive decline, and dementia: the three-city study. J. Alzheimers Dis. 30, 629–637 (2012).
Wolozin, B. et al. Simvastatin is associated with a reduced incidence of dementia and Parkinson's disease. BMC Med. 5, 20 (2007).
Chou, C.-Y., Chou, Y.-C., Chou, Y.-J., Yang, Y.-F. & Huang, N. Statin use and incident dementia: a nationwide cohort study of Taiwan. Int. J. Cardiol. 173, 305–310 (2014).
Moher, D., Liberati, A., Tetzlaff, J., Altman, D. G. & PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 6, e1000097 (2009).
Hernán, M. A., Hernández-Diaz, S. & Robins, J. M. A structural approach to selection bias. Epidemiology 15, 615–625 (2004).
Fox, N. C., Warrington, E. K. & Rossor, M. N. Serial magnetic resonance imaging of cerebral atrophy in preclinical Alzheimer's disease. Lancet 353, 2125 (1999).
Jack, C. R. Jr et al. Hypothetical model of dynamic biomarkers of the Alzheimer's pathological cascade. Lancet Neurol. 9, 119–128 (2010).
Sperling, R. A. et al. Toward defining the preclinical stages of Alzheimer's disease: recommendations from the National Institute on Aging-Alzheimer's Association workgroups on diagnostic guidelines for Alzheimer's disease. Alzheimers Dement. 7, 280–292 (2011).
Power, M. C., Tchetgen, E. J., Sparrow, D., Schwartz, J. & Weisskopf, M. G. Blood pressure and cognition: factors that may account for their inconsistent association. Epidemiology 24, 886–893 (2013).
Qiu, C., Winblad, B. & Fratiglioni, L. The age-dependent relation of blood pressure to cognitive function and dementia. Lancet Neurol. 4, 487–499 (2005).
Paglieri, C. et al. Hypertension and cognitive function. Clin. Exp. Hypertens. 30, 701–710 (2008).
Dufouil, C. et al. APOE genotype, cholesterol level, lipid-lowering treatment, and dementia: the Three-City Study. Neurology 64, 1531–1538 (2005).
Rait, G. et al. Survival of people with clinical diagnosis of dementia in primary care: cohort study. BMJ 341, c3584 (2010).
van den Dungen, P. et al. The accuracy of family physicians' dementia diagnoses at different stages of dementia: a systematic review. Int. J. Geriatr. Psychiatry 27, 342–354 (2012).
Taylor, D. H. Jr, Østbye, T., Langa, K. M., Weir, D. & Plassman, B. L. The accuracy of Medicare claims as an epidemiological tool: the case of dementia revisited. J. Alzheimers Dis. 17, 807–815 (2009).
Butler, D., Kowall, N. W., Lawler, E., Michael Gaziano, J. & Driver, J. A. Underuse of diagnostic codes for specific dementias in the Veterans Affairs New England healthcare system. J. Am. Geriatr. Soc. 60, 910–915 (2012).
Brookhart, M. A. et al. Adherence to lipid-lowering therapy and the use of preventive health services: an investigation of the healthy user effect. Am. J. Epidemiol. 166, 348–354 (2007).
Button, K. S. et al. Power failure: why small sample size undermines the reliability of neuroscience. Nat. Rev. Neurosci. 14, 365–376 (2013).
Greenland, S. & Morgenstern, H. Confounding in health research. Annu. Rev. Public Health 22, 189–212 (2001).
Benner, J. S. et al. Long-term persistence in use of statin therapy in elderly patients. JAMA 288, 455–461 (2002).
Robins, J. M., Hernán, M. A. & Brumback, B. Marginal structural models and causal inference in epidemiology. Epidemiology 11, 550–560 (2000).
Hernán, M. A., Brumback, B. & Robins, J. M. Marginal structural models to estimate the causal effect of zidovudine on the survival of HIV-positive men. Epidemiology 11, 561–570 (2000).
Robins, J. M. in Statistical Models in Epidemiology: The Environment and Clinical Trials (eds Berry, D. & Halloran, M. E.) 95–134 (Springer-Verlag, 1999).
Acknowledgements
M.C.P. receives a grant from the National Institute on Aging (T32 AG027668). Funding was provided to M.C.P., J.W. and D.B. from the Alzheimer's Drug Discovery Foundation (ADDF). The ADDF catalyses and funds drug discovery and drug development for Alzheimer disease (AD) and related disorders. To learn more about the ADDF, visit the website at www.alzdiscovery.org. The funding agencies had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; or decision to submit the manuscript for publication. This work has not previously been presented in any form. However, the literature review and quality assessment were completed in parallel with work completed for the AlzRisk website (www.alzrisk.org), which attempts to catalogue epidemiological reports on risk factors for AD and to provide a continually updated, publically available assessment of the state of the literature. The AlzRisk database entry on statins, which is currently limited to studies reporting on AD as an end point, will be updated as new studies are published. We would like to thank John Jackson for his work in developing AlzRisk search strategies, which informed our search strategy, and his insight into the accuracy of electronic medical records in ascertaining dementia status.
Author information
Authors and Affiliations
Contributions
M.C.P. conducted the literature search, determined study eligibility, extracted the data, conducted the quality assessment, and drafted the manuscript. All authors made substantial contributions to the analysis and interpretation of the data and critically revised the manuscript for important intellectual content. All authors gave final approval for publication.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Box 1
Database search terms used for the literature search (DOCX 24 kb)
Supplementary Box 2
Details of data extraction from eligible articles (DOCX 22 kb)
Supplementary Box 3
Exploration of the potential magnitude of selection bias (DOCX 55 kb)
Supplementary Table 1
Summary of RCTs that reported on statins and cognitive outcomes according to the length of the follow-up period (DOCX 32 kb)
Supplementary Table 2
Summary of observational studies that reported on the association between baseline statin use and either cognitive change or incident dementia (DOCX 27 kb)
Supplementary Table 3
Summary of observational studies that reported on the association between time-updated statin use and incident dementia (DOCX 27 kb)
Supplementary Table 4
Summary of observational studies that considered participants' history of statin use during follow-up, and their cognitive outcomes (DOCX 29 kb)
Rights and permissions
About this article
Cite this article
Power, M., Weuve, J., Sharrett, A. et al. Statins, cognition, and dementia—systematic review and methodological commentary. Nat Rev Neurol 11, 220–229 (2015). https://doi.org/10.1038/nrneurol.2015.35
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrneurol.2015.35
This article is cited by
-
Statin use is associated with lower risk of dementia in stroke patients: a community-based cohort study with inverse probability weighted marginal structural model analysis
European Journal of Epidemiology (2022)
-
Ameliorative role of rolipram, PDE-4 inhibitor, against sodium arsenite–induced vascular dementia in rats
Environmental Science and Pollution Research (2021)
-
Preventing Diabetes and Atherosclerosis in the Cardiometabolic Syndrome
Current Atherosclerosis Reports (2021)
-
The Cognitive Effects of Statins are Modified by Age
Scientific Reports (2020)
-
Evidence for benefit of statins to modify cognitive decline and risk in Alzheimer’s disease
Alzheimer's Research & Therapy (2017)