Abstract
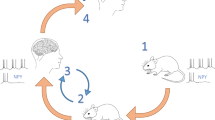
Epilepsy represents a major burden to society, not least because approximately 25% of patients do not respond satisfactorily to antiepileptic medication, and only a minority with pharmacoresistant epilepsy are eligible for potentially curative surgery. Several studies have explored gene therapy as a treatment strategy. The translation of scientific breakthroughs into the clinic faces several challenges, including the validation of experimental models of human pharmacoresistant epilepsy, establishment of sensitive and specific measures of therapeutic efficacy, and evaluation of the long-term safety of gene therapy. On the basis of successful reports of gene therapy in experimental models of epilepsy, a roadmap toward clinical trials is proposed.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Picot, M.-C., Baldy-Moulinier, M., Daurès, J.-P., Dujols, P. & Crespel, A. The prevalence of epilepsy and pharmacoresistant epilepsy in adults: a population-based study in a Western European country. Epilepsia 49, 1230–1238 (2008).
Kwan, P., Schachter, S. C. & Brodie, M. J. Drug-resistant epilepsy. N. Engl. J. Med. 365, 919–926 (2011).
Schuele, S. U. & Lüders, H. O. Intractable epilepsy: management and therapeutic alternatives. Lancet Neurol. 7, 514–524 (2008).
Kahane, P. & Depaulis, A. Deep brain stimulation in epilepsy: what is next? Curr. Opin. Neurol. 23, 177–182 (2010).
Van Dycke, A., Raedt, R., Vonck, K. & Boon, P. Local delivery strategies in epilepsy: a focus on adenosine. Seizure 20, 376–382 (2011).
Rothman, S. M. The therapeutic potential of focal cooling for neocortical epilepsy. Neurotherapeutics 6, 251–257 (2009).
Sebe, J. Y. & Baraban, S. C. The promise of an interneuron-based cell therapy for epilepsy. Dev. Neurobiol. 71, 107–117 (2011).
Simonato, M. et al. Progress in gene therapy for neurological disorders. Nat. Rev. Neurol. 9, 277–291 (2013).
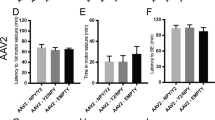
Richichi, C. et al. Anticonvulsant and antiepileptogenic effects mediated by adeno-associated virus vector neuropeptide Y expression in the rat hippocampus. J. Neurosci. 24, 3051–3059 (2004).
Noè, F. et al. Neuropeptide Y gene therapy decreases chronic spontaneous seizures in a rat model of temporal lobe epilepsy. Brain 131, 1506–1515 (2008).
Paradiso, B. et al. Localized delivery of fibroblast growth factor-2 and brain-derived neurotrophic factor reduces spontaneous seizures in an epilepsy model. Proc. Natl Acad. Sci. USA 106, 7191–7196 (2009).
Haberman, R. P., Samulski, R. J. & McCown, T. J. Attenuation of seizures and neuronal death by adeno-associated virus vector galanin expression and secretion. Nat. Med. 9, 1076–1080 (2003).
McCown, T. J. Adeno-associated virus-mediated expression and constitutive secretion of galanin suppresses limbic seizure activity in vivo. Mol. Ther. 14, 63–68 (2006).
Wykes, R. C. et al. Optogenetic and potassium channel gene therapy in a rodent model of focal neocortical epilepsy. Sci. Transl. Med. 4, 161ra152 (2012).
Kanter-Schlifke, I., Georgievska, B., Kirik, D. & Kokaia, M. Seizure suppression by GDNF gene therapy in animal models of epilepsy. Mol. Ther. 15, 1106–1113 (2007).
Woldbye, D. P. D. et al. Adeno-associated viral vector-induced overexpression of neuropeptide Y Y2 receptors in the hippocampus suppresses seizures. Brain 133, 2778–2788 (2010).
Paradiso, B. et al. Localized overexpression of FGF-2 and BDNF in hippocampus reduces mossy fiber sprouting and spontaneous seizures up to 4 weeks after pilocarpine-induced status epilepticus. Epilepsia 52, 572–578 (2011).
Bovolenta, R. et al. Hippocampal FGF-2 and BDNF overexpression attenuates epileptogenesis-associated neuroinflammation and reduces spontaneous recurrent seizures. J. Neuroinflammation 7, 81 (2010).
Raol, Y. H. et al. Enhancing GABAA receptor α1 subunit levels in hippocampal dentate gyrus inhibits epilepsy development in an animal model of temporal lobe epilepsy. J. Neurosci. 26, 11342–11346 (2006).
McClelland, S. et al. Neuron-restrictive silencer factor-mediated hyperpolarization-activated cyclic nucleotide gated channelopathy in experimental temporal lobe epilepsy. Ann. Neurol. 70, 454–464 (2011).
Jimenez-Mateos, E. M. et al. Silencing microRNA-134 produces neuroprotective and prolonged seizure-suppressive effects. Nat. Med. 18, 1087–1094 (2012).
Annegers, J. F., Hauser, W. A., Coan, S. P. & Rocca, W. A. A population-based study of seizures after traumatic brain injuries. N. Engl. J. Med. 338, 20–24 (1998).
Annegers, J. F., Hauser, W. A., Beghi, E., Nicolosi, A. & Kurland, L. T. The risk of unprovoked seizures after encephalitis and meningitis. Neurology 38, 1407–1410 (1988).
Burn, J. et al. Epileptic seizures after a first stroke: the Oxfordshire Community Stroke Project. BMJ 315, 1582–1587 (1997).
Galanopoulou, A. S. et al. Identification of new epilepsy treatments: issues in preclinical methodology. Epilepsia 53, 571–582 (2012).
Wilcox, K. S. et al. Issues related to development of new antiseizure treatments. Epilepsia 54 (Suppl. 4), 24–34 (2013).
Bien, C. G. et al. Trends in presurgical evaluation and surgical treatment of epilepsy at one centre from 1988–2009. J. Neurol. Neurosurg. Psychiatr. 84, 54–61 (2013).
De Tisi, J. et al. The long-term outcome of adult epilepsy surgery, patterns of seizure remission, and relapse: a cohort study. Lancet 378, 1388–1395 (2011).
Hermann, B. P., Wyler, A. R., Bush, A. J. & Tabatabai, F. R. Differential effects of left and right anterior temporal lobectomy on verbal learning and memory performance. Epilepsia 33, 289–297 (1992).
Kelly, K. M. et al. Photothrombotic brain infarction results in seizure activity in aging Fischer 344 and Sprague Dawley rats. Epilepsy Res. 47, 189–203 (2001).
Kharlamov, E. A., Jukkola, P. I., Schmitt, K. L. & Kelly, K. M. Electrobehavioral characteristics of epileptic rats following photothrombotic brain infarction. Epilepsy Res. 56, 185–203 (2003).
Paz, J. T. et al. Closed-loop optogenetic control of thalamus as a tool for interrupting seizures after cortical injury. Nat. Neurosci. 16, 64–70 (2013).
Whittington, M. A. & Jefferys, J. G. Epileptic activity outlasts disinhibition after intrahippocampal tetanus toxin in the rat. J. Physiol. (Lond.) 481, 593–604 (1994).
Mellanby, J., George, G., Robinson, A. & Thompson, P. Epileptiform syndrome in rats produced by injecting tetanus toxin into the hippocampus. J. Neurol. Neurosurg. Psychiatr. 40, 404–414 (1977).
Nilsen, K. E., Walker, M. C. & Cock, H. R. Characterization of the tetanus toxin model of refractory focal neocortical epilepsy in the rat. Epilepsia 46, 179–187 (2005).
Louis, E. D., Williamson, P. D. & Darcey, T. M. Chronic focal epilepsy induced by microinjection of tetanus toxin into the cat motor cortex. Electroencephalogr. Clin. Neurophysiol. 75, 548–557 (1990).
Hagemann, G., Hoeller, M., Bruehl, C., Lutzenburg, M. & Witte, O. W. Effects of tetanus toxin on functional inhibition after injection in separate cortical areas in rat. Brain Res. 818, 127–134 (1999).
Mainardi, M., Pietrasanta, M., Vannini, E., Rossetto, O. & Caleo, M. Tetanus neurotoxin-induced epilepsy in mouse visual cortex. Epilepsia 53, e132–e136 (2012).
Kullmann, D. M. Neurological channelopathies. Annu. Rev. Neurosci. 33, 151–172 (2010).
Lin, E.-J. D., Young, D., Baer, K., Herzog, H. & During, M. J. Differential actions of NPY on seizure modulation via Y1 and Y2 receptors: evidence from receptor knockout mice. Epilepsia 47, 773–780 (2006).
Boison, D. in Jasper's Basic Mechanisms of the Epilepsies (eds Noebels, J. L. et al.) (National Center for Biotechnology Information, 2012).
Haberman, R. et al. Therapeutic liabilities of in vivo viral vector tropism: adeno-associated virus vectors, NMDAR1 antisense, and focal seizure sensitivity. Mol. Ther. 6, 495–500 (2002).
Boison, D. Inhibitory RNA in epilepsy: research tools and therapeutic perspectives. Epilepsia 51, 1659–1668 (2010).
Mazzuferi, M. et al. Nrf2 defense pathway: Experimental evidence for its protective role in epilepsy. Ann. Neurol. 74, 560–568 (2013).
Tønnesen, J., Sørensen, A. T., Deisseroth, K., Lundberg, C. & Kokaia, M. Optogenetic control of epileptiform activity. Proc. Natl Acad. Sci. USA 106, 12162–12167 (2009).
Sukhotinsky, I. et al. Optogenetic delay of status epilepticus onset in an in vivo rodent epilepsy model. PLoS ONE 8, e62013 (2013).
Krook-Magnuson, E., Armstrong, C., Oijala, M. & Soltesz, I. On-demand optogenetic control of spontaneous seizures in temporal lobe epilepsy. Nat. Commun. 4, 1376 (2013).
Manfredsson, F. P. & Mandel, R. J. Development of gene therapy for neurological disorders. Discov. Med. 9, 204–211 (2010).
Gray, S. J. et al. Directed evolution of a novel adeno-associated virus (AAV) vector that crosses the seizure-compromised blood–brain barrier (BBB). Mol. Ther. 18, 570–578 (2010).
Abordo-Adesida, E. et al. Stability of lentiviral vector-mediated transgene expression in the brain in the presence of systemic antivector immune responses. Hum. Gene Ther. 16, 741–751 (2005).
Rodnitzky, R. L. Upcoming treatments in Parkinson's disease, including gene therapy. Parkinsonism Relat. Disord. 18 (Suppl. 1), S37–S40 (2012).
Rahim, A. A. et al. Efficient gene delivery to the adult and fetal CNS using pseudotyped non-integrating lentiviral vectors. Gene Ther. 16, 509–520 (2009).
Bartlett, D. L. et al. Oncolytic viruses as therapeutic cancer vaccines. Mol. Cancer 12, 103 (2013).
Binnie, C. D. Cognitive impairment during epileptiform discharges: is it ever justifiable to treat the EEG? Lancet Neurol. 2, 725–730 (2003).
Noebels, J. A perfect storm: converging paths of epilepsy and Alzheimer's dementia intersect in the hippocampal formation. Epilepsia 52 (Suppl. 1), 39–46 (2011).
Mavilio, F. Gene therapies need new development models. Nature 490, 7 (2012).
Wilson, J. M. Moving to the clinic with gene therapy through our new journal expansion, human gene therapy clinical development. Hum. Gene Ther. 23, 1029–1030 (2012).
NGVB National Gene Vector Biorepository [online], (2014).
Tremblay, J. P. et al. Translating the genomics revolution: the need for an international gene therapy consortium for monogenic diseases. Mol. Ther. 21, 266–268 (2013).
Lerchner, W., Corgiat, B., Der Minassian, V., Saunders, R. C. & Richmond, B. J. Injection parameters and virus dependent choice of promoters to improve neuron targeting in the nonhuman primate brain. Gene Ther. http://dx.doi.org/10.1038/gt.2013.75.
Acknowledgements
Work in the authors' laboratories is supported by the Wellcome Trust, Medical Research Council, European Research Council and the Royal Society.
Author information
Authors and Affiliations
Contributions
All authors researched the data for the article, provided substantial contributions to discussions of its content, wrote the article and undertook review and/or editing of the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
PowerPoint slides
Rights and permissions
About this article
Cite this article
Kullmann, D., Schorge, S., Walker, M. et al. Gene therapy in epilepsy—is it time for clinical trials?. Nat Rev Neurol 10, 300–304 (2014). https://doi.org/10.1038/nrneurol.2014.43
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrneurol.2014.43
This article is cited by
-
Identification of potential crucial genes and therapeutic targets for epilepsy
European Journal of Medical Research (2024)
-
Autologous olfactory mucosa mesenchymal stem cells treatment improves the neural network in chronic refractory epilepsy
Stem Cell Research & Therapy (2023)
-
Combinatorial gene therapy for epilepsy: Gene sequence positioning and AAV serotype influence expression and inhibitory effect on seizures
Gene Therapy (2023)
-
Activated astrocytes attenuate neocortical seizures in rodent models through driving Na+-K+-ATPase
Nature Communications (2022)
-
Integrative Analyses Identify KCNJ15 as a Candidate Gene in Patients with Epilepsy
Neurology and Therapy (2022)