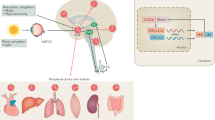
Key Points
-
Sleep and circadian disruption are common in Alzheimer disease, Parkinson disease and Huntington disease
-
Symptoms of circadian disruption range from subjective sleep complaints and alterations of sleep timing to severe disruptions in rest–activity cycles and sleep architecture
-
Behavioural, physiological and molecular markers of circadian system function suggest progressive deterioration of circadian rhythmicity as the disease progresses
-
The relationship between circadian dysfunction and neurodegenerative processes might be bidirectional: circadian dysfunction might exacerbate the disease processes
-
Behavioural or pharmaceutical interventions that target sleep–wake cycles might reverse circadian disruption
Abstract
Circadian rhythms are physiological and behavioural cycles generated by an endogenous biological clock, the suprachiasmatic nucleus. The circadian system influences the majority of physiological processes, including sleep–wake homeostasis. Impaired sleep and alertness are common symptoms of neurodegenerative disorders, and circadian dysfunction might exacerbate the disease process. The pathophysiology of sleep–wake disturbances in these disorders remains largely unknown, and is presumably multifactorial. Circadian rhythm dysfunction is often observed in patients with Alzheimer disease, in whom it has a major impact on quality of life and represents one of the most important factors leading to institutionalization of patients. Similarly, sleep and circadian problems represent common nonmotor features of Parkinson disease and Huntington disease. Clinical studies and experiments in animal models of neurodegenerative disorders have revealed the progressive nature of circadian dysfunction throughout the course of neurodegeneration, and suggest strategies for the restoration of circadian rhythmicity involving behavioural and pharmacological interventions that target the sleep–wake cycle. In this Review, we discuss the role of the circadian system in the regulation of the sleep–wake cycle, and outline the implications of disrupted circadian timekeeping in neurodegenerative diseases.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Mohawk, J. A., Green, C. B. & Takahashi, J. S. Central and peripheral circadian clocks in mammals. Annu. Rev. Neurosci. 35, 445–462 (2012).
Swaab, D. F., Fliers, E. & Partiman, T. S. The suprachiasmatic nucleus of the human brain in relation to sex, age and senile dementia. Brain Res. 342, 37–44 (1985).
Cassone, V. M., Speh, J. C., Card, J. P. & Moore, R. Y. Comparative anatomy of the mammalian hypothalamic suprachiasmatic nucleus. J. Biol. Rhythms 3, 71–91 (1988).
Golombek, D. A. & Rosenstein, R. E. Physiology of circadian entrainment. Physiol. Rev. 90, 1063–1102 (2010).
Dardente, H. & Cermakian, N. Molecular circadian rhythms in central and peripheral clocks in mammals. Chronobiol. Int. 24, 195–213 (2007).
Moore, R. Y. The suprachiasmatic nucleus and the circadian timing system. Prog. Mol. Biol. Transl. Sci. 119, 1–28 (2013).
Ballard, C. et al. Alzheimer's disease. Lancet 377, 1019–1031 (2011).
Ittner, L. M. & Götz, J. Amyloid-β and tau-—a toxic pas de deux in Alzheimer's disease. Nat. Rev. Neurosci. 12, 65–72 (2011).
Braak, H. & Braak, E. Neuropathological stageing of Alzheimer-related changes. Acta Neuropathol. 82, 239–259 (1991).
Bianchetti, A. et al. Predictors of mortality and institutionalization in Alzheimer disease patients 1 year after discharge from an Alzheimer dementia unit. Dementia 6, 108–112 (1995).
Pollak, C. P. & Perlick, D. Sleep problems and institutionalization of the elderly. J. Geriatr. Psychiatry Neurol. 4, 204–210 (1991).
Harper, D. G. et al. Dementia severity and Lewy bodies affect circadian rhythms in Alzheimer disease. Neurobiol. Aging 25, 771–781 (2004).
Satlin, A., Volicer, L., Stopa, E. G. & Harper, D. Circadian locomotor activity and core-body temperature rhythms in Alzheimer's disease. Neurobiol. Aging 16, 765–771 (1995).
Harper, D. G. et al. Disturbance of endogenous circadian rhythm in aging and Alzheimer disease. Am. J. Geriatr Psychiatry 13, 359–368 (2005).
Most, E. I., Scheltens, P. & Van Someren, E. J. Increased skin temperature in Alzheimer's disease is associated with sleepiness. J. Neural Transm. 119, 1185–1194 (2012).
Witting, W., Kwa, I. H., Eikelenboom, P., Mirmiran, M. & Swaab, D. F. Alterations in the circadian rest–activity rhythm in aging and Alzheimer's disease. Biol. Psychiatry 27, 563–572 (1990).
van Someren, E. J. et al. Circadian rest–activity rhythm disturbances in Alzheimer's disease. Biol. Psychiatry 40, 259–270 (1996).
Tranah, G. J. et al. Circadian activity rhythms and risk of incident dementia and mild cognitive impairment in older women. Ann. Neurol. 70, 722–732 (2011).
Hatfield, C. F., Herbert, J., van Someren, E. J., Hodges, J. R. & Hastings, M. H. Disrupted daily activity/rest cycles in relation to daily cortisol rhythms of home-dwelling patients with early Alzheimer's dementia. Brain 127, 1061–1074 (2004).
Manconi, M., Hutchins, W., Feroah, T. R., Zucconi, M. & Ferini-Strambi, L. On the pathway of an animal model for restless legs syndrome. Neurol. Sci. 28 (Suppl. 1), S53–S60 (2007).
Hu, K., Van Someren, E. J., Shea, S. A. & Scheer, F. A. Reduction of scale invariance of activity fluctuations with aging and Alzheimer's disease: Involvement of the circadian pacemaker. Proc. Natl Acad. Sci. USA 106, 2490–2494 (2009).
Skene, D. J. & Swaab, D. F. Melatonin rhythmicity: effect of age and Alzheimer's disease. Exp. Gerontol. 38, 199–206 (2003).
Rosales-Corral, S. A. et al. Alzheimer's disease: pathological mechanisms and the beneficial role of melatonin. J. Pineal Res. 52, 167–202 (2012).
Lin, L. et al. Melatonin in Alzheimer's disease. Int. J. Mol. Sci. 14, 14575–14593 (2013).
Liu, R. Y., Zhou, J. N., van Heerikhuize, J., Hofman, M. A. & Swaab, D. F. Decreased melatonin levels in postmortem cerebrospinal fluid in relation to aging, Alzheimer's disease, and apolipoprotein E-ε4/4 genotype. J. Clin. Endocrinol. Metab. 84, 323–327 (1999).
Zhou, J. N., Liu, R. Y., Kamphorst, W., Hofman, M. A. & Swaab, D. F. Early neuropathological Alzheimer's changes in aged individuals are accompanied by decreased cerebrospinal fluid melatonin levels. J. Pineal Res. 35, 125–130 (2003).
Wu, Y. H. et al. Molecular changes underlying reduced pineal melatonin levels in Alzheimer disease: alterations in preclinical and clinical stages. J. Clin. Endocrinol. Metab. 88, 5898–5906 (2003).
Mishima, K. et al. Melatonin secretion rhythm disorders in patients with senile dementia of Alzheimer's type with disturbed sleep–waking. Biol. Psychiatry 45, 417–421 (1999).
Shan, L., Bossers, K., Unmehopa, U., Bao, A. M. & Swaab, D. F. Alterations in the histaminergic system in Alzheimer's disease: a postmortem study. Neurobiol. Aging 33, 2585–2598 (2012).
Shan, L. et al. Diurnal fluctuation in histidine decarboxylase expression, the rate limiting enzyme for histamine production, and its disorder in neurodegenerative diseases. Sleep 35, 713–715 (2012).
Wu, Y. H., Zhou, J. N., Van Heerikhuize, J., Jockers, R. & Swaab, D. F. Decreased MT1 melatonin receptor expression in the suprachiasmatic nucleus in aging and Alzheimer's disease. Neurobiol. Aging 28, 1239–1247 (2007).
Stopa, E. G. et al. Pathologic evaluation of the human suprachiasmatic nucleus in severe dementia. J. Neuropathol. Exp. Neurol. 58, 29–39 (1999).
Coogan, A. N., Rawlings, N., Luckman, S. M. & Piggins, H. D. Effects of neurotensin on discharge rates of rat suprachiasmatic nucleus neurons in vitro. Neuroscience 103, 663–672 (2001).
Zhou, J. N., Hofman, M. A. & Swaab, D. F. VIP neurons in the human SCN in relation to sex, age, and Alzheimer's disease. Neurobiol. Aging 16, 571–576 (1995).
Liu, R. Y. et al. Decreased vasopressin gene expression in the biological clock of Alzheimer disease patients with and without depression. J. Neuropathol. Exp. Neurol. 59, 314–322 (2000).
Coogan, A. N. et al. The circadian system in Alzheimer's disease: disturbances, mechanisms, and opportunities. Biol. Psychiatry 74, 333–339 (2013).
Friedland, R. P., Luxenberg, J. S. & Koss, E. A quantitative study of intracranial calcification in dementia of the Alzheimer type. Int. Psychogeriatr. 2, 36–43 (1990).
Wu, Y. H. et al. Pineal clock gene oscillation is disturbed in Alzheimer's disease, due to functional disconnection from the “master clock”. FASEB J. 20, 1874–1876 (2006).
Cermakian, N., Lamont, E. W., Boudreau, P. & Boivin, D. B. Circadian clock gene expression in brain regions of Alzheimer's disease patients and control subjects. J. Biol. Rhythms 26, 160–170 (2011).
Sterniczuk, R., Dyck, R. H., Laferla, F. M. & Antle, M. C. Characterization of the 3xTg-AD mouse model of Alzheimer's disease: part 1. Circadian changes. Brain Res. 1348, 139–148 (2010).
Knight, E. M. et al. Age-related changes in core body temperature and activity in triple-transgenic Alzheimer's disease (3xTgAD) mice. Dis. Model Mech. 6, 160–170 (2013).
Gorman, M. R. & Yellon, S. Lifespan daily locomotor activity rhythms in a mouse model of amyloid-induced neuropathology. Chronobiol. Int. 27, 1159–1177 (2010).
Wisor, J. P. et al. Sleep and circadian abnormalities in a transgenic mouse model of Alzheimer's disease: a role for cholinergic transmission. Neuroscience 131, 375–385 (2005).
Ambree, O. et al. Activity changes and marked stereotypic behavior precede Aβ pathology in TgCRND8 Alzheimer mice. Neurobiol. Aging 27, 955–964 (2006).
Slats, D., Claassen, J. A., Verbeek, M. M. & Overeem, S. Reciprocal interactions between sleep, circadian rhythms and Alzheimer's disease: focus on the role of hypocretin and melatonin. Ageing Res. Rev. 12, 188–200 (2013).
Wang, J. Z. & Wang, Z. F. Role of melatonin in Alzheimer-like neurodegeneration. Acta Pharmacol. Sin. 27, 41–49 (2006).
Ju, Y. E., Lucey, B. P. & Holtzman, D. M. Sleep and Alzheimer disease pathology—a bidirectional relationship. Nat. Rev. Neurol. 10, 115–119 (2014).
Kang, J. E. et al. Amyloid-β dynamics are regulated by orexin and the sleep–wake cycle. Science 326, 1005–1007 (2009).
Xie, L. et al. Sleep drives metabolite clearance from the adult brain. Science 342, 373–377 (2013).
Ooms, S. et al. Effect of 1 night of total sleep deprivation on cerebrospinal fluid β-amyloid 42 in healthy middle-aged men: a randomized clinical trial. JAMA Neurol. 71, 971–977 (2014).
Pappolla, M. et al. Inhibition of Alzheimer β-fibrillogenesis by melatonin. J. Biol. Chem. 273, 7185–7188 (1998).
Lahiri, D. K., Chen, D., Ge, Y. W., Bondy, S. C. & Sharman, E. H. Dietary supplementation with melatonin reduces levels of amyloid beta-peptides in the murine cerebral cortex. J. Pineal Res. 36, 224–231 (2004).
Matsubara, E. et al. Melatonin increases survival and inhibits oxidative and amyloid pathology in a transgenic model of Alzheimer's disease. J. Neurochem. 85, 1101–1108 (2003).
Asayama, K. et al. Double blind study of melatonin effects on the sleep–wake rhythm, cognitive and non-cognitive functions in Alzheimer type dementia. J. Nippon Med. Sch. 70, 334–341 (2003).
Cardinali, D. P., Furio, A. M. & Brusco, L. I. Clinical aspects of melatonin intervention in Alzheimer's disease progression. Curr. Neuropharmacol. 8, 218–227 (2010).
Dowling, G. A. et al. Melatonin and bright-light treatment for rest–activity disruption in institutionalized patients with Alzheimer's disease. J. Am. Geriatr Soc. 56, 239–246 (2008).
Mahlberg, R. & Walther, S. Actigraphy in agitated patients with dementia. Monitoring treatment outcomes. Z. Gerontol. Geriatr. 40, 178–184 (2007).
Singer, C. et al. A multicenter, placebo-controlled trial of melatonin for sleep disturbance in Alzheimer's disease. Sleep 26, 893–901 (2003).
Gehrman, P. R. et al. Melatonin fails to improve sleep or agitation in double-blind randomized placebo-controlled trial of institutionalized patients with Alzheimer disease. Am. J. Geriatr Psychiatry 17, 166–169 (2009).
Jansen, S. L., Forbes, D. A., Duncan, V. & Morgan, D. G. Melatonin for cognitive impairment. Cochrane Database of Systematic Reviews, Issue 1. Art. No.: CD003802. http://dx.doi.org/10.1002/14651858.CD003802.pub3.
Lyketsos, C. G., Lindell Veiel, L., Baker, A. & Steele, C. A randomized, controlled trial of bright light therapy for agitated behaviors in dementia patients residing in long-term care. Int. J. Geriatr Psychiatry 14, 520–525 (1999).
Ancoli-Israel, S. et al. Increased light exposure consolidates sleep and strengthens circadian rhythms in severe Alzheimer's disease patients. Behav. Sleep Med. 1, 22–36 (2003).
Burns, A., Allen, H., Tomenson, B., Duignan, D. & Byrne, J. Bright light therapy for agitation in dementia: a randomized controlled trial. Int. Psychogeriatr. 21, 711–721 (2009).
McCurry, S. M. et al. Increasing walking and bright light exposure to improve sleep in community-dwelling persons with Alzheimer's disease: results of a randomized, controlled trial. J. Am. Geriatr. Soc. 59, 1393–1402 (2011).
Dowling, G. A. et al. Effect of morning bright light treatment for rest–activity disruption in institutionalized patients with severe Alzheimer's disease. Int. Psychogeriatr. 17, 221–236 (2005).
Dowling, G. A., Mastick, J., Hubbard, E. M., Luxenberg, J. S. & Burr, R. L. Effect of timed bright light treatment for rest–activity disruption in institutionalized patients with Alzheimer's disease. Int. J. Geriatr. Psychiatry 20, 738–743 (2005).
Alves, G., Forsaa, E. B., Pedersen, K. F., Dreetz Gjerstad, M. & Larsen, J. P. Epidemiology of Parkinson's disease. J. Neurol. 255 (Suppl. 5), 18–32 (2008).
Dorsey, E. R. et al. Projected number of people with Parkinson disease in the most populous nations, 2005 through 2030. Neurology 68, 384–386 (2007).
Bruguerolle, B. & Simon, N. Biologic rhythms and Parkinson's disease: a chronopharmacologic approach to considering fluctuations in function. Clin. Neuropharmacol. 25, 194–201 (2002).
Willis, G. L. Parkinson's disease as a neuroendocrine disorder of circadian function: dopamine-melatonin imbalance and the visual system in the genesis and progression of the degenerative process. Rev. Neurosci. 19, 245–316 (2008).
Videnovic, A. & Golombek, D. Circadian and sleep disorders in Parkinson's disease. Exp. Neurol. 243, 45–56 (2013).
Bonuccelli, U. et al. Diurnal motor variations to repeated doses of levodopa in Parkinson's disease. Clin. Neuropharmacol. 23, 28–33 (2000).
Nutt, J. G., Woodward, W. R., Carter, J. H. & Trotman, T. L. Influence of fluctuations of plasma large neutral amino acids with normal diets on the clinical response to levodopa. J. Neurol. Neurosurg. Psychiatry 52, 481–487 (1989).
van Hilten, J. J. et al. Assessment of response fluctuations in Parkinson's disease by ambulatory wrist activity monitoring. Acta Neurol. Scand. 87, 171–177 (1993).
van Hilten, J. J., Middelkoop, H. A., Kerkhof, G. A. & Roos, R. A. A new approach in the assessment of motor activity in Parkinson's disease. J. Neurol. Neurosurg. Psychiatry 54, 976–979 (1991).
Mihci, E., Kardelen, F., Dora, B. & Balkan, S. Orthostatic heart rate variability analysis in idiopathic Parkinson's disease. Acta Neurol. Scand. 113, 288–293 (2006).
Pathak, A. & Senard, J. M. Blood pressure disorders during Parkinson's disease: epidemiology, pathophysiology and management. Expert Rev. Neurother. 6, 1173–1180 (2006).
Pursiainen, V. et al. Circadian heart rate variability in Parkinson's disease. J. Neurol. 249, 1535–1540 (2002).
Comella, C. L. Sleep disorders in Parkinson's disease: an overview. Mov Disord. 22 (Suppl. 17), S367–S373 (2007).
Porter, B., Macfarlane, R. & Walker, R. The frequency and nature of sleep disorders in a community-based population of patients with Parkinson's disease. Eur. J. Neurol. 15, 50–54 (2008).
Verbaan, D., van Rooden, S. M., Visser, M., Marinus, J. & van Hilten, J. J. Nighttime sleep problems and daytime sleepiness in Parkinson's disease. Mov. Disord. 23, 35–41 (2008).
Struck, L. K., Rodnitzky, R. L. & Dobson, J. K. Circadian fluctuations of contrast sensitivity in Parkinson's disease. Neurology 40, 467–470 (1990).
Piccini, P. et al. Diurnal worsening in Parkinson patients treated with levodopa [Italian]. Riv. Neurol. 61, 219–224 (1991).
van Hilten, B. et al. Sleep disruption in Parkinson's disease. Assessment by continuous activity monitoring. Arch. Neurol. 51, 922–928 (1994).
Whitehead, D. L., Davies, A. D., Playfer, J. R. & Turnbull, C. J. Circadian rest–activity rhythm is altered in Parkinson's disease patients with hallucinations. Mov. Disord. 23, 1137–1145 (2008).
Kallio, M. et al. Heart rate variability in patients with untreated Parkinson's disease. Eur. J. Neurol. 7, 667–672 (2000).
Senard, J. M., Chamontin, B., Rascol, A. & Montastruc, J. L. Ambulatory blood pressure in patients with Parkinson's disease without and with orthostatic hypotension. Clin. Auton. Res. 2, 99–104 (1992).
Devos, D. et al. Heart rate variability and Parkinson's disease severity. J. Neural Transm. 110, 997–1011 (2003).
Wakabayashi, K. & Takahashi, H. Neuropathology of autonomic nervous system in Parkinson's disease. Eur. Neurol. 38 (Suppl. 2), 2–7 (1997).
Langston, J. The hypothalamus in Parkinson's disease. Ann. Neurol. 3, 129–133 (1978).
Wirz-Justice, A., Da Prada, M. & Reme, C. Circadian rhythm in rat retinal dopamine. Neurosci. Lett. 45, 21–25 (1984).
Dearry, A. & Burnside, B. Dopaminergic regulation of cone retinomotor movement in isolated teleost retinas: I. Induction of cone contraction is mediated by D2 receptors. J. Neurochem. 46, 1006–1021 (1986).
Raggi, A., Bella, R., Pennisi, G., Neri, W. & Ferri, R. Sleep disorders in Parkinson's disease: a narrative review of the literature. Rev. Neurosci. 24, 279–291 (2013).
Braak, H. & Del Tredici, K. Invited Article: Nervous system pathology in sporadic Parkinson disease. Neurology 70, 1916–1925 (2008).
Bordet, R. et al. Study of circadian melatonin secretion pattern at different stages of Parkinson's disease. Clin. Neuropharmacol. 26, 65–72 (2003).
Fertl, E., Auff, E., Doppelbauer, A. & Waldhauser, F. Circadian secretion pattern of melatonin in de novo parkinsonian patients: evidence for phase-shifting properties of l-dopa. J. Neural Transm. Park. Dis. Dement. Sect. 5, 227–234 (1993).
Hartmann, A., Veldhuis, J. D., Deuschle, M., Standhardt, H. & Heuser, I. Twenty-four hour cortisol release profiles in patients with Alzheimer's and Parkinson's disease compared to normal controls: ultradian secretory pulsatility and diurnal variation. Neurobiol. Aging 18, 285–289 (1997).
Aziz, N. A., Pijl, H., Frolich, M., Roelfsema, F. & Roos, R. A. Diurnal secretion profiles of growth hormone, thyrotrophin and prolactin in Parkinson's disease. J. Neuroendocrinol. 23, 519–524 (2011).
Pierangeli, G. et al. Nocturnal body core temperature falls in Parkinson's disease but not in multiple-system atrophy. Mov. Disord. 16, 226–232 (2001).
Cagnacci, A. et al. Effect of naloxone on body temperature in postmenopausal women with Parkinson's disease. Life Sci. 46, 1241–1247 (1990).
Suzuki, K. et al. Circadian variation of core body temperature in Parkinson disease patients with depression: a potential biological marker for depression in Parkinson disease. Neuropsychobiology 56, 172–179 (2007).
Videnovic, A. et al. Circadian melatonin rhythm and excessive daytime sleepiness in Parkinson disease. JAMA Neurol. 71, 463–469 (2014).
Breen, D. P. et al. Sleep and circadian rhythm regulation in early Parkinson disease. JAMA Neurol. 71, 589–595 (2014).
Bolitho, S. J. et al. Disturbances in melatonin secretion and circadian sleep–wake regulation in Parkinson disease. Sleep Med. 15, 342–347 (2014).
Boivin, D. B. et al. Circadian clock genes oscillate in human peripheral blood mononuclear cells. Blood 102, 4143–4145 (2003).
Takimoto, M. et al. Daily expression of clock genes in whole blood cells in healthy subjects and a patient with circadian rhythm sleep disorder. Am. J. Physiol. Regul. Integr. Comp. Physiol. 289, R1273–R1279 (2005).
Teboul, M. et al. Atypical patterns of circadian clock gene expression in human peripheral blood mononuclear cells. J. Mol. Med. 83, 693–699 (2005).
Cai, Y., Liu, S., Sothern, R. B., Xu, S. & Chan, P. Expression of clock genes Per1 and Bmal1 in total leukocytes in health and Parkinson's disease. Eur. J. Neurol. 17, 550–554 (2009).
Witkovsky, P. Dopamine and retinal function. Doc. Ophthalmol. 108, 17–40 (2004).
Harrell, L. E. & Balagura, S. The effects of dark and light on the functional recovery following lateral hypothalamic lesions. Life Sci. 15, 2079–2087 (1974).
Willis, G. L. & Turner, E. J. Primary and secondary features of Parkinson's disease improve with strategic exposure to bright light: a case series study. Chronobiol. Int. 24, 521–537 (2007).
Willis, G. L., Moore, C. & Armstrong, S. M. A historical justification for and retrospective analysis of the systematic application of light therapy in Parkinson's disease. Rev. Neurosci. 23, 199–226 (2012).
Paus, S. et al. Bright light therapy in Parkinson's disease: a pilot study. Mov. Disord. 22, 1495–1498 (2007).
Barraud, Q. et al. Sleep disorders in Parkinson's disease: the contribution of the MPTP non-human primate model. Exp. Neurol. 219, 574–582 (2009).
Fifel, K. et al. Alteration of daily and circadian rhythms following dopamine depletion in MPTP treated non-human primates. PLoS ONE 9, e86240 (2014).
Vezoli, J. et al. Early presymptomatic and long-term changes of rest activity cycles and cognitive behavior in a MPTP-monkey model of Parkinson's disease. PLoS ONE 6, e23952 (2011).
Ben, V. & Bruguerolle, B. Effects of bilateral striatal 6-OHDA lesions on circadian rhythms in the rat: a radiotelemetric study. Life Sci. 67, 1549–1558 (2000).
Isobe, Y. & Nishino, H. Circadian rhythm of drinking and running-wheel activity in rats with 6-hydroxydopamine lesions of the ventral tegmental area. Brain Res. 899, 187–192 (2001).
Baier, P. C. et al. Circadian distribution of motor-activity in unilaterally 6-hydroxy-dopamine lesioned rats. Exp. Brain Res. 169, 283–288 (2006).
Gravotta, L., Gavrila, A. M., Hood, S. & Amir, S. Global depletion of dopamine using intracerebroventricular 6-hydroxydopamine injection disrupts normal circadian wheel-running patterns and PERIOD2 expression in the rat forebrain. J. Mol. Neurosci. 45, 162–171 (2011).
Kudo, T., Loh, D. H., Truong, D., Wu, Y. & Colwell, C. S. Circadian dysfunction in a mouse model of Parkinson's disease. Exp. Neurol. 232, 66–75 (2011).
Willison, L. D., Kudo, T., Loh, D. H., Kuljis, D. & Colwell, C. S. Circadian dysfunction may be a key component of the non-motor symptoms of Parkinson's disease: insights from a transgenic mouse model. Exp. Neurol. 243, 57–66 (2013).
Morrison, P. J. Accurate prevalence and uptake of testing for Huntington's disease. Lancet Neurol. 9, 1147 (2010).
Harper, P. S. The epidemiology of Huntington's disease. Hum. Genet. 89, 365–376 (1992).
Snell, R. G. et al. Relationship between trinucleotide repeat expansion and phenotypic variation in Huntington's disease. Nat. Genet. 4, 393–397 (1993).
Gusella, J. F. et al. A polymorphic DNA marker genetically linked to Huntington's disease. Nature 306, 234–238 (1983).
Gilliam, T. C. et al. A DNA segment encoding two genes very tightly linked to Huntington's disease. Science 238, 950–952 (1987).
[No authors listed] A novel gene containing a trinucleotide repeat that is expanded and unstable on Huntington's disease chromosomes. The Huntington's Disease Collaborative Research Group. Cell 72, 971–983 (1993).
Bamford, K. A., Caine, E. D., Kido, D. K., Cox, C. & Shoulson, I. A prospective evaluation of cognitive decline in early Huntington's disease: functional and radiographic correlates. Neurology 45, 1867–1873 (1995).
Paulsen, J. S., Ready, R. E., Hamilton, J. M., Mega, M. S. & Cummings, J. L. Neuropsychiatric aspects of Huntington's disease. J. Neurol. Neurosurg. Psychiatry 71, 310–314 (2001).
Stoy, N. & McKay, E. Weight loss in Huntington's disease. Ann. Neurol. 48, 130–131 (2000).
Goodman, A. O. et al. Asymptomatic sleep abnormalities are a common early feature in patients with Huntington's disease. Curr. Neurol. Neurosci. Rep. 11, 211–217 (2011).
Hansotia, P., Wall, R. & Berendes, J. Sleep disturbances and severity of Huntington's disease. Neurology 35, 1672–1674 (1985).
Emser, W., Brenner, M., Stober, T. & Schimrigk, K. Changes in nocturnal sleep in Huntington's and Parkinson's disease. J. Neurol. 235, 177–179 (1988).
Silvestri, R. et al. Sleep features in Tourette's syndrome, neuroacanthocytosis and Huntington's chorea. Neurophysiol. Clin. 25, 66–77 (1995).
Cuturic, M., Abramson, R. K., Vallini, D., Frank, E. M. & Shamsnia, M. Sleep patterns in patients with Huntington's disease and their unaffected first-degree relatives: a brief report. Behav. Sleep Med. 7, 245–254 (2009).
Arnulf, I. et al. Rapid eye movement sleep disturbances in Huntington disease. Arch. Neurol. 65, 482–488 (2008).
Morton, A. J. et al. Disintegration of the sleep–wake cycle and circadian timing in Huntington's disease. J. Neurosci. 25, 157–163 (2005).
Kudo, T. et al. Dysfunctions in circadian behavior and physiology in mouse models of Huntington's disease. Exp. Neurol. 228, 80–90 (2011).
Lazar, A. S. et al. Sleep, diurnal preference, health, and psychological well-being: a prospective single-allelic-variation study. Chronobiol. Int. 29, 131–146 (2012).
Epping, E. A. & Paulsen, J. S. Depression in the early stages of Huntington disease. Neurodegener. Dis. Manag. 1, 407–414 (2011).
Goodman, A. O., Morton, A. J. & Barker, R. A. Identifying sleep disturbances in Huntington's disease using a simple disease-focused questionnaire. PLoS Curr. 2, RRN1189 (2010).
Aziz, N. A., Anguelova, G. V., Marinus, J., Lammers, G. J. & Roos, R. A. Sleep and circadian rhythm alterations correlate with depression and cognitive impairment in Huntington's disease. Parkinsonism Relat. Disord. 16, 345–350 (2010).
Taylor, N. & Bramble, D. Sleep disturbance and Huntingdon's disease. Br. J. Psychiatry 171, 393 (1997).
Videnovic, A., Leurgans, S., Fan, W., Jaglin, J. & Shannon, K. M. Daytime somnolence and nocturnal sleep disturbances in Huntington disease. Parkinsonism Relat. Disord. 15, 471–474 (2009).
Starr, A. A disorder of rapid eye movements in Huntington's chorea. Brain 90, 545–564 (1967).
Wiegand, M. et al. Nocturnal sleep in Huntington's disease. J. Neurol. 238, 203–208 (1991).
van Vugt, J. P., van Hilten, B. J. & Roos, R. A. Hypokinesia in Huntington's disease. Mov. Disord. 11, 384–388 (1996).
van Vugt, J. P. et al. Quantitative assessment of daytime motor activity provides a responsive measure of functional decline in patients with Huntington's disease. Mov. Disord. 16, 481–488 (2001).
Hurelbrink, C. B., Lewis, S. J. & Barker, R. A. The use of the Actiwatch-Neurologica system to objectively assess the involuntary movements and sleep–wake activity in patients with mild-moderate Huntington's disease. J. Neurol. 252, 642–647 (2005).
Pouladi, M. A., Morton, A. J. & Hayden, M. R. Choosing an animal model for the study of Huntington's disease. Nat. Rev. Neurosci. 14, 708–721 (2013).
Shirbin, C. A. et al. Cortisol and depression in pre-diagnosed and early stage Huntington's disease. Psychoneuroendocrinology 38, 2439–2447 (2013).
Kantor, S., Szabo, L., Varga, J., Cuesta, M. & Morton, A. J. Progressive sleep and electroencephalogram changes in mice carrying the Huntington's disease mutation. Brain 136, 2147–2158 (2013).
Fisher, S. P. et al. Longitudinal analysis of the electroencephalogram and sleep phenotype in the R6/2 mouse model of Huntington's disease. Brain 136, 2159–2172 (2013).
Fahrenkrug, J., Popovic, N., Georg, B., Brundin, P. & Hannibal, J. Decreased VIP and VPAC2 receptor expression in the biological clock of the R6/2 Huntington's disease mouse. J. Mol. Neurosci. 31, 139–148 (2007).
Rudenko, O., Tkach, V., Berezin, V. & Bock, E. Detection of early behavioral markers of Huntington's disease in R6/2 mice employing an automated social home cage. Behav. Brain Res. 203, 188–199 (2009).
Bode, F. J. et al. Increased numbers of motor activity peaks during light cycle are associated with reductions in adrenergic α-receptor levels in a transgenic Huntington's disease rat model. Behav. Brain Res. 205, 175–182 (2009).
Pietropaolo, S., Delage, P., Cayzac, S., Crusio, W. E. & Cho, Y. H. Sex-dependent changes in social behaviors in motor pre-symptomatic R6/1 mice. PLoS ONE 6, e19965 (2011).
Loh, D. H., Kudo, T., Truong, D., Wu, Y. & Colwell, C. S. The Q175 mouse model of Huntington's disease shows gene dosage- and age-related decline in circadian rhythms of activity and sleep. PLoS ONE 8, e69993 (2013).
Heuser, I. J., Chase, T. N. & Mouradian, M. M. The limbic-hypothalamic-pituitary-adrenal axis in Huntington's disease. Biol. Psychiatry 30, 943–952 (1991).
Pallier, P. N. et al. Pharmacological imposition of sleep slows cognitive decline and reverses dysregulation of circadian gene expression in a transgenic mouse model of Huntington's disease. J. Neurosci. 27, 7869–7878 (2007).
Pallier, P. N. & Morton, A. J. Management of sleep/wake cycles improves cognitive function in a transgenic mouse model of Huntington's disease. Brain Res. 1279, 90–98 (2009).
Wood, N. I. et al. Responses to environmental enrichment differ with sex and genotype in a transgenic mouse model of Huntington's disease. PLoS ONE 5, e9077 (2010).
Aziz, N. A. et al. Delayed onset of the diurnal melatonin rise in patients with Huntington's disease. J. Neurol. 256, 1961–1965 (2009).
Kalliolia, E. et al. Plasma melatonin is reduced in Huntington's disease. Mov. Disord. (2014).
Saleh, N. et al. Neuroendocrine disturbances in Huntington's disease. PLoS ONE 4, e4962 (2009).
Aziz, N. A. et al. Increased hypothalamic–pituitary–adrenal axis activity in Huntington's disease. J. Clin. Endocrinol. Metab. 94, 1223–1228 (2009).
Kalsbeek, A., Buijs, R. M., van Heerikhuize, J. J., Arts, M. & van der Woude, T. P. Vasopressin-containing neurons of the suprachiasmatic nuclei inhibit corticosterone release. Brain Res. 580, 62–67 (1992).
van Wamelen, D. J. et al. Suprachiasmatic nucleus neuropeptide expression in patients with Huntington's Disease. Sleep 36, 117–125 (2013).
Kudo, T. et al. Circadian dysfunction in response to in vivo treatment with the mitochondrial toxin 3-nitropropionic acid. ASN Neuro 6, e00133 (2014).
Robinson, I. & Reddy, A. B. Molecular mechanisms of the circadian clockwork in mammals. FEBS Lett. 588, 2477–2483 (2014).
Acknowledgements
A.V. has received research grant support from the NIH (K23 NS072283). A.S.L. has received research grant support from the CHDI Foundation (RG50786). R.A.B. has received research grant support from the CHDI Foundation (RG50786), Evelyn Trust (RG66030), Butterfield Trust (RG68592), and the NIHR Biomedical Research Centre and Biomedical Research Unit Award to Addenbrooke's Hospital/University of Cambridge (RG68592). S.O. has received research grant support from the Netherlands Organization for Scientific Research (grant no. 016.116.371).
Author information
Authors and Affiliations
Contributions
A.V., A.S.L. and S.O. researched data for the article. All authors provided substantial contributions to discussion of the content, wrote the article and reviewed and/or edited the manuscript before and after submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
PowerPoint slides
Rights and permissions
About this article
Cite this article
Videnovic, A., Lazar, A., Barker, R. et al. 'The clocks that time us'—circadian rhythms in neurodegenerative disorders. Nat Rev Neurol 10, 683–693 (2014). https://doi.org/10.1038/nrneurol.2014.206
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrneurol.2014.206
This article is cited by
-
Regularity of bedtime, wake-up time, and time in bed in mid-life: associations with cardiometabolic health markers with adjustment for physical activity and sedentary time
Journal of Activity, Sedentary and Sleep Behaviors (2024)
-
Metabolic characteristics of transmembrane prolyl 4-hydroxylase (P4H-TM) deficient mice
Pflügers Archiv - European Journal of Physiology (2024)
-
Mitigation role of physical exercise participation in the relationship between blood cadmium and sleep disturbance: a cross-sectional study
BMC Public Health (2023)
-
Circadian disruption and sleep disorders in neurodegeneration
Translational Neurodegeneration (2023)
-
Proteostasis failure exacerbates neuronal circuit dysfunction and sleep impairments in Alzheimer’s disease
Molecular Neurodegeneration (2023)