Key Points
-
Noninvasive brain stimulation (NIBS) is a promising approach to enhance recovery after stroke, but its beneficial effect is limited and the technique is not yet ready for broad clinical use
-
We suggest that the disappointments in NIBS trials are related to over-reliance on the interhemispheric competition and vicariation models of recovery, which are oversimplified and do not apply to all patients with stroke
-
The concept of 'structural reserve' integrates the effects that interhemispheric inhibition and vicariation exert on the unlesioned residual network
-
We propose a unified 'bimodal balance–recovery model' that takes into account this individual residual network
-
The model could be used to tailor treatment for individual patients and increase the efficacy of NIBS in stroke rehabilitation
Abstract
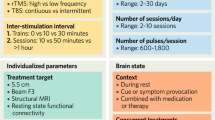
Noninvasive brain stimulation (NIBS) techniques can be used to monitor and modulate the excitability of intracortical neuronal circuits. Long periods of cortical stimulation can produce lasting effects on brain function, paving the way for therapeutic applications of NIBS in chronic neurological disease. The potential of NIBS in stroke rehabilitation has been of particular interest, because stroke is the main cause of permanent disability in industrial nations, and treatment outcomes often fail to meet the expectations of patients. Despite promising reports from many clinical trials on NIBS for stroke recovery, the number of studies reporting a null effect remains a concern. One possible explanation is that the interhemispheric competition model—which posits that suppressing the excitability of the hemisphere not affected by stroke will enhance recovery by reducing interhemispheric inhibition of the stroke hemisphere, and forms the rationale for many studies—is oversimplified or even incorrect. Here, we critically review the proposed mechanisms of synaptic and functional reorganization after stroke, and suggest a bimodal balance–recovery model that links interhemispheric balancing and functional recovery to the structural reserve spared by the lesion. The proposed model could enable NIBS to be tailored to the needs of individual patients.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Kolominsky-Rabas, P. L., Weber, M., Gefeller, O., Neundoerfer, B. & Heuschmann, P. U. Epidemiology of ischemic stroke subtypes according to TOAST criteria: incidence, recurrence, and long-term survival in ischemic stroke subtypes: a population-based study. Stroke 32, 2735–2740 (2001).
Roger, V. L. et al. Heart disease and stroke statistics. A report from the American Heart Association. Circulation 123, e18–e209 (2011).
Veerbeek, J. M., Kwakkel, G., van Wegen, E. E., Ket, J. C. & Heymans, M. W. Early prediction of outcome of activities of daily living after stroke: a systematic review. Stroke 42, 1482–1488 (2011).
Lansberg, M. G., Bluhmki, E. & Thijs, V. N. Efficacy and safety of tissue plasminogen activator 3 to 4.5 hours after acute ischemic stroke: a meta-analysis. Stroke 40, 2438–2441 (2009).
Maulden, S. A., Gassaway, J., Horn, S. D., Smout, R. J. & DeJong, G. Timing of initiation of rehabilitation after stroke. Arch. Phys. Med. Rehabil. 86, S34–S40 (2005).
Dobkin, B. H. Clinical practice. Rehabilitation after stroke. N. Engl. J. Med. 352, 1677–1684 (2005).
Wagner, T. H. et al. An economic analysis of robot-assisted therapy for long-term upper-limb impairment after stroke. Stroke 42, 2630–2632 (2011).
Lo, A. C. et al. Robot-assisted therapy for long-term upper-limb impairment after stroke. N. Engl. J. Med. 362, 1772–1783 (2010).
Klamroth-Marganska, V. et al. Three-dimensional, task-specific robot therapy of the arm after stroke: a multicentre, parallel-group randomised trial. Lancet Neurol. 13, 159–166 (2014).
Hao, Z., Wang, D., Zeng, Y. & Liu, M. Repetitive transcranial magnetic stimulation for improving function after stroke. Cochrane Database of Systematic Reviews, Issue 5. Art. No.: Cd008862. http://dx.doi.org/10.1002/14651858.CD008862.pub2.
Elsner, B., Kugler, J., Pohl, M. & Mehrholz, J. Transcranial direct current stimulation (tDCS) for improving function and activities of daily living in patients after stroke. Cochrane Database of Systematic Reviews, Issue 11. Art. No.: Cd009645. http://dx.doi.org/10.1002/14651858.CD009645.pub2.
Adeyemo, B. O., Simis, M., Macea, D. D. & Fregni, F. Systematic review of parameters of stimulation, clinical trial design characteristics, and motor outcomes in non-invasive brain stimulation in stroke. Front. Psychiatry 3, 88 (2012).
Hsu, W. Y., Cheng, C. H., Liao, K. K., Lee, I. H. & Lin, Y. Y. Effects of repetitive transcranial magnetic stimulation on motor functions in patients with stroke: a meta-analysis. Stroke 43, 1849–1857 (2012).
Di Lazzaro, V. et al. I-wave origin and modulation. Brain Stimul. 5, 512–525 (2012).
Attwell, D. & Laughlin, S. B. An energy budget for signaling in the grey matter of the brain. J. Cereb. Blood Flow Metab. 21, 1133–1145 (2001).
Astrup, J., Siesjo, B. K. & Symon, L. Thresholds in cerebral ischemia: the ischemic penumbra. Stroke 12, 723–725 (1981).
Symon, L. The relationship between CBF, evoked potentials and the clinical features in cerebral ischaemia. Acta Neurol. Scand. Suppl. 78, 175–190 (1980).
Krnjevic´, K. & Xu, Y. Mechanisms underlying anoxic hyperpolarization of hippocampal neurons. Can. J. Physiol. Pharmacol. 68, 1609–1613 (1990).
Somjen, G. G. (Ed.) Ions in the Brain: Normal Function, Seizures, and Stroke: Normal Function, Seizures, and Stroke (Oxford University Press, 2004).
Gerard, R. The response of nerve to oxygen lack. Am. J. Physiol. 92, 498–541 (1930).
Zhu, P. & Krnjevic´, K. Anoxia selectively depresses excitatory synaptic transmission in hippocampal slices. Neurosci. Lett. 166, 27–30 (1994).
Khazipov, R., Bregestovski, P. & Ben-Ari, Y. Hippocampal inhibitory interneurons are functionally disconnected from excitatory inputs by anoxia. J. Neurophysiol. 70, 2251–2259 (1993).
Murphy, T. H. & Corbett, D. Plasticity during stroke recovery: from synapse to behaviour. Nat. Rev. Neurosci. 10, 861–872 (2009).
Clarkson, A. N., Huang, B. S., MacIsaac, S. E., Mody, I. & Carmichael, S. T. Reducing excessive GABA-mediated tonic inhibition promotes functional recovery after stroke. Nature 468, 305–309 (2010).
Carmichael, S. T. Brain excitability in stroke: the yin and yang of stroke progression. Arch. Neurol. 69, 161 (2012).
Buchkremer-Ratzmann, I., August, M., Hagemann, G. & Witte, O. W. Electrophysiological transcortical diaschisis after cortical photothrombosis in rat brain. Stroke 27, 1105–1111 (1996).
Schiene, K. et al. Neuronal hyperexcitability and reduction of GABAA-receptor expression in the surround of cerebral photothrombosis. J. Cereb. Blood Flow Metab. 16, 906–914 (1996).
Bruehl, C., Neumann-Haefelin, T. & Witte, O. Enhancement of whole cell calcium currents following transient MCAO. Brain Res. 884, 129–138 (2000).
Bahn, M. M., Oser, A. B. & Cross, D. T. CT and MRI of stroke. J. Magn. Reson. Imaging 6, 833–845 (1996).
Cicinelli, P., Traversa, R. & Rossini, P. Post-stroke reorganization of brain motor output to the hand: a 2–4 month follow-up with focal magnetic transcranial stimulation. Electroencephalogr. Clin. Neurophysiol. 105, 438–450 (1997).
Traversa, R., Cicinelli, P., Bassi, A., Rossini, P. M. & Bernardi, G. Mapping of motor cortical reorganization after stroke. A brain stimulation study with focal magnetic pulses. Stroke 28, 110–117 (1997).
Traversa, R., Cicinelli, P., Pasqualetti, P., Filippi, M. & Rossini, P. M. Follow-up of interhemispheric differences of motor evoked potentials from the 'affected'and 'unaffected' hemispheres in human stroke. Brain Res. 803, 1–8 (1998).
Traversa, R. et al. Neurophysiological follow-up of motor cortical output in stroke patients. Clin. Neurophysiol. 111, 1695–1703 (2000).
Catano, A., Houa, M., Caroyer, J., Ducarne, H. & Noel, P. Magnetic transcranial stimulation in acute stroke: early excitation threshold and functional prognosis. Electroencephalogr. Clin. Neurophysiol. 101, 233–239 (1996).
Heald, A., Bates, D., Cartlidge, N., French, J. & Miller, S. Longitudinal study of central motor conduction time following stroke. 2. Central motor conduction measured within 72 h after stroke as a predictor of functional outcome at 12 months. Brain 116, 1371–1385 (1993).
Manganotti, P. et al. Motor disinhibition in affected and unaffected hemisphere in the early period of recovery after stroke. Clin. Neurophysiol. 113, 936–943 (2002).
Thickbroom, G. W., Byrnes, M. L., Archer, S. A. & Mastaglia, F. L. Motor outcome after subcortical stroke: MEPs correlate with hand strength but not dexterity. Clin. Neurophysiol. 113, 2025–2029 (2002).
Turton, A., Wroe, S., Trepte, N., Fraser, C. & Lemon, R. Contralateral and ipsilateral EMG responses to transcranial magnetic stimulation during recovery of arm and hand function after stroke. Electroencephalogr. Clin. Neurophysiol. 101, 316–328 (1996).
Catano, A., Houa, M. & Noël, P. Magnetic transcranial stimulation: dissociation of excitatory and inhibitory mechanisms in acute strokes. Electroencephalogr. Clin. Neurophysiol. 105, 29–36 (1997).
Palliyath, S. Role of central conduction time and motor evoked response amplitude in predicting stroke outcome. Electromyogr. Clin. Neurophys. 40, 315–320 (2000).
Rapisarda, G., Bastings, E., de Noordhout, A. M., Pennisi, G. & Delwaide, P. Can motor recovery in stroke patients be predicted by early transcranial magnetic stimulation? Stroke 27, 2191–2196 (1996).
Hendricks, H. T., Zwarts, M. J., Plat, E. F. & van Limbeek, J. Systematic review for the early prediction of motor and functional outcome after stroke by using motor-evoked potentials. Arch. Phys. Med. Rehabil. 83, 1303–1308 (2002).
Byrnes, M. L., Thickbroom, G. W., Phillips, B. A. & Mastaglia, F. L. Long-term changes in motor cortical organisation after recovery from subcortical stroke. Brain Res. 889, 278–287 (2001).
Pennisi, G. et al. Transcranial magnetic stimulation after pure motor stroke. Clin. Neurophysiol. 113, 1536–1543 (2002).
Pennisi, G. et al. Absence of response to early transcranial magnetic stimulation in ischemic stroke patients: prognostic value for hand motor recovery. Stroke 30, 2666–2670 (1999).
Delvaux, V. et al. Post-stroke reorganization of hand motor area: a 1-year prospective follow-up with focal transcranial magnetic stimulation. Clin. Neurophysiol. 114, 1217–1225 (2003).
Stinear, C. M., Barber, P. A., Petoe, M., Anwar, S. & Byblow, W. D. The PREP algorithm predicts potential for upper limb recovery after stroke. Brain 135, 2527–2535 (2012).
Liepert, J., Storch, P., Fritsch, A. & Weiller, C. Motor cortex disinhibition in acute stroke. Clin. Neurophysiol. 111, 671–676 (2000).
Cicinelli, P. et al. Interhemispheric asymmetries of motor cortex excitability in the postacute stroke stage: a paired-pulse transcranial magnetic stimulation study. Stroke 34, 2653–2658 (2003).
Swayne, O. B., Rothwell, J. C., Ward, N. S. & Greenwood, R. J. Stages of motor output reorganization after hemispheric stroke suggested by longitudinal studies of cortical physiology. Cereb. Cortex 18, 1909–1922 (2008).
Bütefisch, C. M., Netz, J., Wessling, M., Seitz, R. J. & Hömberg, V. Remote changes in cortical excitability after stroke. Brain 126, 470–481 (2003).
Di Lazzaro, V. et al. The level of cortical afferent inhibition in acute stroke correlates with long-term functional recovery in humans. Stroke 43, 250–252 (2012).
Foltys, H. et al. Motor representation in patients rapidly recovering after stroke: a functional magnetic resonance imaging and transcranial magnetic stimulation study. Clin. Neurophysiol. 114, 2404–2415 (2003).
Shimizu, T. et al. Motor cortical disinhibition in the unaffected hemisphere after unilateral cortical stroke. Brain 125, 1896–1907 (2002).
Fridman, E. A. et al. Reorganization of the human ipsilesional premotor cortex after stroke. Brain 127, 747–758 (2004).
Liepert, J., Hamzei, F. & Weiller, C. Motor cortex disinhibition of the unaffected hemisphere after acute stroke. Muscle Nerve 23, 1761–1763 (2000).
Murase, N., Duque, J., Mazzocchio, R. & Cohen, L. G. Influence of interhemispheric interactions on motor function in chronic stroke. Ann. Neurol. 55, 400–409 (2004).
Jacobs, K. M. & Donoghue, J. P. Reshaping the cortical motor map by unmasking latent intracortical connections. Science 251, 944–947 (1991).
Pineiro, R., Pendlebury, S., Johansen-Berg, H. & Matthews, P. Functional MRI detects posterior shifts in primary sensorimotor cortex activation after stroke: evidence of local adaptive reorganization? Stroke 32, 1134–1139 (2001).
Rossini, P. et al. Hand motor cortical area reorganization in stroke: a study with fMRI, MEG and TCS maps. Neuroreport 9, 2141–2146 (1998).
Calautti, C., Leroy, F., Guincestre, J.-Y. & Baron, J.-C. Displacement of primary sensorimotor cortex activation after subcortical stroke: a longitudinal PET study with clinical correlation. Neuroimage 19, 1650–1654 (2003).
Seitz, R. J. et al. Role of the premotor cortex in recovery from middle cerebral artery infarction. Arch. Neurol. 55, 1081–1088 (1998).
Chollet, F. et al. The functional anatomy of motor recovery after stroke in humans: a study with positron emission tomography. Ann. Neurol. 29, 63–71 (1991).
Weiller, C., Chollet, F., Friston, K. J., Wise, R. J. & Frackowiak, R. S. Functional reorganization of the brain in recovery from striatocapsular infarction in man. Ann. Neurol. 31, 463–472 (1992).
Weiller, C., Ramsay, S., Wise, R., Friston, K. & Frackowiak, R. Individual patterns of functional reorganization in the human cerebral cortex after capsular infarction. Ann. Neurol. 33, 181–189 (1993).
Johansen-Berg, H. et al. The role of ipsilateral premotor cortex in hand movement after stroke. Proc. Natl Acad. Sci. 99, 14518–14523 (2002).
Lotze, M. et al. The role of multiple contralesional motor areas for complex hand movements after internal capsular lesion. J. Neurosci. 26, 6096–6102 (2006).
Jang, S. H. A review of the ipsilateral motor pathway as a recovery mechanism in patients with stroke. NeuroRehabilitation 24, 315–320 (2009).
Ago, T. et al. Deterioration of pre-existing hemiparesis brought about by subsequent ipsilateral lacunar infarction. J. Neurol. Neurosurg. Psychiatry 74, 1152–1153 (2003).
Song, Y.-M., Lee, J.-Y., Park, J.-M., Yoon, B.-W. & Roh, J.-K. Ipsilateral hemiparesis caused by a corona radiata infarct after a previous stroke on the opposite side. Arch. Neurol. 62, 809–811 (2005).
Yamamoto, S., Takasawa, M., Kajiyama, K., Baron, J.-C. & Yamaguchi, T. Deterioration of hemiparesis after recurrent stroke in the unaffected hemisphere: three further cases with possible interpretation. Cerebrovasc. Dis. 23, 35–39 (2006).
Biernaskie, J., Szymanska, A., Windle, V. & Corbett, D. Bi-hemispheric contribution to functional motor recovery of the affected forelimb following focal ischemic brain injury in rats. Eur. J. Neurosci. 21, 989–999 (2005).
Lemon, R. N. Descending pathways in motor control. Annu. Rev. Neurosci. 31, 195–218 (2008).
Boudrias, M.-H., McPherson, R. L., Frost, S. B. & Cheney, P. D. Output properties and organization of the forelimb representation of motor areas on the lateral aspect of the hemisphere in rhesus macaques. Cerebr. Cortex 20, 169–186 (2010).
Baker, S. N. The primate reticulospinal tract, hand function and functional recovery. J. Physiol. 589, 5603–5612 (2011).
Eisner-Janowicz, I. et al. Early and late changes in the distal forelimb representation of the supplementary motor area after injury to frontal motor areas in the squirrel monkey. J. Neurophysiol. 100, 1498–1512 (2008).
Brus-Ramer, M., Carmel, J. B., Chakrabarty, S. & Martin, J. H. Electrical stimulation of spared corticospinal axons augments connections with ipsilateral spinal motor circuits after injury. J. Neurosci. 27, 13793–13801 (2007).
Verstynen, T., Diedrichsen, J., Albert, N., Aparicio, P. & Ivry, R. B. Ipsilateral motor cortex activity during unimanual hand movements relates to task complexity. J. Neurophysiol. 93, 1209–1222 (2005).
Hummel, F., Kirsammer, R. & Gerloff, C. Ipsilateral cortical activation during finger sequences of increasing complexity: representation of movement difficulty or memory load? Clin. Neurophys. 114, 605–613 (2003).
Rose, D. & Winstein, C. The co-ordination of bimanual rapid aiming movements following stroke. Clin. Rehabil. 19, 452–462 (2005).
Lewis, G. N. & Byblow, W. D. Bimanual coordination dynamics in poststroke hemiparetics. J. Mot. Behav. 36, 174–188 (2004).
Grefkes, C. et al. Cortical connectivity after subcortical stroke assessed with functional magnetic resonance imaging. Ann. Neurol. 63, 236–246 (2008).
Finger, S. In Handbook of Clinical Neurology Vol. 95 Ch. 51 (eds Aminoff, J. A. et al.) 833–841 (Elsevier, 2009).
Duque, J. et al. Transcallosal inhibition in chronic subcortical stroke. Neuroimage 28, 940–946 (2005).
Werhahn, K. J., Mortensen, J., Van Boven, R. W., Zeuner, K. E. & Cohen, L. G. Enhanced tactile spatial acuity and cortical processing during acute hand deafferentation. Nat. Neurosci. 5, 936–938 (2002).
Floel, A. et al. Influence of somatosensory input on motor function in patients with chronic stroke. Ann. Neurol. 56, 206–212 (2004).
Ward, N. S. & Cohen, L. G. Mechanisms underlying recovery of motor function after stroke. Arch. Neurol. 61, 1844–1848 (2004).
Stinear, C. M. et al. Functional potential in chronic stroke patients depends on corticospinal tract integrity. Brain 130, 170–180 (2007).
Lindenberg, R. et al. Structural integrity of corticospinal motor fibers predicts motor impairment in chronic stroke. Neurology 74, 280–287 (2010).
Bradnam, L. V., Stinear, C. M., Barber, P. A. & Byblow, W. D. Contralesional hemisphere control of the proximal paretic upper limb following stroke. Cerebr. Cortex 22, 2662–2671 (2012).
Werhahn, K. J., Conforto, A. B., Kadom, N., Hallett, M. & Cohen, L. G. Contribution of the ipsilateral motor cortex to recovery after chronic stroke. Ann. Neurol. 54, 464–472 (2003).
Seghier, M. L. Laterality index in functional MRI: methodological issues. Magn. Res. Imaging 26, 594–601 (2008).
Johansen-Berg, H. Functional imaging of stroke recovery: what have we learnt and where do we go from here? Int. J. Stroke 2, 7–16 (2007).
Johansen-Berg, H., Scholz, J. & Stagg, C. J. Relevance of structural brain connectivity to learning and recovery from stroke. Front. Syst. Neurosci. 4, 146 (2010).
Bestmann, S. et al. The role of contralesional dorsal premotor cortex after stroke as studied with concurrent TMS–fMRI. J. Neurosci. 30, 11926–11937 (2010).
Mochizuki, H., Huang, Y.-Z. & Rothwell, J. C. Interhemispheric interaction between human dorsal premotor and contralateral primary motor cortex. J. Physiol. 561, 331–338 (2004).
Koch, G. et al. Time course of functional connectivity between dorsal premotor and contralateral motor cortex during movement selection. J. Neurosci. 26, 7452–7459 (2006).
Lomarev, M., Kim, D., Richardson, S. P., Voller, B. & Hallett, M. Safety study of high-frequency transcranial magnetic stimulation in patients with chronic stroke. Clin. Neurophysiol. 118, 2072–2075 (2007).
Magee, J. C. & Johnston, D. A synaptically controlled, associative signal for Hebbian plasticity in hippocampal neurons. Science 275, 209–213 (1997).
Bi, G. & Poo, M. Synaptic modification by correlated activity: Hebb's postulate revisited. Annu. Rev. Neurosci. 24, 139–166 (2001).
Bi, G. Q. & Poo, M. M. Synaptic modifications in cultured hippocampal neurons: dependence on spike timing, synaptic strength, and postsynaptic cell type. J. Neurosci. 18, 10464–10472 (1998).
Hoogendam, J. M., Ramakers, G. M. & Di Lazzaro, V. Physiology of repetitive transcranial magnetic stimulation of the human brain. Brain Stimul. 3, 95–118 (2010).
Huang, Y.-Z., Rothwell, J. C., Chen, R.-S., Lu, C.-S. & Chuang, W.-L. The theoretical model of theta burst form of repetitive transcranial magnetic stimulation. Clin. Neurophysiol. 122, 1011–1018 (2011).
Wolters, A. et al. A temporally asymmetric Hebbian rule governing plasticity in the human motor cortex. J. Neurophysiol. 89, 2339–2345 (2003).
Stefan, K., Kunesch, E., Cohen, L. G., Benecke, R. & Classen, J. Induction of plasticity in the human motor cortex by paired associative stimulation. Brain 123, 572–584 (2000).
Conforto, A. B. et al. Transcranial magnetic stimulation in mild to severe hemiparesis early after stroke: a proof of principle and novel approach to improve motor function. J. Neurol. 259, 1399–1405 (2012).
Talelli, P. et al. Theta burst stimulation in the rehabilitation of the upper limb: a semirandomized, placebo-controlled trial in chronic stroke patients. Neurorehabil. Neural Repair 26, 976–987 (2012).
Lindenberg, R., Zhu, L. L. & Schlaug, G. Combined central and peripheral stimulation to facilitate motor recovery after stroke: the effect of number of sessions on outcome. Neurorehabil. Neural Repair 26, 479–483 (2012).
Avenanti, A., Coccia, M., Ladavas, E., Provinciali, L. & Ceravolo, M. Low-frequency rTMS promotes use-dependent motor plasticity in chronic stroke: a randomized trial. Neurology 78, 256–264 (2012).
Kakuda, W. et al. A multi-center study on low-frequency rTMS combined with intensive occupational therapy for upper limb hemiparesis in post-stroke patients. J. Neuroeng. Rehabil. 9, 4 (2012).
Chang, W. H. et al. rTMS with motor training modulates cortico-basal ganglia-thalamocortical circuits in stroke patients. Restor. Neurol. Neurosci. 30, 179–189 (2012).
Seniów, J. et al. Transcranial magnetic stimulation combined with physiotherapy in rehabilitation of poststroke hemiparesis: a randomized, double-blind, placebo-controlled study. Neurorehabil. Neural Repair 26, 1072–1079 (2012).
Cazzoli, D. et al. Theta burst stimulation reduces disability during the activities of daily living in spatial neglect. Brain 135, 3426–3439 (2012).
Ameli, M. et al. Differential effects of high-frequency repetitive transcranial magnetic stimulation over ipsilesional primary motor cortex in cortical and subcortical middle cerebral artery stroke. Ann. Neurol. 66, 298–309 (2009).
Boggio, P. S. et al. Hand function improvement with low-frequency repetitive transcranial magnetic stimulation of the unaffected hemisphere in a severe case of stroke. Am. J. Phys. Med. Rehabil. 85, 927–930 (2006).
Koch, G. et al. Theta-burst stimulation of the left hemisphere accelerates recovery of hemispatial neglect. Neurology 78, 24–30 (2012).
Di Lazzaro, V. et al. Modulation of motor cortex neuronal networks by rTMS: comparison of local and remote effects of six different protocols of stimulation. J. Neurophysiol. 105, 2150–2156 (2011).
Datta, A., Truong, D., Minhas, P., Parra, L. C. & Bikson, M. Inter-individual variation during transcranial direct current stimulation and normalization of dose using MRI-derived computational models. Front. Psychiatry 3, 91 (2012).
Maeda, F., Keenan, J. P., Tormos, J. M., Topka, H. & Pascual-Leone, A. Interindividual variability of the modulatory effects of repetitive transcranial magnetic stimulation on cortical excitability. Exp. Brain Res. 133, 425–430 (2000).
Hamada, M., Murase, N., Hasan, A., Balaratnam, M. & Rothwell, J. C. The role of interneuron networks in driving human motor cortical plasticity. Cereb. Cortex 23, 1593–1605 (2013).
Cheeran, B. et al. A common polymorphism in the brain-derived neurotrophic factor gene (BDNF) modulates human cortical plasticity and the response to rTMS. J. Physiol. 586, 5717–5725 (2008).
Nitsche, M. A., Müller-Dahlhaus, F., Paulus, W. & Ziemann, U. The pharmacology of neuroplasticity induced by non-invasive brain stimulation: building models for the clinical use of CNS active drugs. J. Physiol. 590, 4641–4662 (2012).
Wu, D. et al. Effects on decreasing upper-limb poststroke muscle tone using transcranial direct current stimulation: a randomized sham-controlled study. Arch. Phys. Med. Rehabil. 94, 1–8 (2013).
Khedr, E. M., Etraby, A. E., Hemeda, M., Nasef, A. M. & Razek, A. A. Long-term effect of repetitive transcranial magnetic stimulation on motor function recovery after acute ischemic stroke. Acta Neurol. Scand. 121, 30–37 (2010).
Hesse, S. et al. Combined transcranial direct current stimulation and robot-assisted arm training in subacute stroke patients: an exploratory, randomized multicenter trial. Neurorehabil. Neural Repair 25, 838–846 (2011).
Bolognini, N. et al. Neurophysiological and behavioral effects of tDCS combined with constraint-induced movement therapy in poststroke patients. Neurorehabil. Neural Repair 25, 819–829 (2011).
Hummel, F. C. & Cohen, L. G. Non-invasive brain stimulation: a new strategy to improve neurorehabilitation after stroke? Lancet Neurol. 5, 708–712 (2006).
Khedr, E. M., Ahmed, M. A., Fathy, N. & Rothwell, J. C. Therapeutic trial of repetitive transcranial magnetic stimulation after acute ischemic stroke. Neurology 65, 466–468 (2005).
Nair, D. G., Renga, V., Lindenberg, R., Zhu, L. & Schlaug, G. Optimizing recovery potential through simultaneous occupational therapy and non-invasive brain-stimulation using tDCS. Restor. Neurol. Neurosci. 29, 411–420 (2011).
Grefkes, C. & Fink, G. R. Disruption of motor network connectivity post-stroke and its noninvasive neuromodulation. Curr. Opin. Neurol. 25, 670–675 (2012).
Schlaug, G., Renga, V. & Nair, D. Transcranial direct current stimulation in stroke recovery. Arch. Neurol. 65, 1571–1576 (2008).
Hummel, F. C. et al. Controversy: noninvasive and invasive cortical stimulation show efficacy in treating stroke patients. Brain Stim. 1, 370–382 (2008).
Plow, E. B., Carey, J. R., Nudo, R. J., & Pascual-Leone, A. Invasive cortical stimulation to promote recovery of function after stroke: a critical appraisal. Stroke 40, 1926–1931 (2009).
Assenza, G. et al. Neuronal functionality assessed by magnetoencephalography is related to oxidative stress system in acute ischemic stroke. Neuroimage 44, 1267–1273 (2009).
Sheorajpanday, R. V., Nagels, G., Weeren, A. J., van Putten, M. J. & De Deyn, P. P. Quantitative EEG in ischemic stroke: correlation with functional status after 6 months. Clin. Neurophysiol. 122, 874–883 (2011).
Finnigan, S., Rose, S. E. & Chalk, J. B. Contralateral hemisphere delta EEG in acute stroke precedes worsening of symptoms and death. Clin. Neurophysiol. 119, 1690–1694 (2008).
Tecchio, F. et al. Outcome prediction in acute monohemispheric stroke via magnetoencephalography. J. Neurol. 254, 296–305 (2007).
Assenza, G., Zappasodi, F., Pasqualetti, P., Vernieri, F. & Tecchio, F. A contralesional EEG power increase mediated by interhemispheric disconnection provides negative prognosis in acute stroke. Restor. Neurol. Neurosci. 31, 177–188 (2013).
Finnigan, S. & van Putten, M. J. EEG in ischaemic stroke: quantitative EEG can uniquely inform (sub-) acute prognoses and clinical management. Clin. Neurophysiol. 124, 10–9 (2012).
Graziadio, S., Tomasevic, L., Assenza, G., Tecchio, F. & Eyre, J. The myth of the 'unaffected' side after unilateral stroke: is reorganisation of the non-infarcted corticospinal system to re-establish balance the price for recovery? Exp. Neurol. 238, 168–175 (2012).
Ilmoniemi, R. J. et al. Neuronal responses to magnetic stimulation reveal cortical reactivity and connectivity. Neuroreport 8, 3537–3540 (1997).
Ferreri, F. et al. Does an intraneural interface short-term implant for robotic hand control modulate sensorimotor cortical integration? An EEG–TMS co-registration study on a human amputee. Restor. Neurol. Neurosci. 32, 281–292 (2014).
Premoli, I. et al. TMS-EEG signatures of GABAergic neurotransmission in the human cortex. J. Neurosci. 34, 5603–5612 (2014).
Nikulin, V. V., Kicic, D., Kahkonen, S. & Ilmoniemi, R. J. Modulation of electroencephalographic responses to transcranial magnetic stimulation: evidence for changes in cortical excitability related to movement. Eur. J. Neurosci. 18, 1206–1212 (2003).
Ferreri, F. et al. Human brain connectivity during single and paired pulse transcranial magnetic stimulation. Neuroimage 54, 90–102 (2011).
Kujirai, T. et al. Corticocortical inhibition in human motor cortex. J. Physiol. 471, 501–519 (1993).
Di Lazzaro, V. et al. Direct demonstration of the effect of lorazepam on the excitability of the human motor cortex. Clin. Neurophys. 111, 794–799 (2000).
Calancie, B., Nordin, M., Wallin, U. & Hagbarth, K.-E. Motor-unit responses in human wrist flexor and extensor muscles to transcranial cortical stimuli. J. Neurophysiol. 58, 1168–1185 (1987).
Valls-Solé, J., Pascual-Leone, A., Wassermann, E. M. & Hallett, M. Human motor evoked responses to paired transcranial magnetic stimuli. Electroencephalogr. Clin. Neurophysiol. 85, 355–364 (1992).
Di Lazzaro, V. et al. Segregating two inhibitory circuits in human motor cortex at the level of GABAA receptor subtypes: a TMS study. Clin. Neurophys. 118, 2207–2214 (2007).
Ziemann, U., Rothwell, J. C. & Ridding, M. C. Interaction between intracortical inhibition and facilitation in human motor cortex. J. Physiol. 496, 873–881 (1996).
Di Lazzaro, V. et al. Origin of facilitation of motor-evoked potentials after paired magnetic stimulation: direct recording of epidural activity in conscious humans. J. Neurophysiol. 96, 1765–1771 (2006).
Siebner, H. R. et al. Consensus paper: combining transcranial stimulation with neuroimaging. Brain Stimul. 2, 58–80 (2009).
Ziemann, U. Transcranial magnetic stimulation at the interface with other techniques: a powerful tool for studying the human cortex. Neuroscientist 17, 368–381 (2011).
Ilmoniemi, R. J. & Kicic, D. Methodology for combined TMS and EEG. Brain Topogr. 22, 233–248 (2010).
Ziemann, U. et al. Consensus: motor cortex plasticity protocols. Brain Stim. 1, 164–182 (2008).
Thickbroom, G. W. Transcranial magnetic stimulation and synaptic plasticity: experimental framework and human models. Exp. Brain Res. 180, 583–593 (2007).
Huang, Y. Z., Edwards, M. J., Rounis, E., Bhatia, K. P. & Rothwell, J. C. Theta burst stimulation of the human motor cortex. Neuron 45, 201–206 (2005).
Di Lazzaro, V. et al. Motor cortex plasticity predicts recovery in acute stroke. Cereb. Cortex 20, 1523–1528 (2010).
Müller-Dahlhaus, F., Ziemann, U. & Classen, J. Plasticity resembling spike-timing dependent synaptic plasticity: the evidence in human cortex. Front. Synaptic Neurosci. 2, 34 (2010).
Stefan, K., Kunesch, E., Benecke, R., Cohen, L. G. & Classen, J. Mechanisms of enhancement of human motor cortex excitability induced by interventional paired associative stimulation. J. Physiol. 543, 699–708 (2002).
Wolters, A. et al. Timing-dependent plasticity in human primary somatosensory cortex. J. Physiol. 565, 1039–1052 (2005).
Heidegger, T., Krakow, K. & Ziemann, U. Effects of antiepileptic drugs on associative LTP-like plasticity in human motor cortex. Eur. J. Neurosci. 32, 1215–1222 (2010).
Korchounov, A. & Ziemann, U. Neuromodulatory neurotransmitters influence LTP-like plasticity in human cortex: a pharmaco-TMS study. Neuropsychopharmacology 36, 1894–1902 (2011).
Nitsche, M. A. et al. Transcranial direct current stimulation: state of the art 2008. Brain Stim. 1, 206–223 (2008).
Ranieri, F. et al. Modulation of LTP at rat hippocampal CA3-CA1 synapses by direct current stimulation. J. Neurophysiol. 107, 1868–1880 (2012).
Medeiros, L. F. et al. Neurobiological effects of transcranial direct current stimulation: a review. Front. Psychiatry 3, 110 (2012).
Nitsche, M. A. et al. Modulating parameters of excitability during and after transcranial direct current stimulation of the human motor cortex. J. Physiol. 568, 291–303 (2005).
Liebetanz, D., Nitsche, M. A., Tergau, F. & Paulus, W. Pharmacological approach to the mechanisms of transcranial DC-stimulation-induced after-effects of human motor cortex excitability. Brain 125, 2238–2247 (2002).
Powell, J., Pandyan, A. D., Granat, M., Cameron, M. & Stott, D. J. Electrical stimulation of wrist extensors in poststroke hemiplegia. Stroke 30, 1384–1389 (1999).
Conforto, A. B., Kaelin-Lang, A. & Cohen, L. G. Increase in hand muscle strength of stroke patients after somatosensory stimulation. Ann. Neurol. 51, 122–125 (2002).
Ng, S. S. & Hui-Chan, C. W. Transcutaneous electrical nerve stimulation combined with task-related training improves lower limb functions in subjects with chronic stroke. Stroke 38, 2953–2959 (2007).
Ada, L. & Foongchomcheay, A. Efficacy of electrical stimulation in preventing or reducing subluxation of the shoulder after stroke: a meta-analysis. Aust. J. Physiother. 48, 257–267 (2002).
Price, C. I. & Pandyan, A. Electrical stimulation for preventing and treating post-stroke shoulder pain: a systematic Cochrane review. Clin. Rehabil. 15, 5–19 (2001).
Sujith, O. Functional electrical stimulation in neurological disorders. Eur. J. Neurol. 15, 437–444 (2008).
Wu, C. W.-H. & Kaas, J. H. The effects of long-standing limb loss on anatomical reorganization of the somatosensory afferents in the brainstem and spinal cord. Somatosens. Mot. Res. 19, 153–163 (2002).
Author information
Authors and Affiliations
Contributions
G.D.P. developed the model and wrote the article. G.P. searched the literature and wrote the section on the stimulation protocols and clinical trials. G.A. searched the literature and wrote the section on EEG and repetitive electric stimulation. F.C. searched the literature and participated in writing the section on predictors of recovery. F.F. searched the literature and wrote the section on TMS–EEG in the multimodal diagnostic approach. D.F. was involved in the computational development of the model. M.T. searched the literature and wrote the section on synaptic dysfunctions following stroke. F.R. provided substantial contributions to discussion of the content. U.Z., J.C.R. and V.D.L. guided the manuscript development and revised the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Di Pino, G., Pellegrino, G., Assenza, G. et al. Modulation of brain plasticity in stroke: a novel model for neurorehabilitation. Nat Rev Neurol 10, 597–608 (2014). https://doi.org/10.1038/nrneurol.2014.162
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrneurol.2014.162
This article is cited by
-
Determining the effects of targeted high-definition transcranial direct current stimulation on reducing post-stroke upper limb motor impairments—a randomized cross-over study
Trials (2024)
-
Electric field simulation and appropriate electrode positioning for optimized transcranial direct current stimulation of stroke patients: an in Silico model
Scientific Reports (2024)
-
Non-Invasive Brain Stimulation Combined with Neuromuscular Electrical Stimulation for Upper Limb Rehabilitation in Stroke Survivors: A Systematic Review
Current Physical Medicine and Rehabilitation Reports (2024)
-
The effects of rTMS on motor recovery after stroke: a systematic review of fMRI studies
Neurological Sciences (2024)
-
Protocol for a single-blind randomized clinical trial to test the efficacy of bilateral transcranial magnetic stimulation on upper extremity motor function in patients recovering from stroke
Trials (2023)