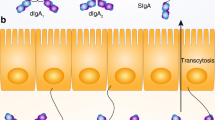
Key Points
-
Intestinal immunoglobulin A (IgA)+ plasma cells are derived from conventional B2 cells located in the Peyer's patches or isolated lymphoid follicles of the gut, and from B1 cells, which reside in the body cavities, in particular the peritoneal cavity.
-
CCL25 is produced by the small intestine epithelium and specifically attracts IgA-committed B cells derived from conventional B2 cells.
-
The presence of IgM+ B cells and IgA+ plasma cells in the gut depends on interactions between lymphotoxin (LT) and LTβ receptor, which are present on lymphocytes and lamina-propria stromal cells, respectively.
-
IgM+ B cells switch to IgA+ B cells and differentiate to IgA+ plasma cells in situ in the lamina propria, with the help of local dendritic cells and stromal cells.
-
The presence of somatically mutated IgAs is essential for the maintenance of gut homeostasis. AID-deficient mice develop hyperplasia of isolated lymphoid follicles, which is triggered by a large increase in the number of anaerobic bacteria in the small intestine.
-
Dysregulation of the gut flora causes activation of all the immune systems of the body.
Abstract
Immunoglobulin A is the most abundant immunoglobulin isotype in mucosal secretions. In this review, we summarize recent advances in our understanding of the sites, mechanisms and functions of intestinal IgA synthesis in mice. On the basis of these recent findings, we propose an updated model for the induction and regulation of IgA responses in the gut. In addition, we discuss new insights into the role of IgA in the maintenance of gut homeostasis and into the reciprocal interactions between gut B cells and the bacterial flora.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Mestecky, J., Moro, I. & Underdown, B. J. in Mucosal Immunology (eds Ogra, P. et al.) 133–152 (Academic Press, San Diego, 1999).
Matsunaga, T. & Rahman, A. What brought the adaptive immune system to vertebrates? The jaw hypothesis and the seahorse. Immunol. Rev. 166, 177–186 (1998).
Brandtzaeg, P. et al. Regional specialization in the mucosal immune system: what happens in the microcompartments? Immunol. Today 20, 141–151 (1999).
van Egmond, M. et al. IgA and the IgA Fc receptor. Trends Immunol. 22, 205–211 (2001).
Mostov, K. E. Transepithelial transport of immunoglobulins. Annu. Rev. Immunol. 12, 63–84 (1994).
Hanson, L. A. Comparative immunological studies of the immunoglobulins of human milk and of blood serum. Int. Arch. Allergy Appl. Immunol. 18, 241–253 (1961).
Tomasi, T. B., Tan, E. M., Solomon, A. & Predergast, R. A. Characteristics of an immune system common to certain external secretions. J. Exp. Med. 121, 101–142 (1965).
Cebra, J. J. & Shroff, K. E. in Handbook of Mucosal Immunology (eds Ogra, P. et al.) 151–157 (Academic Press, San Diego, 1994).
Kelsall, B. & Strober, W. in Mucosal Immunology (eds Ogra, P. et al.) 293–317 (Academic Press, San Diego, 1999).
Moghaddami, M., Cummins, A. & Mayrhofer, G. Lymphocyte-filled villi: comparison with other lymphoid aggregations in the mucosa of the human small intestine. Gastroenterology 115, 1414–1425 (1998).
Hamada, H. et al. Identification of multiple isolated lymphoid follicles on the antimesenteric wall of the mouse small intestine. J. Immunol. 168, 57–64 (2002). References 10 and 11 give detailed morphological descriptions of isolated lymphoid follicles in human and mouse small intestine, respectively.
Fagarasan, S. et al. Critical roles of activation-induced cytidine deaminase (AID) in the homeostasis of gut flora. Science 298, 1424–1427 (2002). This paper implicates class-switch recombination and somatic hypermutation of gut B cells as being essential for the maintenance of bacterial homeostasis in the gut.
McIntyre, T. & Strobel, W. in Mucosal Immunology (eds Ogra, P. et al.) 319–356 (Academic Press, San Diego, 1999).
Craig, S. W. & Cebra, J. J. Peyer's patches: an enriched source of precursors for IgA-producing immunocytes in the rabbit. J. Exp. Med. 134, 188–200 (1971).
Craig, S. W. & Cebra, J. J. Rabbit Peyer's patches, appendix, and popliteal lymph node B lymphocytes: a comparative analysis of their membrane immunoglobulin components and plasma-cell precursor potential. J. Immunol. 114, 492–502 (1975).
Tseng, J. Transfer of lymphocytes of Peyer's patches between immunoglobulin allotype congenic mice: repopulation of the IgA plasma cells in the gut lamina propria. J. Immunol. 127, 2039–2043 (1981).
Tseng, J. A population of resting IgM–IgD double-bearing lymphocytes in Peyer's patches: the major precursor cells for IgA plasma cells in the gut lamina propria. J. Immunol. 132, 2730–2735 (1984).
Butcher, E. C. et al. Surface phenotype of Peyer's patch germinal center cells: implications for the role of germinal centers in B-cell differentiation. J. Immunol. 129, 2698–2707 (1982).
Weinstein, P. D. & Cebra, J. J. The preference for switching to IgA expression by Peyer's patch germinal-center B cells is likely due to the intrinsic influence of their microenvironment. J. Immunol. 147, 4126–4135 (1991).
Cebra, J. J. Influences of microbiota on intestinal immune system development. Am. J. Clin. Nutr. 69, 1046S–1051S (1999).
Lebman, D. A., Griffin, P. M. & Cebra, J. J. Relationship between expression of IgA by Peyer's patch cells and functional IgA memory cells. J. Exp. Med. 166, 1405–1418 (1987).
Lycke, N. in Mucosal T Cells (ed. MacDonald, T.) 209–234 (Karger, Basel, 1998).
Crabbe, P. A., Nash, D. R., Bazin, H., Eyssen, H. & Heremans, J. F. Immunohistochemical observations on lymphoid tissues from conventional and germ-free mice. Lab. Invest. 22, 448–457 (1970).
Boursier, L., Farstad, I. N., Mellembakken, J. R., Brandtzaeg, P. & Spencer, J. IgVH gene analysis suggests that peritoneal B cells do not contribute to the gut immune system in man. Eur. J. Immunol. 32, 2427–2436 (2002).
Kroese, F. G. et al. Many of the IgA-producing plasma cells in murine gut are derived from self-replenishing precursors in the peritoneal cavity. Int. Immunol. 1, 75–84 (1989). This study provides the first evidence that many IgA-producing plasma cells in the lamina propria of the mouse small intestine are derived from peritoneal B1 cells.
Macpherson, A. J. et al. A primitive T-cell-independent mechanism of intestinal mucosal IgA responses to commensal bacteria. Science 288, 2222–2226 (2000). The first demonstration that anti-commensal IgA antibodies derived from B1 cells are not natural antibodies, but are induced specifically in response to antigenic stimulation; this study also indicates that this pathway is independent of T cells or follicular lymphoid-tissue organization.
Bos, N. A. et al. Monoclonal immunoglobulin A derived from peritoneal B cells is encoded by both germ line and somatically mutated VH genes and is reactive with commensal bacteria. Infect. Immun. 64, 616–623 (1996).
Guy-Grand, D., Griscelli, C. & Vassalli, P. The gut-associated lymphoid system: nature and properties of the large dividing cells. Eur. J. Immunol. 4, 435–443 (1974).
McWilliams, M., Phillips-Quagliata, J. M. & Lamm, M. E. Mesenteric lymph node B lymphoblasts which home to the small intestine are precommitted to IgA synthesis. J. Exp. Med. 145, 866–875 (1977).
Springer, T. A. Traffic signals for lymphocyte recirculation and leukocyte emigration: the multistep paradigm. Cell 76, 301–314 (1994).
Holzmann, B., McIntyre, B. W. & Weissman, I. L. Identification of a murine Peyer's patch-specific lymphocyte homing receptor as an integrin molecule with an α-chain homologous to human VLA-4. Cell 56, 37–46 (1989).
Briskin, M. J., McEvoy, L. M. & Butcher, E. C. MAdCAM-1 has homology to immunoglobulin and mucin-like adhesion receptors and to IgA1. Nature 363, 461–464 (1993).
Berlin, C. et al. α4β7 integrin mediates lymphocyte binding to the mucosal vascular addressin MAdCAM-1. Cell 74, 185 (1993).
Wagner, N. et al. Critical role for β7 integrins in formation of the gut-associated lymphoid tissue. Nature 382, 366–370 (1996).
Cyster, J. G. Chemokines and cell migration in secondary lymphoid organs. Science 286, 2098–2102 (1999).
Okada, T. et al. Chemokine requirements for B-cell entry to lymph nodes and Peyer's patches. J. Exp. Med. 196, 65–75 (2002).
Bowman, E. P. et al. The intestinal chemokine thymus-expressed chemokine (CCL25) attracts IgA antibody-secreting cells. J. Exp. Med. 195, 269–275 (2002). Butcher and colleagues provide evidence that TECK (CCL25) is an IgA+ B-cell chemotactic factor, which attracts IgA-committed B cells from the spleen, Peyer's patches and mesenteric lymph nodes to the lamina propria.
Kunkel, E. J. et al. Lymphocyte CC-chemokine receptor 9 and epithelial thymus-expressed chemokine (TECK) expression distinguish the small intestinal immune compartment: epithelial expression of tissue-specific chemokines as an organizing principle in regional immunity. J. Exp. Med. 192, 761–768 (2000).
Papadakis, K. A. et al. The role of thymus-expressed chemokine and its receptor CCR9 on lymphocytes in the regional specialization of the mucosal immune system. J. Immunol. 165, 5069–5076 (2000).
Kang, H. S. et al. Signaling via LTβR on the lamina propria stromal cells of the gut is required for IgA production. Nature Immunol. 3, 576–582 (2002).
Newberry, R. D., McDonough, J. S., McDonald, K. G. & Lorenz, R. G. Postgestational lymphotoxin/lymphotoxin-β receptor interactions are essential for the presence of intestinal B lymphocytes. J. Immunol. 168, 4988–4997 (2002). References 40 and 41 were the first to show that lymphotoxin-β receptor signalling on lamina-propria stromal cells is required for the presence of B cells in the lamina propria and for IgA production; these studies emphasize the sufficiency of the lamina-propria environment for the generation of IgA+ plasma cells.
Chaplin, D. D. & Fu, Y. Cytokine regulation of secondary lymphoid organ development. Curr. Opin. Immunol. 10, 289–297 (1998).
Fagarasan, S. et al. Alymphoplasia (aly)-type nuclear factor-κB-inducing kinase (NIK) causes defects in secondary lymphoid tissue chemokine receptor signaling and homing of peritoneal cells to the gut-associated lymphatic tissue system. J. Exp. Med. 191, 1477–1486 (2000).
Miyawaki, S. et al. A new mutation, aly, that induces a generalized lack of lymph nodes accompanied by immunodeficiency in mice. Eur. J. Immunol. 24, 429–434 (1994).
Shinkura, R. et al. Alymphoplasia is caused by a point mutation in the mouse gene encoding NF-κB-inducing kinase. Nature Genet. 22, 74–77 (1999).
Koike, R. et al. Analysis of expression of lymphocyte homing-related adhesion molecules in ALY mice deficient in lymph nodes and Peyer's patches. Cell. Immunol. 180, 62–69 (1997).
Ansel, K. M., Harris, R. B. & Cyster, J. G. CXCL13 is required for B1-cell homing, natural antibody production and body-cavity immunity. Immunity 16, 67–76 (2002).
Neumann, B., Luz, A., Pfeffer, K. & Holzmann, B. Defective Peyer's patch organogenesis in mice lacking the 55-kD receptor for tumor-necrosis factor. J. Exp. Med. 184, 259–264 (1996).
Vajdy, M., Kosco-Vilbois, M. H., Kopf, M., Kohler, G. & Lycke, N. Impaired mucosal immune responses in interleukin-4-targeted mice. J. Exp. Med. 181, 41–53 (1995).
Rennert, P. D., James, D., Mackay, F., Browning, J. L. & Hochman, P. S. Lymph node genesis is induced by signaling through the lymphotoxin-β receptor. Immunity 9, 71–79 (1998).
Yamamoto, M. et al. Alternate mucosal immune system: organized Peyer's patches are not required for IgA responses in the gastrointestinal tract. J. Immunol. 164, 5184–5191 (2000).
Muramatsu, M. et al. Class-switch recombination and hypermutation require activation-induced cytidine deaminase (AID), a potential RNA-editing enzyme. Cell 102, 553–563 (2000). Together with reference 54, this paper shows that class-switch recombination and somatic hypermutation depend on AID, a putative RNA-editing enzyme, which is expressed specifically by activated B cells.
Muramatsu, M. et al. Specific expression of activation-induced cytidine deaminase (AID), a novel member of the RNA-editing deaminase family in germinal-center B cells. J. Biol. Chem. 274, 18470–18476 (1999).
Revy, P. et al. Activation-induced cytidine deaminase (AID) deficiency causes the autosomal recessive form of the hyper-IgM syndrome (HIGM2). Cell 102, 565–575 (2000).
Fagarasan, S., Kinoshita, K., Muramatsu, M., Ikuta, K. & Honjo, T. In situ class switching and differentiation to IgA-producing cells in the gut lamina propria. Nature 413, 639–643 (2001). The first demonstration at the molecular level (methodology described in reference 57) that class-switching to IgA takes place in situ in the lamina propria; the dominance of IgA in the gut is explained by the capacity of lamina-propria stromal cells to enhance preferential class-switching and differentiation to IgA-producing plasma cells.
Kamata, T. et al. Increased frequency of surface IgA-positive plasma cells in the intestinal lamina propria and decreased IgA excretion in hyper IgA (HIGA) mice, a murine model of IgA nephropathy with hyperserum IgA. J. Immunol. 165, 1387–1394 (2000).
Kinoshita, K., Harigai, M., Fagarasan, S., Muramatsu, M. & Honjo, T. A hallmark of active class-switch recombination: transcripts directed by I promoters on looped-out circular DNAs. Proc. Natl Acad. Sci. USA 98, 12620–12623 (2001).
Nakamura, M. et al. High frequency class switching of an IgM+ B-lymphoma clone CH12F3 to IgA+ cells. Int. Immunol. 8, 193–201 (1996).
Stavnezer-Nordgren, J. & Sirlin, S. Specificity of immunoglobulin heavy-chain switch correlates with activity of germline heavy chain genes prior to switching. EMBO J. 5, 95–102 (1986).
Iwasato, T., Shimizu, A., Honjo, T. & Yamagishi, H. Circular DNA is excised by immunoglobulin class-switch recombination. Cell 62, 143–149 (1990).
von Schwedler, U., Jack, H. M. & Wabl, M. Circular DNA is a product of the immunoglobulin class-switch rearrangement. Nature 345, 452–456 (1990).
Matsuoka, M., Yoshida, K., Maeda, T., Usuda, S. & Sakano, H. Switch circular DNA formed in cytokine-treated mouse splenocytes: evidence for intramolecular DNA deletion in immunoglobulin class switching. Cell 62, 135–142 (1990).
Kraehenbuhl, J. P. & Neutra, M. R. Epithelial M cells: differentiation and function. Annu. Rev. Cell. Dev. Biol. 16, 301–332 (2000).
Rosner, A. J. & Keren, D. F. Demonstration of M cells in the specialized follicle-associated epithelium overlying isolated lymphoid follicles in the gut. J. Leukocyte Biol. 35, 397–404 (1984).
Maric, I., Holt, P. G., Perdue, M. H. & Bienenstock, J. Class II MHC antigen (Ia)-bearing dendritic cells in the epithelium of the rat intestine. J. Immunol. 156, 1408–1414 (1996).
Rescigno, M. et al. Dendritic cells express tight junction proteins and penetrate gut epithelial monolayers to sample bacteria. Nature Immunol. 2, 361–367 (2001). This paper describes a new mechanism by which lamina-propria dendritic cells sample bacteria directly from the gut lumen.
Farquhar, M. G. & Palade, G. E. Junctional complexes in various epithelia. J. Cell Biol. 17, 375–412 (1963).
Harriman, G. R., Kunimoto, D. Y., Elliott, J. F., Paetkau, V. & Strober, W. The role of IL-5 in IgA B-cell differentiation. J. Immunol. 140, 3033–3039 (1988).
Beagley, K. W. et al. Interleukins and IgA synthesis. Human and murine interleukin-6 induce high-rate IgA secretion in IgA-committed B cells. J. Exp. Med. 169, 2133–2148 (1989).
Coffman, R. L., Lebman, D. A. & Shrader, B. Transforming growth factor-β specifically enhances IgA production by lipopolysaccharide-stimulated murine B lymphocytes. J. Exp. Med. 170, 1039–1044 (1989).
Sonoda, E. et al. Transforming growth factor-β induces IgA production and acts additively with interleukin-5 for IgA production. J. Exp. Med. 170, 1415–1420 (1989).
van Ginkel, F. W. et al. Partial IgA deficiency with increased TH2-type cytokines in TGF-β1 knockout mice. J. Immunol. 163, 1951–1957 (1999).
Cazac, B. B. & Roes, J. TGF-β receptor controls B-cell responsiveness and induction of IgA in vivo. Immunity 13, 443–451 (2000).
Ramsay, A. J. et al. The role of interleukin-6 in mucosal IgA antibody responses in vivo. Science 264, 561–563 (1994).
Fayette, J. et al. Human dendritic cells skew isotype switching of CD40-activated naive B cells towards IgA1 and IgA2. J. Exp. Med. 185, 1909–1918 (1997).
Litinskiy, M. B. et al. DCs induce CD40-independent immunoglobulin class switching through BLyS and APRIL. Nature Immunol. 3, 822–829 (2002). The first demonstration that dendritic cells can trigger class-switch recombination of B cells directly, in a CD40-independent manner, through BAFF and APRIL.
Fagarasan, S. & Honjo, T. T-independent immune response: new aspects of B-cell biology. Science 290, 89–92 (2000).
Mestecky, J. et al. in Mucosal Immunology (eds Ogra, P. et al.) xxiii–xliii (Academic Press, San Diego, 1999).
Levy, Y. et al. Defect in IgV gene somatic hypermutation in common variable immuno-deficiency syndrome. Proc. Natl Acad. Sci. USA 95, 13135–13140 (1998).
Bastlein, C. et al. Common variable immunodeficiency syndrome and nodular lymphoid hyperplasia in the small intestine. Endoscopy 20, 272–275 (1988).
Burt, R. W. & Jacoby, R. F. in Textbook of Gastroenterology (eds Yamada, T., Alpers, D., Laine, L., Owyang, C. & Powell, D.) 1995–2022 (Lippincott William & Wilkins, Philadelphia, 1999).
Russell, M., Kilian, M. & Lamm, M. in Mucosal Immunology (eds Ogra, P. et al.) 225–240 (Academic Press, San Diego, 1999).
Harriman, G. R. et al. Targeted deletion of the IgA constant region in mice leads to IgA deficiency with alterations in expression of other Ig isotypes. J. Immunol. 162, 2521–2529 (1999).
Lycke, N., Erlandsson, L., Ekman, L., Schon, K. & Leanderson, T. Lack of J chain inhibits the transport of gut IgA and abrogates the development of intestinal antitoxic protection. J. Immunol. 163, 913–919 (1999).
Johansen, F. E. et al. Absence of epithelial immunoglobulin A transport, with increased mucosal leakiness, in polymeric immunoglobulin receptor/secretory component-deficient mice. J. Exp. Med. 190, 915–922 (1999).
Berek, C., Berger, A. & Apel, M. Maturation of the immune response in germinal centers. Cell 67, 1121–1129 (1991).
Gonzalez-Fernandez, A. & Milstein, C. Analysis of somatic hypermutation in mouse Peyer's patches using immunoglobulin κ light-chain transgenes. Proc. Natl Acad. Sci. USA 90, 9862–9866 (1993).
Nagaoka, H., Muramatsu, M., Yamamura, N., Kinoshita, K. & Honjo, T. Activation-induced deaminase (AID)-directed hypermutation in the immunoglobulin Sμ region: implication of AID involvement in a common step of class-switch recombination and somatic hypermutation. J. Exp. Med. 195, 529–534 (2002).
Kinoshita, K. & Honjo, T. Linking class-switch recombination with somatic hypermutation. Nature Rev. Mol. Cell. Biol. 2, 493–503 (2001).
Honjo, T., Kinoshita, K. & Muramatsu, M. Molecular mechanism of class-switch recombination: linkage with somatic hypermutation. Annu. Rev. Immunol. 20, 165–196 (2002).
Umesaki, Y. & Setoyama, H. Structure of the intestinal flora responsible for development of the gut immune system in a rodent model. Microbes Infect. 2, 1343–1351 (2000).
Fischer, M. & Kuppers, R. Human IgA- and IgM-secreting intestinal plasma cells carry heavily mutated VH region genes. Eur. J. Immunol. 28, 2971–2977 (1998).
Boursier, L., Dunn-Walters, D. K. & Spencer, J. Characteristics of IgVH genes used by human intestinal plasma cells from childhood. Immunology 97, 558–564 (1999).
Boursier, L., Dunn-Walters, D. K. & Spencer, J. Sequence analysis of light-chain genes from human intestinal plasma cells demonstrates that λ genes are almost all in-frame and highly mutated, and most κ genes are highly mutated when in-frame and minimally mutated when out-of-frame. Eur. J. Immunol. 30, 2908–2917 (2000).
Macpherson, A. J. et al. IgA production without μ or δ chain expression in developing B cells. Nature Immunol. 2, 625–631 (2001).
Kitamura, D., Roes, J., Kuhn, R. & Rajewsky, K. A B-cell-deficient mouse by targeted disruption of the membrane exon of the immunoglobulin μ chain gene. Nature 350, 423–426 (1991).
Lam, K. P., Kuhn, R. & Rajewsky, K. In vivo ablation of surface immunoglobulin on mature B cells by inducible gene targeting results in rapid cell death. Cell 90, 1073–1083 (1997).
Hooper, L. V. & Gordon, J. I. Commensal host–bacterial relationships in the gut. Science 292, 1115–1118 (2001).
Hooper, L. V. et al. Molecular analysis of commensal host–microbial relationships in the intestine. Science 291, 881–884 (2001).
Okazaki, I. M., Kinoshita, K., Muramatsu, M., Yoshikawa, K. & Honjo, T. The AID enzyme induces class-switch recombination in fibroblasts. Nature 416, 340–345 (2002).
Acknowledgements
This study was supported in part by a Center of Excellence Grant from the Ministry of Education, Science, Sports and Culture of Japan. We thank Y. Doi, K. Kinoshita, M. Muramatsu, H. Nagaoka and K. Suzuki for their contributions to both the work cited and the writing of this manuscript. Because of the extent and complexity of the mucosal immunology field, we could not discuss many interesting studies, and we apologize to those excellent scientists whose work could not be cited.
Author information
Authors and Affiliations
Corresponding author
Glossary
- J CHAIN
-
A polypeptide produced by immunocytes that is essential for the polymerization of immunoglobulin A and IgM, which is required for binding to the polymeric-immunoglobulin receptor and transport through epithelia.
- B2 CELLS
-
IgMlowIgDhiMac1−B220hiCD23+ cells that are produced continuously in adult bone marrow and secrete antibodies with high affinity and fine specificity.
- CLASS-SWITCH RECOMBINATION
-
(CSR). Alters the immunoglobulin heavy-chain constant-region (CH) gene that will be expressed from the Cμ region to one of the other CH genes. This results in a switch of immunoglobulin isotype from IgM/IgD to IgG, IgA or IgE, without altering antigen specificity.
- SOMATIC HYPERMUTATION
-
(SHM). Results in the accumulation of point mutations in the variable-region genes of immunoglobulin heavy and light chains. B cells that express high-affinity immunoglobulins on their surface are selected by limited amounts of the antigens, giving rise to high-affinity antibodies.
- B1 CELLS
-
Self-renewing IgMhiIgDlowMac1+B220loCD23− cells that are dominant in the peritoneal and pleural cavities. B1 cells recognize self-components, as well as common bacterial antigens, and they secrete antibodies that tend to have low affinity and broad specificity.
- NATURAL IgMS
-
These antibodies normally circulate in the blood of non-immunized mice. They are highly cross-reactive, and bind with low affinity to both microbial and self-antigens. A large proportion of natural IgMs is derived from peritoneal B1 cells.
- OMENTUM
-
A bilayered sheet of mesothelial cells connecting the spleen, pancreas, stomach and transverse colon, terminating in an 'apron-like' structure that contains adipocytes.
- M CELLS
-
(Microfold cells). Specialized epithelial cells that deliver antigens by transepithelial vesicular transport from the gut lumen directly to intraepithelial lymphocytes and to subepithelial lymphoid tissues.
- COMMON VARIABLE IMMUNODEFICIENCY SYNDROME
-
(CVID). The most common symptomatic primary antibody deficiency, characterized by decreased levels of serum immunoglobulin. Most patients suffer from recurrent infections, predominantly of the respiratory and gastrointestinal tracts. The incidence of malignancies, such as gastric carcinoma or lymphoma, is increased in patients with CVID.
- INTUSSUSCEPTION
-
The telescoping or prolapse of one portion of the intestine into an immediately adjacent segment.
- μMT−/− MICE
-
These mice carry a stop codon in the first membrane exon of the μ-chain constant region. They lack IgM+ B cells, and B-cell development is arrested at the pre-B-cell stage.
Rights and permissions
About this article
Cite this article
Fagarasan, S., Honjo, T. Intestinal IgA synthesis: regulation of front-line body defences. Nat Rev Immunol 3, 63–72 (2003). https://doi.org/10.1038/nri982
Issue Date:
DOI: https://doi.org/10.1038/nri982
This article is cited by
-
Bacteroides-derived isovaleric acid enhances mucosal immunity by facilitating intestinal IgA response in broilers
Journal of Animal Science and Biotechnology (2023)
-
A two-minute assay for electronic quantification of antibodies in saliva enabled through a reusable microfluidic multi-frequency impedance cytometer and machine learning analysis
Biomedical Microdevices (2023)
-
Gender dimorphism in IgA subclasses in T2-high asthma
Clinical and Experimental Medicine (2022)
-
Evaluation of Microencapsulated Synbiotic Preparations Containing Lactobionic Acid
Applied Biochemistry and Biotechnology (2021)
-
Humoral signatures of protective and pathological SARS-CoV-2 infection in children
Nature Medicine (2021)