Key Points
-
Toxoplasma gondii is a protozoan parasite that infects more than 1 billion people in the world. In addition to humans, T. gondii can infect all mammals and birds. Mice are a natural host for T. gondii and are a thoroughly studied animal model for T. gondii infection.
-
In mice, Toll-like receptor 11 (TLR11) is the principal innate immune sensor for T. gondii. TLR11 recognizes the unconventional actin-binding protein profilin, which is essential for parasite invasion into host cells.
-
In humans, TLR11 is a nonfunctional pseudogene. Consequently, the mechanisms through which the human immune system recognizes T. gondii are not well understood. Additional TLRs, in particular TLR2, TLR7 and TLR9, as well as the NLRP1 (NOD-, LRR- and pyrin domain-containing 1) inflammasome are all possible candidates for innate immune sensors that could be involved in human defence against T. gondii.
-
In a mouse model, activation of TLR11 and myeloid differentiation primary-response protein 88 (MYD88) in dendritic cells leads to the induction of interleukin-12 (IL-12) expression and the activation of interferon-γ (IFNγ) production by natural killer (NK) cells. In addition, both CD4+ T cell-derived and CD8+ T cell-derived IFNγ is essential for host resistance to the parasite.
-
An emerging source of IFNγ that does not require TLR-mediated parasite recognition is the neutrophil.
-
IFNγ mediates host protection via multiple mechanisms including induction of immunity-related GTPases (IRGs) and guanylate-binding proteins (GBPs). IFNγ also triggers the induction of the antimicrobial molecules nitric oxide and reactive oxygen species and is responsible for changes in host metabolism that restrict T. gondii replication.
Abstract
Toxoplasma gondii is a protozoan parasite of global importance. In the laboratory setting, T. gondii is frequently used as a model pathogen to study mechanisms of T helper 1 (TH1) cell-mediated immunity to intracellular infections. However, recent discoveries have shown that innate type 1 immune responses that involve interferon-γ (IFNγ)-producing natural killer (NK) cells and neutrophils, rather than IFNγ-producing T cells, predetermine host resistance to T. gondii. This Review summarizes the Toll-like receptor (TLR)-dependent mechanisms that are responsible for parasite recognition and for the induction of IFNγ production by NK cells, as well as the emerging data about the TLR-independent mechanisms that lead to the IFNγ-mediated elimination of T. gondii.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Jordan, K. A. & Hunter, C. A. Regulation of CD8+ T cell responses to infection with parasitic protozoa. Exp. Parasitol. 126, 318–325 (2010).
Gaddi, P. J. & Yap, G. S. Cytokine regulation of immunopathology in toxoplasmosis. Immunol. Cell Biol. 85, 155–159 (2007).
Denkers, E. Y. T lymphocyte-dependent effector mechanisms of immunity to Toxoplasma gondii. Microbes Infect. 1, 699–708 (1999).
Hunter, C. A. & Sibley, L. D. Modulation of innate immunity by Toxoplasma gondii virulence effectors. Nature Rev. Microbiol. 10, 766–778 (2012).
Takeuchi, O. & Akira, S. Pattern recognition receptors and inflammation. Cell 140, 805–820 (2010).
Yarovinsky, F. et al. TLR11 activation of dendritic cells by a protozoan profilin-like protein. Science 308, 1626–1629 (2005). This study defines the first protein derived from T. gondii that functions as an immunostimulatory ligand for mammalian TLRs, specifically for mouse TLR11; T. gondii profilin is the first identified ligand for TLR11.
Plattner, F. et al. Toxoplasma profilin is essential for host cell invasion and TLR11-dependent induction of an interleukin-12 response. Cell Host Microbe 3, 77–87 (2008). This study defines the essential biological function for T. gondii profilin in parasite invasion by using a parasite strain with conditional inactivation of proflin.
Campos, M. A. et al. Activation of Toll-like receptor-2 by glycosylphosphatidylinositol anchors from a protozoan parasite. J. Immunol. 167, 416–423 (2001).
Debierre-Grockiego, F. et al. Activation of TLR2 and TLR4 by glycosylphosphatidylinositols derived from Toxoplasma gondii. J. Immunol. 179, 1129–1137 (2007).
Benson, A., Pifer, R., Behrendt, C. L., Hooper, L. V. & Yarovinsky, F. Gut commensal bacteria direct a protective immune response against Toxoplasma gondii. Cell Host Microbe 6, 187–196 (2009). This study reveals that intestinal bacteria can induce protective immunity to T. gondii during a mucosal response to the parasite.
Scanga, C. A. et al. Cutting edge: MyD88 is required for resistance to Toxoplasma gondii infection and regulates parasite-induced IL-12 production by dendritic cells. J. Immunol. 168, 5997–6001 (2002). This study shows that the adaptor molecule MYD88, which is downstream of TLRs and IL-1 receptor, is essential for host resistance to T. gondii . It is also the first study that shows the susceptibility of MYD88-deficient mice to microbial infection.
Jankovic, D. et al. In the absence of IL-12, CD4+ T cell responses to intracellular pathogens fail to default to a TH2 pattern and are host protective in an IL-10−/− setting. Immunity 16, 429–439 (2002).
Yarovinsky, F., Kanzler, H., Hieny, S., Coffman, R. L. & Sher, A. Toll-like receptor recognition regulates immunodominance in an antimicrobial CD4+ T cell response. Immunity 25, 655–664 (2006).
Hou, B., Benson, A., Kuzmich, L., DeFranco, A. L. & Yarovinsky, F. Critical coordination of innate immune defense against Toxoplasma gondii by dendritic cells responding via their Toll-like receptors. Proc. Natl Acad. Sci. USA 108, 278–283 (2011). This study shows the role of TLR signalling in DCs and the regulation of protective immunity against T. gondii infection using a novel cell-specific MYD88 conditional knockout mouse model. The paper shows that TLR signalling in DCs is required for host protection through the IL-12-dependent activation of NK cells but not through T cell-derived IFNγ expression.
Janeway, C. A. Jr. Approaching the asymptote? Evolution and revolution in immunology. Cold Spring Harb. Symp. Quant. Biol. 1, 1–13 (1989).
Pifer, R., Benson, A., Sturge, C. R. & Yarovinsky, F. UNC93B1 is essential for TLR11 activation and IL-12-dependent host resistance to Toxoplasma gondii. J. Biol. Chem. 286, 3307–3314 (2011).
Pifer, R. & Yarovinsky, F. Innate responses to Toxoplasma gondii in mice and humans. Trends Parasitol. 27, 388–393 (2011).
Raetz, M. et al. Cooperation of TLR12 and TLR11 in the IRF8-dependent IL-12 response to Toxoplasma gondii profilin. J. Immunol. 191, 4818–4827 (2013).
Koblansky, A. A. et al. Recognition of profilin by Toll-like receptor 12 is critical for host resistance to Toxoplasma gondii. Immunity 38, 119–130 (2013).
Andrade, W. A. et al. Combined action of nucleic acid-sensing Toll-like receptors and TLR11/TLR12 heterodimers imparts resistance to Toxoplasma gondii in mice. Cell Host Microbe 13, 42–53 (2013).
Pepper, M. et al. Plasmacytoid dendritic cells are activated by Toxoplasma gondii to present antigen and produce cytokines. J. Immunol. 180, 6229–6236 (2008).
Melo, M. B. et al. UNC93B1 mediates host resistance to infection with Toxoplasma gondii. PLoS Pathog. 6, 1001071 (2010).
Diebold, S. S., Kaisho, T., Hemmi, H., Akira, S. & Reis e Sousa, C. Innate antiviral responses by means of TLR7-mediated recognition of single-stranded RNA. Science 303, 1529–1531 (2004).
Heil, F. et al. Species-specific recognition of single-stranded RNA via toll-like receptor 7 and 8. Science 303, 1526–1529 (2004).
Mancuso, G. et al. Bacterial recognition by TLR7 in the lysosomes of conventional dendritic cells. Nature Immunol. 10, 587–594 (2009).
Diebold, S. S. et al. Nucleic acid agonists for Toll-like receptor 7 are defined by the presence of uridine ribonucleotides. Eur. J. Immunol. 36, 3256–3267 (2006).
Pisitkun, P. et al. Autoreactive B cell responses to RNA-related antigens due to TLR7 gene duplication. Science 312, 1669–1672 (2006).
Edwards, A. D. et al. Toll-like receptor expression in murine DC subsets: lack of TLR7 expression by CD8α+ DC correlates with unresponsiveness to imidazoquinolines. Eur. J. Immunol. 33, 827–833 (2003).
Reis e Sousa, C. et al. In vivo microbial stimulation induces rapid CD40 ligand-independent production of interleukin 12 by dendritic cells and their redistribution to T cell areas. J. Exp. Med. 186, 1819–1829 (1997). This study reveals for the first time that CD8+ DCs are the major IL-12-producing cells in an immune response to T. gondii.
Hemmi, H. et al. A Toll-like receptor recognizes bacterial DNA. Nature 408, 740–745 (2000).
Haas, T. et al. The DNA sugar backbone 2′ deoxyribose determines toll-like receptor 9 activation. Immunity 28, 315–323 (2008).
Leadbetter, E. A. et al. Chromatin-IgG complexes activate B cells by dual engagement of IgM and Toll-like receptors. Nature 416, 603–607 (2002).
Christensen, S. R. et al. Toll-like receptor 9 controls anti-DNA autoantibody production in murine lupus. J. Exp. Med. 202, 321–331 (2005).
Gissot, M., Choi, S. W., Thompson, R. F., Greally, J. M. & Kim, K. Toxoplasma gondii and Cryptosporidium parvum lack detectable DNA cytosine methylation. Eukaryot. Cell 7, 537–540 (2008).
Coban, C. et al. Toll-like receptor 9 mediates innate immune activation by the malaria pigment hemozoin. J. Exp. Med. 201, 19–25 (2005).
Parroche, P. et al. Malaria hemozoin is immunologically inert but radically enhances innate responses by presenting malaria DNA to Toll-like receptor 9. Proc. Natl Acad. Sci. USA 104, 1919–1924 (2007).
Hall, J. A. et al. Commensal DNA limits regulatory T cell conversion and is a natural adjuvant of intestinal immune responses. Immunity 29, 637–649 (2008). References 10 and 37 show the adjuvant activity of the intestinal microbiota during oral T. gondii infection.
Rose, W. A., Sakamoto, K. & Leifer, C. A. TLR9 is important for protection against intestinal damage and for intestinal repair. Sci. Rep. 2, 14 (2012).
Egan, C. E., Cohen, S. B. & Denkers, E. Y. Insights into inflammatory bowel disease using Toxoplasma gondii as an infectious trigger. Immunol. Cell Biol. 90, 668–675 (2012).
Campos, M. A. et al. Impaired production of proinflammatory cytokines and host resistance to acute infection with Trypanosoma cruzi in mice lacking functional myeloid differentiation factor 88. J. Immunol. 172, 1711–1718 (2004).
Krishnegowda, G. et al. Induction of proinflammatory responses in macrophages by the glycosylphosphatidylinositols of Plasmodium falciparum: cell signaling receptors, glycosylphosphatidylinositol (GPI) structural requirement, and regulation of GPI activity. J. Biol. Chem. 280, 8606–8616 (2005).
Del Rio, L. et al. Toxoplasma gondii triggers myeloid differentiation factor 88-dependent IL-12 and chemokine ligand 2 (monocyte chemoattractant protein 1) responses using distinct parasite molecules and host receptors. J. Immunol. 172, 6954–6960 (2004).
Chang, H. R., Grau, G. E. & Pechere, J. C. Role of TNF and IL-1 in infections with Toxoplasma gondii. Immunology 69, 33–37 (1990).
Sibley, L. D., Adams, L. B., Fukutomi, Y. & Krahenbuhl, J. L. Tumor necrosis factor-α triggers antitoxoplasmal activity of IFN-γ primed macrophages. J. Immunol. 147, 2340–2345 (1991).
Deckert-Schluter, M., Bluethmann, H., Rang, A., Hof, H. & Schluter, D. Crucial role of TNF receptor type 1 (p55), but not of TNF receptor type 2 (p75), in murine toxoplasmosis. J. Immunol. 160, 3427–3436 (1998).
Yap, G. S., Scharton-Kersten, T., Charest, H. & Sher, A. Decreased resistance of TNF receptor p55- and p75-deficient mice to chronic toxoplasmosis despite normal activation of inducible nitric oxide synthase in vivo. J. Immunol. 160, 1340–1345 (1998).
Hunter, C. A., Chizzonite, R. & Remington, J. S. IL-1β is required for IL-12 to induce production of IFNγ by NK cells. A role for IL-1β in the T cell-independent mechanism of resistance against intracellular pathogens. J. Immunol. 155, 4347–4354 (1995). This study shows that IL-1 β , together with IL-12, is a central mediator for NK cell activation in response to T. gondii.
Ju, C. H., Chockalingam, A. & Leifer, C. A. Early response of mucosal epithelial cells during Toxoplasma gondii infection. J. Immunol. 183, 7420–7427 (2009).
Mun, H. S. et al. TLR2 as an essential molecule for protective immunity against Toxoplasma gondii infection. Int. Immunol. 15, 1081–1087 (2003).
Erridge, C. Endogenous ligands of TLR2 and TLR4: agonists or assistants? J. Leukoc. Biol. 87, 989–999 (2010).
Reiling, N. et al. Cutting edge: Toll-like receptor (TLR)2- and TLR4-mediated pathogen recognition in resistance to airborne infection with Mycobacterium tuberculosis. J. Immunol. 169, 3480–3484 (2002).
Debierre-Grockiego, F. & Schwarz, R. T. Immunological reactions in response to apicomplexan glycosylphosphatidylinositols. Glycobiology 20, 801–811 (2010).
Zhu, J., Krishnegowda, G., Li, G. & Gowda, D. C. Proinflammatory responses by glycosylphosphatidyl-inositols (GPIs) of Plasmodium falciparum are mainly mediated through the recognition of TLR2/TLR1. Exp. Parasitol. 128, 205–211 (2011).
Mun, H. S. et al. Toll-like receptor 4 mediates tolerance in macrophages stimulated with Toxoplasma gondii-derived heat shock protein 70. Infect. Immun. 73, 4634–4642 (2005).
Witola, W. H. et al. NALP1 influences susceptibility to human congenital toxoplasmosis, proinflammatory cytokine response, and fate of Toxoplasma gondii-infected monocytic cells. Infect. Immun. 79, 756–766 (2011).
Gov, L., Karimzadeh, A., Ueno, N. & Lodoen, M. B. Human innate immunity to Toxoplasma gondii is mediated by host caspase-1 and ASC and parasite GRA15. mBio 4, e00255–e00213 (2013).
Ewald, S. E., Chavarria-Smith, J. & Boothroyd, J. C. NLRP1 is an inflammasome sensor for Toxoplasma gondii. Infect. Immun. 11, 11 (2013).
Gazzinelli, R. T. et al. Parasite-induced IL-12 stimulates early IFNγ synthesis and resistance during acute infection with Toxoplasma gondii. J. Immunol. 153, 2533–2543 (1994). This study shows an essential role for IL-12 in the activation of IFNγ secretion by NK and CD4+ T cells.
Hunter, C. A., Candolfi, E., Subauste, C., Van Cleave, V. & Remington, J. S. Studies on the role of interleukin-12 in acute murine toxoplasmosis. Immunology 84, 16–20 (1995).
Khan, I. A., Matsuura, T. & Kasper, L. H. Interleukin-12 enhances murine survival against acute toxoplasmosis. Infect. Immun. 62, 1639–1642 (1994).
Scharton-Kersten, T. M., Yap, G., Magram, J. & Sher, A. Inducible nitric oxide is essential for host control of persistent but not acute infection with the intracellular pathogen Toxoplasma gondii. J. Exp. Med. 185, 1261–1273 (1997). This study establishes a role for iNOS in host resistance to T. gondii.
Lieberman, L. A. et al. IL-23 provides a limited mechanism of resistance to acute toxoplasmosis in the absence of IL-12. J. Immunol. 173, 1887–1893 (2004).
Trinchieri, G. Interleukin-12 and the regulation of innate resistance and adaptive immunity. Nature Rev. Immunol. 3, 133–146 (2003).
Scharton-Kersten, T. M. et al. In the absence of endogenous IFNγ, mice develop unimpaired IL-12 responses to Toxoplasma gondii while failing to control acute infection. J. Immunol. 157, 4045–4054 (1996). This study establishes that IL-12 functions upstream of IFNγ in vivo.
Scharton-Kersten, T., Contursi, C., Masumi, A., Sher, A. & Ozato, K. Interferon consensus sequence binding protein-deficient mice display impaired resistance to intracellular infection due to a primary defect in interleukin 12 p40 induction. J. Exp. Med. 186, 1523–1534 (1997). An essential role for IRF8 (also known as ICSBP) in the induction of IL-12 and host resistance to T. gondii is established in this study.
Mashayekhi, M. et al. CD8α+ dendritic cells are the critical source of interleukin-12 that controls acute infection by Toxoplasma gondii tachyzoites. Immunity 35, 249–259 (2011). This study reveals that IL-12 production is the major fucntion of CD8+ DCs in mediating host protection against T. gondii.
Honda, K. et al. Spatiotemporal regulation of MyD88–IRF-7 signalling for robust type-I interferon induction. Nature 434, 1035–1040 (2005).
Takaoka, A. et al. Integral role of IRF-5 in the gene induction programme activated by Toll-like receptors. Nature 434, 243–249 (2005).
Gazzinelli, R. T., Hieny, S., Wynn, T. A., Wolf, S. & Sher, A. Interleukin 12 is required for the T-lymphocyte-independent induction of interferon-γ by an intracellular parasite and induces resistance in T-cell-deficient hosts. Proc. Natl Acad. Sci. USA 90, 6115–6119 (1993). References 47, 58 and 69 establish IL-12 as a crucial cytokine that regulates NK cell IFNγ production.
Hunter, C. A., Subauste, C. S., Van Cleave, V. H. & Remington, J. S. Production of γ-interferon by natural killer cells from Toxoplasma gondii-infected SCID mice: regulation by interleukin-10, interleukin-12, and tumor necrosis factor-α. Infect. Immun. 62, 2818–2824 (1994).
Hildner, K. et al. Batf3 deficiency reveals a critical role for CD8α+ dendritic cells in cytotoxic T cell immunity. Science 322, 1097–1100 (2008).
Schiavoni, G. et al. ICSBP is essential for the development of mouse type I interferon-producing cells and for the generation and activation of CD8α+ dendritic cells. J. Exp. Med. 196, 1415–1425 (2002).
Tsujimura, H., Tamura, T. & Ozato, K. Cutting edge: IFN consensus sequence binding protein/IFN regulatory factor 8 drives the development of type I IFN-producing plasmacytoid dendritic cells. J. Immunol. 170, 1131–1135 (2003).
Tussiwand, R. et al. Compensatory dendritic cell development mediated by BATF–IRF interactions. Nature 490, 502–507 (2012).
Seillet, C. et al. CD8α+ DCs can be induced in the absence of transcription factors Id2, Nfil3, and Batf3. Blood 121, 1574–1583 (2013).
Iwasaki, A. & Medzhitov, R. Regulation of adaptive immunity by the innate immune system. Science 327, 291–295 (2010).
Blander, J. M. & Medzhitov, R. On regulation of phagosome maturation and antigen presentation. Nature Immunol. 7, 1029–1035 (2006).
West, M. A. et al. Enhanced dendritic cell antigen capture via Toll-like receptor-induced actin remodeling. Science 305, 1153–1157 (2004).
Atif, S. M., Uematsu, S., Akira, S. & McSorley, S. J. CD103−CD11b+ dendritic cells regulate the sensitivity of CD4 T-cell responses to bacterial flagellin. Mucosal Immunol. 1, 25 (2013).
Letran, S. E. et al. TLR5 functions as an endocytic receptor to enhance flagellin-specific adaptive immunity. Eur. J. Immunol. 41, 29–38 (2011).
Raetz, M. et al. Parasite-induced TH1 cells and intestinal dysbiosis cooperate in IFNγ-dependent elimination of Paneth cells. Nature Immunol. 14, 136–142 (2013). This study reveals that MYD88 signalling in T cells, but not in DCs, regulates T H 1 cell polarization.
Sukhumavasi, W. et al. TLR adaptor MyD88 is essential for pathogen control during oral Toxoplasma gondii infection but not adaptive immunity induced by a vaccine strain of the parasite. J. Immunol. 181, 3464–3473 (2008).
Sibley, L. D. Invasion and intracellular survival by protozoan parasites. Immunol. Rev. 240, 72–91 (2011).
Mordue, D. G., Hakansson, S., Niesman, I. & Sibley, L. D. Toxoplasma gondii resides in a vacuole that avoids fusion with host cell endocytic and exocytic vesicular trafficking pathways. Exp. Parasitol. 92, 87–99 (1999). This study reveals that parasitophorous vacuoles containing tachyzoites are not fused with host endolysosomal compartments.
McKee, A. S., Dzierszinski, F., Boes, M., Roos, D. S. & Pearce, E. J. Functional inactivation of immature dendritic cells by the intracellular parasite Toxoplasma gondii. J. Immunol. 173, 2632–2640 (2004).
Lang, C., Algner, M., Beinert, N., Gross, U. & Luder, C. G. Diverse mechanisms employed by Toxoplasma gondii to inhibit IFNγ-induced major histocompatibility complex class II gene expression. Microbes Infect. 8, 1994–2005 (2006).
Leng, J., Butcher, B. A. & Denkers, E. Y. Dysregulation of macrophage signal transduction by Toxoplasma gondii: past progress and recent advances. Parasite Immunol. 31, 717–728 (2009).
Mordue, D. G. & Sibley, L. D. Intracellular fate of vacuoles containing Toxoplasma gondii is determined at the time of formation and depends on the mechanism of entry. J. Immunol. 159, 4452–4459 (1997).
Gubbels, M. J., Striepen, B., Shastri, N., Turkoz, M. & Robey, E. A. Class I major histocompatibility complex presentation of antigens that escape from the parasitophorous vacuole of Toxoplasma gondii. Infect. Immun. 73, 703–711 (2005).
Dzierszinski, F. et al. Presentation of Toxoplasma gondii antigens via the endogenous major histocompatibility complex class I pathway in nonprofessional and professional antigen-presenting cells. Infect. Immun. 75, 5200–5209 (2007). References 89 and 90 establish that infected cells are responsible for the activation of CD8+ T cell responses to T. gondii.
Sinai, A. P., Webster, P. & Joiner, K. A. Association of host cell endoplasmic reticulum and mitochondria with the Toxoplasma gondii parasitophorous vacuole membrane: a high affinity interaction. J. Cell Sci. 110, 2117–2128 (1997).
Goldszmid, R. S. et al. Host ER-parasitophorous vacuole interaction provides a route of entry for antigen cross-presentation in Toxoplasma gondii-infected dendritic cells. J. Exp. Med. 206, 399–410 (2009).
Blanchard, N. et al. Immunodominant, protective response to the parasite Toxoplasma gondii requires antigen processing in the endoplasmic reticulum. Nature Immunol. 9, 937–944 (2008).
Gregg, B. et al. Subcellular antigen location influences T-cell activation during acute infection with Toxoplasma gondii. PLoS ONE 6, 28 (2011).
John, B. et al. Dynamic Imaging of CD8+ T cells and dendritic cells during infection with Toxoplasma gondii. PLoS Pathog. 5, 3 (2009).
Koshy, A. A. et al. Toxoplasma co-opts host cells it does not invade. PLoS Pathog. 8, 26 (2012). This study reveals that T. gondii can manipulate uninfected host cells by injecting proteins into their cytoplasm.
Wilson, D. C. et al. Differential regulation of effector- and central-memory responses to Toxoplasma gondii Infection by IL-12 revealed by tracking of Tgd057-specific CD8+ T cells. PLoS Pathog. 6, 1000815 (2010).
Yap, G., Pesin, M. & Sher, A. Cutting edge: IL-12 is required for the maintenance of IFNγ production in T cells mediating chronic resistance to the intracellular pathogen, Toxoplasma gondii. J. Immunol. 165, 628–631 (2000).
Scharton-Kersten, T., Caspar, P., Sher, A. & Denkers, E. Y. Toxoplasma gondii: evidence for interleukin-12-dependent and-independent pathways of interferon-γ production induced by an attenuated parasite strain. Exp. Parasitol. 84, 102–114 (1996).
Sturge, C. R. et al. TLR-independent neutrophil-derived IFNγ is important for host resistance to intracellular pathogens. Proc. Natl Acad. Sci. USA 110, 10711–10716 (2013). This study reveals that neutrophils are an important source of IFNγ, which is required for host resistance to the parasite in the absence of TLR11-mediated recogntion of profilin.
Yarovinsky, F., Hieny, S. & Sher, A. Recognition of Toxoplasma gondii by TLR11 prevents parasite-induced immunopathology. J. Immunol. 181, 8478–8484 (2008).
Nathan, C. F., Murray, H. W., Wiebe, M. E. & Rubin, B. Y. Identification of interferon-γ as the lymphokine that activates human macrophage oxidative metabolism and antimicrobial activity. J. Exp. Med. 158, 670–689 (1983). This study establishes that IFNγ is required and is sufficient for the induction of antimicrobial activity against T. gondii in human macrophages.
Murray, H. W., Gellene, R. A., Libby, D. M., Rothermel, C. D. & Rubin, B. Y. Activation of tissue macrophages from AIDS patients: in vitro response of AIDS alveolar macrophages to lymphokines and interferon-γ. J. Immunol. 135, 2374–2377 (1985).
Pfefferkorn, E. R. Interferon-γ blocks the growth of Toxoplasma gondii in human fibroblasts by inducing the host cells to degrade tryptophan. Proc. Natl Acad. Sci. USA 81, 908–912 (1984). This is a pioneering study that shows an IFNγ-mediated mechanism for the restriction of T. gondii growth through tryptophan starvation.
Pfefferkorn, E. R., Rebhun, S. & Eckel, M. Characterization of an indoleamine 2,3-dioxygenase induced by γ-interferon in cultured human fibroblasts. J. Interferon Res. 6, 267–279 (1986).
Ball, H. J. et al. Characterization of an indoleamine 2,3-dioxygenase-like protein found in humans and mice. Gene 396, 203–213 (2007).
Metz, R. et al. Novel tryptophan catabolic enzyme IDO2 is the preferred biochemical target of the antitumor indoleamine 2,3-dioxygenase inhibitory compound D-1-methyl-tryptophan. Cancer Res. 67, 7082–7087 (2007).
Divanovic, S. et al. Opposing biological functions of tryptophan catabolizing enzymes during intracellular infection. J. Infect. Dis. 205, 152–161 (2012).
Adams, L. B., Hibbs, J. B. Jr., Taintor, R. R. & Krahenbuhl, J. L. Microbiostatic effect of murine-activated macrophages for Toxoplasma gondii. Role for synthesis of inorganic nitrogen oxides from L-arginine. J. Immunol. 144, 2725–2729 (1990).
Fang, F. C. Antimicrobial reactive oxygen and nitrogen species: concepts and controversies. Nature Rev. Microbiol. 2, 820–832 (2004).
Fox, B. A., Gigley, J. P. & Bzik, D. J. Toxoplasma gondii lacks the enzymes required for de novo arginine biosynthesis and arginine starvation triggers cyst formation. Int. J. Parasitol. 34, 323–331 (2004).
Butcher, B. A. et al. Toxoplasma gondii rhoptry kinase ROP16 activates STAT3 and STAT6 resulting in cytokine inhibition and arginase-1-dependent growth control. PLoS Pathog. 7, 8 (2011).
El Kasmi, K. C. et al. Toll-like receptor-induced arginase 1 in macrophages thwarts effective immunity against intracellular pathogens. Nature Immunol. 9, 1399–1406 (2008).
Yap, G. S. & Sher, A. Effector cells of both nonhemopoietic and hemopoietic origin are required for interferon (IFN)-γ- and tumor necrosis factor (TNF)-α-dependent host resistance to the intracellular pathogen, Toxoplasma gondii. J. Exp. Med. 189, 1083–1092 (1999).
Schluter, D. et al. Inhibition of inducible nitric oxide synthase exacerbates chronic cerebral toxoplasmosis in Toxoplasma gondii-susceptible C57BL/6 mice but does not reactivate the latent disease in T. gondii-resistant BALB/c mice. J. Immunol. 162, 3512–3518 (1999).
Khan, I. A., Matsuura, T. & Kasper, L. H. Inducible nitric oxide synthase is not required for long-term vaccine-based immunity against Toxoplasma gondii. J. Immunol. 161, 2994–3000 (1998).
Suzuki, Y., Kang, H., Parmley, S., Lim, S. & Park, D. Induction of tumor necrosis factor-α and inducible nitric oxide synthase fails to prevent toxoplasmic encephalitis in the absence of interferon-γ in genetically resistant BALB/c mice. Microbes Infect. 2, 455–462 (2000).
Murray, H. W. & Teitelbaum, R. F. L-arginine-dependent reactive nitrogen intermediates and the antimicrobial effect of activated human mononuclear phagocytes. J. Infect. Dis. 165, 513–517 (1992).
Murray, H. W., Juangbhanich, C. W., Nathan, C. F. & Cohn, Z. A. Macrophage oxygen-dependent antimicrobial activity. II. The role of oxygen intermediates. J. Exp. Med. 150, 950–964 (1979).
Wilson, C. B., Tsai, V. & Remington, J. S. Failure to trigger the oxidative metabolic burst by normal macrophages: possible mechanism for survival of intracellular pathogens. J. Exp. Med. 151, 328–346 (1980).
Chang, H. R. & Pechere, J. C. Macrophage oxidative metabolism and intracellular Toxoplasma gondii. Microb. Pathog. 7, 37–44 (1989).
Taylor, G. A. & Feng, C. G, & Sher, A. p47 GTPases: regulators of immunity to intracellular pathogens. Nature Rev. Immunol. 4, 100–109 (2004).
Bekpen, C. et al. The interferon-inducible p47 (IRG) GTPases in vertebrates: loss of the cell autonomous resistance mechanism in the human lineage. Genome Biol. 6, 31 (2005).
Taylor, G. A. et al. Pathogen-specific loss of host resistance in mice lacking the IFNγ-inducible gene IGTP. Proc. Natl Acad. Sci. USA 97, 751–755 (2000).
Collazo, C. M. et al. Inactivation of LRG-47 and IRG-47 reveals a family of interferon gamma-inducible genes with essential, pathogen-specific roles in resistance to infection. J. Exp. Med. 194, 181–188 (2001). References 124 and 125 establish a crucial role for IRGs in mediating IFNγ-dependent host resistance T. gondii.
Liesenfeld, O. et al. The IFNγ-inducible GTPase, Irga6, protects mice against Toxoplasma gondii but not against Plasmodium berghei and some other intracellular pathogens. PLoS ONE 6, 17 (2011).
Fentress, S. J. et al. Phosphorylation of immunity-related GTPases by a Toxoplasma gondii-secreted kinase promotes macrophage survival and virulence. Cell Host Microbe 8, 484–495 (2010).
Zhao, Y. O., Khaminets, A., Hunn, J. P. & Howard, J. C. Disruption of the Toxoplasma gondii parasitophorous vacuole by IFNγ-inducible immunity-related GTPases (IRG proteins) triggers necrotic cell death. PLoS Pathog. 5, 6 (2009).
Martens, S. et al. Mechanisms regulating the positioning of mouse p47 resistance GTPases LRG-47 and IIGP1 on cellular membranes: retargeting to plasma membrane induced by phagocytosis. J. Immunol. 173, 2594–2606 (2004).
Butcher, B. A. et al. p47 GTPases regulate Toxoplasma gondii survival in activated macrophages. Infect. Immun. 73, 3278–3286 (2005).
Hunn, J. P. et al. Regulatory interactions between IRG resistance GTPases in the cellular response to Toxoplasma gondii. EMBO J. 27, 2495–2509 (2008).
Zhao, Y. O., Konen-Waisman, S., Taylor, G. A., Martens, S. & Howard, J. C. Localisation and mislocalisation of the interferon-inducible immunity-related GTPase, Irgm1 (LRG-47) in mouse cells. PLoS ONE 5, 0008648 (2010).
Liu, Z. et al. The immunity-related GTPase Irgm3 relieves endoplasmic reticulum stress response during coxsackievirus B3 infection via a PI3K/Akt dependent pathway. Cell. Microbiol. 14, 133–146 (2012).
Taylor, G. A. et al. The inducibly expressed GTPase localizes to the endoplasmic reticulum, independently of GTP binding. J. Biol. Chem. 272, 10639–10645 (1997).
Henry, S. C. et al. Balance of Irgm protein activities determines IFNγ-induced host defense. J. Leukoc. Biol. 85, 877–885 (2009).
Khaminets, A. et al. Coordinated loading of IRG resistance GTPases on to the Toxoplasma gondii parasitophorous vacuole. Cell. Microbiol. 12, 939–961 (2010).
Ling, Y. M. et al. Vacuolar and plasma membrane stripping and autophagic elimination of Toxoplasma gondii in primed effector macrophages. J. Exp. Med. 203, 2063–2071 (2006).
Martens, S. et al. Disruption of Toxoplasma gondii parasitophorous vacuoles by the mouse p47-resistance GTPases. PLoS Pathog. 1, 18 (2005). References 137 and 138 show how IRGs can eliminate T. gondii in infected cells.
Hunn, J. P. & Howard, J. C. The mouse resistance protein Irgm1 (LRG-47): a regulator or an effector of pathogen defense? PLoS Pathog. 6, 1001008 (2010).
Feng, C. G., Zheng, L., Lenardo, M. J. & Sher, A. Interferon-inducible immunity-related GTPase Irgm1 regulates IFNγ-dependent host defense, lymphocyte survival and autophagy. Autophagy 5, 232–234 (2009).
Henry, S. C. et al. Regulation of macrophage motility by Irgm1. J. Leukoc. Biol. 87, 333–343 (2010).
Feng, C. G. et al. The immunity-related GTPase Irgm1 promotes the expansion of activated CD4+ T cell populations by preventing interferon-γ-induced cell death. Nature Immunol. 9, 1279–1287 (2008). This study reveals a novel stem cell-protective function for IRGM1 in host resistance to microbial infection.
Feng, C. G., Weksberg, D. C., Taylor, G. A., Sher, A. & Goodell, M. A. The p47 GTPase Lrg-47 (Irgm1) links host defense and hematopoietic stem cell proliferation. Cell Stem Cell 2, 83–89 (2008).
Selleck, E. M. et al. Guanylate-binding protein 1 (Gbp1) contributes to cell-autonomous immunity against Toxoplasma gondii. PLoS Pathog. 9, 25 (2013).
Yamamoto, M. et al. ATF6β is a host cellular target of the Toxoplasma gondii virulence factor ROP18. J. Exp. Med. 208, 1533–1546 (2011).
Behnke, M. S. et al. The polymorphic pseudokinase ROP5 controls virulence in Toxoplasma gondii by regulating the active kinase ROP18. PLoS Pathog. 8, 8 (2012).
Steinfeldt, T. et al. Phosphorylation of mouse immunity-related GTPase (IRG) resistance proteins is an evasion strategy for virulent Toxoplasma gondii. PLoS Biol. 8, 1000576 (2010).
Bekpen, C., Xavier, R. J. & Eichler, E. E. Human IRGM gene “to be or not to be”. Semin. Immunopathol. 32, 437–444 (2010).
McCarroll, S. A. et al. Deletion polymorphism upstream of IRGM associated with altered IRGM expression and Crohn's disease. Nature Genet. 40, 1107–1112 (2008).
Degrandi, D. et al. Extensive characterization of IFN-induced GTPases mGBP1 to mGBP10 involved in host defense. J. Immunol. 179, 7729–7740 (2007).
Kresse, A. et al. Analyses of murine GBP homology clusters based on in silico, in vitro and in vivo studies. BMC Genomics 9, 1471–2164 (2008).
Yamamoto, M. et al. A cluster of interferon-γ-inducible p65 GTPases plays a critical role in host defense against Toxoplasma gondii. Immunity 37, 302–313 (2012).
Degrandi, D. et al. Murine guanylate binding protein 2 (mGBP2) controls Toxoplasma gondii replication. Proc. Natl Acad. Sci. USA 110, 294–299 (2013).
Haldar, A. K. et al. IRG and GBP host resistance factors target aberrant, “non-self” vacuoles characterized by the missing of “self” IRGM proteins. PLoS Pathog. 9, 13 (2013).
Roach, J. C. et al. The evolution of vertebrate Toll-like receptors. Proc. Natl Acad. Sci. USA 102, 9577–9582 (2005).
Melo, M. B., Jensen, K. D. & Saeij, J. P. Toxoplasma gondii effectors are master regulators of the inflammatory response. Trends Parasitol. 27, 487–495 (2011).
Acknowledgements
F.Y. gratefully thanks colleagues and the members of his laboratory for the many discussions that contributed to this manuscript. Work in F.Y.'s laboratory is supported by the US National Institutes of Health (AI085263) and the Burroughs Wellcome Fund.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The author declares no competing financial interests.
Glossary
- Apicomplexan parasite
-
A member of a large group of protozoan parasites possessing a unique apical complex structure that is involved in host cell invasion. This phylogenetic group includes more than 5,000 species, all of which are parasitic. The best known examples of apicomplexan parasites are Plasmodium spp. (the cause of malaria), Toxoplasma gondii and Cryptosporidium spp.
- Oocysts
-
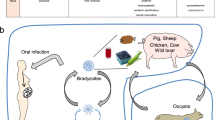
Ovoid structures that contain two sporocysts, each of which contains four sporozoites. The sporozoites may then become tachyzoites or bradyzoites. Cats shed faecal Toxoplasma gondii oocysts in the soil, grass and water. The oocyst wall is an extremely resistant multilayer structure that protects the parasite from mechanical and chemical damage, which enables the parasite to survive for long periods of time.
- Parasitophorous vacuole
-
A vacuole surrounded by a parasitophorous vacuolar membrane (PVM), which forms as a result of the invasion of Toxoplasma gondii into the host cell. The PVM is formed during active invasion of the host cell and depends on actin polymerization in T. gondii, but not in the host. The physical force created by the parasite initiates the formation of a membrane, which surrounds the intracellular parasite within the parasitophorous vacuole and which differs from endosomal or phagolysosomal membranes.
- Soluble tachyzoite antigen
-
(STAg). A soluble extract from Toxoplasma gondii that is obtained by sonication of the parasites.
- Congenital toxoplasmosis
-
A disease that occurs when a developing child is infected with the parasite Toxoplasma gondii. The developing child can be infected during pregnancy, labour or delivery.
- Bradyzoites
-
A slow replicating form of Toxoplasma gondii. Bradyzoites in the form of cysts are responsible for the chronic stage of T. gondii infection.
- Cysts
-
Structures that contain bradyzoites. Cysts grow and remain intracellular despite variations in their size. Young cysts may be as small as 5μm and may contain only two bradyzoites, whereas older cysts may contain hundreds of bradyzoites.
- 'Non-professional' APCs
-
Cells that do not constitutively express MHC class II molecules, but that upregulate MHC class II following stimulation with certain cytokines, particularly interferon-γ. Important examples of non-professional APCs include fibroblasts and thymic epithelial cells.
- Tachyzoites
-
Fast-replicating forms of Toxoplasma gondii that are responsible for the acute stage of infection.
- Sexual cycle
-
Sexual replication of Toxoplasma gondii occurs in the gut of the cat during the enteroepithelial stage of the parasite life cycle, which takes about 310 days. Sexual replication leads to the production of oocysts. A host in which parasites sexually reproduce is known as the definitive, final or primary host.
Rights and permissions
About this article
Cite this article
Yarovinsky, F. Innate immunity to Toxoplasma gondii infection. Nat Rev Immunol 14, 109–121 (2014). https://doi.org/10.1038/nri3598
Published:
Issue Date:
DOI: https://doi.org/10.1038/nri3598
This article is cited by
-
Dynamic RNA profiles in the small intestinal epithelia of cats after Toxoplasma gondii infection
Infectious Diseases of Poverty (2023)
-
Transcriptome analysis of a newly established mouse model of Toxoplasma gondii pneumonia
Parasites & Vectors (2023)
-
Curcumin modulates neurogliogenesis and purinergic receptor expression in neural precursor cells infected with Toxoplasma gondii
Parasitology Research (2023)
-
Functional characterization of goose IRF1 in IFN induction and anti-NDV infection
Veterinary Research (2022)
-
B7-H4 reduction induced by Toxoplasma gondii infection results in dysfunction of decidual dendritic cells by regulating the JAK2/STAT3 pathway
Parasites & Vectors (2022)