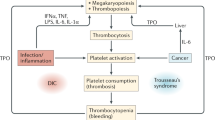
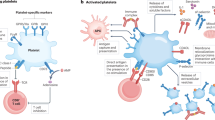
Key Points
-
Platelets have a plethora of soluble and cell-associated immunomodulatory molecules that can enhance and in some instances inhibit immune responses in a variety of diverse settings.
-
Platelets are naturally adhesive and can interact with various cells of the innate and adaptive immune system.
-
Platelets store preformed and synthesized immunomodulatory molecules that can markedly affect immune responses. These molecules include transforming growth factor-β, interleukin-1, platelet-derived growth factor and CC-chemokine ligand 5 (also known as RANTES).
-
Platelets express and secrete CD40 and CD154 and are the largest source of soluble CD154, which can affect both dendritic cell maturation and T cell activation.
-
Platelets express Toll-like receptors and actively bind to microorganisms, and in some instances can be directly cytopathic against bacteria. This, together with their large numbers within the circulation, makes them the ultimate bacterial sentinels in the bloodstream.
-
Platelets have been shown to have a central role in the pathogenesis of a diverse array of immune-mediated and infectious diseases, such as atherosclerosis, sepsis-induced lung damage, rheumatoid arthritis and malaria.
Abstract
Platelets are anucleate cells that are crucial mediators of haemostasis. Most immunologists probably don't think about platelets every day, and may even consider these cells to be 'nuisances' in certain in vitro studies. However, it is becoming increasingly clear that platelets have inflammatory functions and can influence both innate and adaptive immune responses. Here, we discuss the mechanisms by which platelets contribute to immunity: these small cells are more immunologically savvy than we once thought.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Bizzozero, G. Sur un nouvel èlèment morphologique du sang chez les mammiferes et son importance dans la thrombose et dans la coagulation. Arch. Ital. Biol. 1, 1–5 (1882).
Gawaz, M. et al. Platelets in inflammation and atherogenesis. J. Clin. Invest. 115, 3378–3384 (2005).
Nieswandt, B. et al. Integrins in platelet activation. J. Thromb. Haemost. 7 (Suppl. 1), 206–209 (2009).
Smyth, S. S. et al. Platelet functions beyond hemostasis. J. Thromb. Haemost. 7, 1759–1766 (2009).
Kaushansky, K. Lineage-specific hematopoietic growth factors. N. Engl. J. Med. 354, 2034–2045 (2006).
Italiano, J. E. Jr & Hartwig, J. H. in Platelets 2nd edn (eds Michelson, A. D. & Coller, B. S.) 23–44 (Elsevier, Amsterdam, 2007).
Levin, J. in Platelets 2nd edn (eds Michelson, A. D. & Coller, B. S.) 3–22 (Elsevier, Amsterdam, 2007).
Hose, J. E., Martin, G. G. & Gerard, A. S. A decapod hemocyte classification scheme integrating morphology, cytochemistry, and function. Biol. Bull. 178, 33–45 (1990).
Götz, P. & Boman, H. G. in Comprehensive Insect Physiology, Biochemistry and Pharmacology (eds Kerkut, G. A. & Gilbert, L. I.) 453–485 (Pergamon, Oxford, 1985).
Thon, J. N. & Italiano, J. E. Jr. Platelet formation. Semin. Hematol. 47, 220–226 (2010).
Patel, S. R. et al. Differential roles of microtubule assembly and sliding in proplatelet formation by megakaryocytes. Blood. 106, 4076–4085 (2005).
Thon, J. N. et al. Cytoskeletal mechanics of proplatelet maturation and platelet release. J. Cell Biol. 191, 861–874 (2010).
Hartwig, J. Mechanisms of actin rearrangements mediating platelet activation. J. Cell Biol. 118, 1421–1442 (1992).
Diacovo, T. G., Puri, K. D., Warnock, R. A., Springer, T. A. & von Andrian, U. H. Platelet-mediated lymphocyte delivery to high endothelial venules. Science 273, 252–255 (1996).
Italiano, J. E. Jr et al. Angiogenesis is regulated by a novel mechanism: pro- and antiangiogenic proteins are organized into separate platelet α granules and differentially released. Blood. 111, 1227–1233 (2008).
Sehgal, S. & Storrie, B. Evidence that differential packaging of the major platelet granule proteins von Willebrand factor and fibrinogen can support their differential release. J. Thromb. Haemost. 5, 2009–2016 (2007).
White, G. C. & Rompietti, R. Platelet secretion: indiscriminately spewed forth or highly orchestrated? J. Thromb. Haemost. 5, 2006–2008 (2007).
Wicki, A. N. et al. Isolation and characterization of human blood platelet mRNA and construction of a cDNA library in λgt11. Confirmation of the platelet derivation by identification of GPIb coding mRNA and cloning of a GPIb coding cDNA insert. Thromb. Haemost. 61, 448–453 (1989).
Shashkin, P. N., Brown, G. T., Ghosh, A., Marathe, G. K. & McIntyre, T. M. Lipopolysaccharide is a direct agonist for platelet RNA splicing. J. Immunol. 181, 3495–3502 (2008).
Lindemann, S. & Gawaz, M. The active platelet: translation and protein synthesis in an anucleate cell. Semin. Thromb. Hemost. 33, 144–150 (2007).
Denis, M. M. et al. Escaping the nuclear confines: signal-dependent pre-mRNA splicing in anucleate platelets. Cell 122, 379–391 (2005).
Coppinger, J. A. Moderation of the platelet releasate response by aspirin. Blood 109, 4786–4792 (2007).
Mazzucco, L., Borzini, P. & Gope, R. Platelet-derived factors involved in tissue repair — from signal to function. Transfus. Med. Rev. 24, 218–234 (2010).
Assoian, R. K., Komoriya, A., Meyers, C. A., Miller, D. M. & Sporn, M. B. Transforming growth factor-β in human platelets: identification of a major storage site, purification, and characterization. J. Biol. Chem. 258, 7155–7160 (1983).
Andersson, P. O., Stockelberg, D., Jacobsson, S. & Wadenvik, H. A transforming growth factor-β1-mediated bystander immune suppression could be associated with remission of chronic idiopathic thrombocytopenic purpura. Ann. Hematol. 79, 507–513 (2000).
Andersson, P. O., Olsson, A. & Wadenvik, H. Reduced transforming growth factor-β1 production by mononuclear cells from patients with active chronic idiopathic thrombocytopenic purpura. Br. J. Haematol. 116, 862–867 (2002).
Ling, Y., Cao, X., Yu, Z. & Ruan, C. Circulating dendritic cells subsets and CD4+Foxp3+ regulatory T cells in adult patients with chronic ITP before and after treatment with high-dose dexamethasome. Eur. J. Haematol. 79, 310–316 (2007).
Liu, B. et al. Abnormality of CD4+CD25+ regulatory T cells in idiopathic thrombocytopenic purpura. Eur. J. Haematol. 78, 139–143 (2007).
Stasi, R. et al. Analysis of regulatory T cell changes in patients with idiopathic thrombocytopenic purpura receiving B-cell depleting therapy with rituximab. Blood 112, 1147–1150 (2008).
Olsson, B., Ridell, B., Carlsson, L., Jacobsson, S. & Wadenvik, H. Recruitment of T cells into bone marrow of ITP patients possibly due to elevated expression of VLA-4 and CX3CR1. Blood 12, 1078–1084 (2008).
Yu, J. et al. Defective circulating CD25 regulatory T cells in patients with chronic immune thrombocytopenic purpura. Blood 112, 1325–1328 (2008).
Grewal, I. S. & Flavell, R. A. CD40 and CD154 in cell-mediated immunity. Annu. Rev. Immunol. 16, 111–135 (1998).
Henn, V. et al. CD40 ligand on activated platelets triggers an inflammatory reaction of endothelial cells. Nature 391, 591–594 (1998).
Andre, P., Nannizzi-Alaimo, L., Prasad, S. K. & Phillips, D. R. Platelet-derived CD40L: the switch-hitting player of cardiovascular disease. Circulation 106, 896–899 (2002).
Hammwöhner, M. et al. Platelet expression of CD40/CD40 ligand and its relation to inflammatory markers and adhesion molecules in patients with atrial fibrillation. Exp. Biol. Med. 232, 581–589 (2007).
Anand, S. X., Viles-Gonzalez, J. F. & Badimon, J. J. Membrane-associated CD40L and sCD40L in atherothrombotic disease. Thromb. Haemost. 90, 377–384 (2003).
Henn, V., Steinbach, S., Büchner, K., Presek, P. & Kroczek, R. A. The inflammatory action of CD40 ligand (CD154) expressed on activated human platelets is temporally limited by coexpressed CD40. Blood 98, 1047–1054 (2001).
von Hundelshausen, P. & Weber, C. Platelets as immune cells: bridging inflammation and cardiovascular disease. Circ. Res. 100, 27–40 (2007).
Elzey, B. D. et al. Platelet-mediated modulation of adaptive immunity. A communication link between innate and adaptive immune compartments. Immunity 19, 9–19 (2003).
Sprague, D. L. et al. Platelet-mediated modulation of adaptive immunity: unique delivery of CD154 signal by platelet-derived membrane vesicles. Blood 111, 5028–5036 (2008).
Elzey, B. D. et al. Platelet-derived CD154 enables T-cell priming and protection against Listeria monocytogenes challenge. Blood 111, 3684–3691 (2008).
Iannacone, M. et al. Platelets mediate cytotoxic T lymphocyte-induced liver damage. Nature Med. 11, 1167–1169 (2005).
Kissel, K. et al. Human platelets target dendritic cell differentiation and production of proinflammatory cytokines. Transfusion 46, 818–827 (2006).
Janeway, C. A. Jr. The immune system evolved to discriminate infectious non-self from non-infectious self. Immunol. Today 13, 11–15 (1992).
Janeway, C. A. Jr & Medzhitov, R. Innate immune recognition. Annu. Rev. Immunol. 20, 197–216 (2002).
Scott, T. & Owens, M. D. Thrombocytes respond to lipopolysaccharide through Toll-like receptor-4, and MAP kinase and NF-κB pathways leading to expression of interleukin-6 and cyclooxygenase-2 with production of prostaglandin E2. Mol. Immunol. 45, 1001–1008 (2008).
Kuckleburg, C. J., Tiwari, R. & Czuprynski, C. J. Endothelial cell apoptosis induced by bacteria-activated platelets requires caspase-8 and -9 and generation of reactive oxygen species. Thromb. Haemost. 99, 363–372 (2008).
Kuckleburg, C. J., McClenahan, D. J. & Czuprynski, C. J. Platelet activation by Histophilus somni and its lipooligosaccharide induces endothelial cell proinflammatory responses and platelet internalization. Shock 29, 189–196 (2008).
Aslam, R., Freedman, J. & Semple, J. W. Murine platelets express Toll-like receptor 2: a potential regulator of innate and adaptive immunity. Platelets 15, 267–269 (2004).
Shiraki, R. et al. Expression of Toll-like receptors on human platelets. Thromb. Res. 113, 379–385 (2004).
Andonegui, G. et al. Platelets express functional Toll-like receptor-4. Blood 106, 2417–2423 (2005).
Cognasse, F. et al. Evidence of Toll-like receptor molecules on human platelets. Immunol. Cell Biol. 88, 196–198 (2005).
Aslam, R. et al. Platelet Toll-like receptor expression modulates lipopolysaccharide-induced thrombocytopenia and tumor necrosis factor-α production in vivo. Blood 107, 637–641 (2006).
Semple, J. W., Aslam, R., Kim, M., Speck, E. R. & Freedman, J. Platelet-bound lipopolysaccharide enhances Fc receptor-mediated phagocytosis of IgG opsonized platelets. Blood 109, 4803–4805 (2007).
Patrignani, P. et al. Reduced thromboxane biosynthesis in carriers of Toll-like receptor 4 polymorphisms in vivo. Blood 107, 3572–3574 (2006).
Ståhl, A. L. et al. Lipopolysaccharide from enterohemorrhagic Escherichia coli binds to platelets via TLR4 and CD62 and is detected on circulating platelets in patients with hemolytic uremic syndrome. Blood 108, 167–176 (2006).
Zhang, G. et al. Lipopolysaccharide stimulates platelet secretion and potentiates platelet aggregation via TLR4/MyD88 and the cGMP-dependent protein kinase pathway. J. Immunol. 182, 7997–8004 (2009).
Welbourn, C. R. & Young, Y. Endotoxin, septic shock and acute lung injury: neutrophils, macrophages and inflammatory mediators. Br. J. Surg. 79, 998–1003 (1992).
McClenahan, D. J., Evanson, O. A., Walcheck, B. K. & Weiss, D. J. Association among filamentous actin content, CD11b expression, and membrane deformability in stimulated and unstimulated bovine neutrophils. Amer. J. Vet. Res. 61, 380–386 (2000).
Ma, A. C. & Kubes, P. Platelets, neutrophils, and neutrophil extracellular traps (NETs) in sepsis. J. Thromb. Haemost. 6, 415–420 (2008).
Clark, S. R. et al. Platelet TLR4 activates neutrophil extracellular traps to ensnare bacteria in septic blood. Nature Med. 13, 463–469 (2007). This elegant study provided evidence that platelets act as sensors of bacterial infection by demonstrating that activation of platelet TLR4 stimulates the binding of activated platelets to neutrophils, leading to the release of NETs to catch and kill bacteria.
Brinkmann, V. et al. Neutrophil extracellular traps kill bacteria. Science 303, 1532–1535 (2004).
Klesney-Tait, J., Turnbull, I. R. & Colonna, M. The TREM receptor family and signal integration. Nature Immunol. 7, 1266–1273 (2006).
Haselmayer, P., Grosse-Hovest, L., von Landenberg, P., Schild, H. & Radsak, M. P. TREM-1 ligand expression on platelets enhances neutrophil activation. Blood 110, 1029–1035 (2007). In this article, the authors demonstrated that platelets express the ligand for TREM1, and showed that the binding of this molecule to neutrophil TREM1 augments LPS-induced neutrophil activation, and ultimately the innate inflammatory response.
Washington, A. V. et al. TREM-like transcript-1 protects against inflammation-associated hemorrhage by facilitating platelet aggregation in mice and humans. J. Clin. Invest. 119, 1489–1501 (2009).
Ross, R. Atherosclerosis: an inflammatory disease. N. Engl. J. Med. 340, 115–126 (1999).
Dong, Z. M., Brown, A. A. & Wagner, D. D. Prominent role of P-selectin in the development of advanced atherosclerosis in ApoE-deficient mice. Circulation 101, 2290–2295 (2000).
Burger, P. C. & Wagner, D. D. Platelet P-selectin facilitates atherosclerotic lesion development. Blood 101, 2661–2666 (2003).
Lindemann, S. et al. Activated platelets mediate inflammatory signaling by regulated interleukin 1β synthesis. J. Cell Biol. 154, 485–490 (2001).
Langer, H. F. & Gawaz, M. P. Platelet-vessel wall interactions in atherosclerotic disease. Thromb. Haemost. 99, 480–486 (2008).
Davi, G. & Patrono, C. Platelet activation and atherothrombosis. N. Engl. J. Med. 357, 2482–2494 (2007).
Langer, H. F. et al. Platelets recruit human dendritic cells via Mac-1/JAM-C interaction and modulate dendritic cell function in vitro. Arterioscler. Thromb. Vasc. Biol. 27, 1463–1470 (2007).
Lievens, D. et al. Platelet CD40L mediates thrombotic and inflammatory processes in atherosclerosis. Blood 116, 4317–4327 (2010).
Langer, H. F., Baughman, R. P., Lower, E. E., Flessa, H. C. & Tollerud, D. J. Thrombocytopenia in the intensive care unit. Chest 104, 1243–1247 (1993).
Gawaz, M., Dickfeld, T., Bogner, C., Fateh-Moghadam, S. & Neumann, F. J. Platelet function in septic multiple organ dysfunction syndrome. Intensive Care Med. 23, 379–385 (1997).
Jacoby, R. C. et al. Platelet activation and function after trauma. J. Trauma 51, 639–647 (2001).
Ogura, H. et al. Activated platelets enhance microparticle formation and platelet–leukocyte interaction in severe trauma and sepsis. J. Trauma 50, 801–809 (2001).
Mause, S. F., von Hundelshausen, P., Zernecke, A., Koenen, R. R. & Weber, C. Platelet microparticles: a transcellular delivery system for RANTES promoting monocyte recruitment on endothelium. Arterioscler. Thromb. Vasc. Biol. 25, 1512–1518 (2005).
Boman, H. G. Peptide antibiotics and their role in innate immunity. Annu. Rev. Immunol. 13, 61–92 (1995).
Yeaman, M. R., Puentes, S. M., Norman, D. C. & Bayer, A. S. Partial characterization and staphylocidal activity of thrombin-induced platelet microbicidal protein. Infect. Immun. 60, 1202–1209 (1992).
Krijgsveld, J. et al. Thrombocidins, microbicidal proteins from human blood platelets, are C-terminal deletion products of CXC chemokines. J. Biol. Chem. 275, 20374–20381 (2000).
Cox, D. & McConkey, S. The role of platelets in the pathogenesis of cerebral malaria. Cell. Mol. Life Sci. 67, 557–568 (2010).
McMorran, B. J. et al. Platelets kill intraerythrocytic malarial parasites and mediate survival to infection. Science 323, 797–800 (2009). This elegant study demonstrated that platelets can kill parasites in vitro and in mice, and found that platelet-deficient mice are more likely to die from malaria than mice with normal platelet counts. In addition, it showed that a single dose of aspirin may interfere with platelets sufficiently to prevent their killing power.
Choy, E. H. & Panayi, G. S. Cytokine pathways and joint inflammation in rheumatoid arthritis. N. Engl. J. Med. 344, 907–916 (2001).
Knijff-Dutmer, E. A., Koerts, J., Nieuwland, R., Kalsbeek-Batenburg, E. M. & van de Laar, M. A. Elevated levels of platelet microparticles are associated with disease activity in rheumatoid arthritis. Arthritis Rheum. 46, 1498–1503 (2002).
Boilard, E. et al. Platelets amplify inflammation in arthritis via collagen-dependent microparticle production. Science 327, 580–583 (2010). This interesting study described an essential role for platelet-derived microparticles in the development of the autoimmune disease rheumatoid arthritis, and identified the collagen receptor glycoprotein VI as a key stimulator of microparticle generation.
Looney, M. R., Gropper, M. A. & Matthay, M. A. Transfusion-related acute lung injury: a review. Chest 126, 249–258 (2004).
Cognasse, F., Lafarge, S., Chavarin, P., Acquart, S. & Garraud, O. Lipopolysaccharide induces sCD40L release through human platelets TLR4, but not TLR2 and TLR9. Intensive Care Med. 33, 382–384 (2007).
Khan, S. Y. et al. Soluble CD40 ligand accumulates in stored blood components, primes neutrophils through CD40, and is a potential cofactor in the development of transfusion-related acute lung injury. Blood 108, 2455–2462 (2006).
Cines, D. B., Bussel, J. B., Liebman, H. A. & Luning Prak, E. T. The ITP syndrome: pathogenic and clinical diversity. Blood 113, 6511–6521 (2009).
Semple, J. W., Provan, D., Garvey, M. B. & Freedman, J. Recent progress in understanding the pathogenesis of immune thrombocytopenia (ITP). Curr. Opin. Haematol. 17, 590–595 (2010).
Semple, J. W. Infections, antigen presenting cells, T cells and immune tolerance: their role in the pathogenesis of immune thrombocytopenia (ITP). Hematol. Oncol. Clin. North Am. 23, 1177–1192 (2009).
Harrington, W. J., Minnich, V., Hollingsworth, J. W. & Moore, C. V. Demonstration of a thrombocytopenic factor in the blood of patients with thrombocytopenic purpura. J. Lab. Clin. Med. 38, 1–10 (1951).
Evans, R. S., Takahashi, K., Duane, R. T., Payne, R. & Liu, C. Primary thrombocytopenic purpura and acquired hemolytic anemia; evidence for a common etiology. Arch. Intern. Med. 87, 48–65 (1951).
Nardi, M., Tomlinson, S., Greco, M. & Karpatkin, S. Complement-independent, peroxide-induced antibody lysis of platelets in HIV-1-related immune thrombocytopenia. Cell 106, 551–561 (2000).
Olsson, B. et al. T-cell-mediated cytotoxicity toward platelets in chronic idiopathic thrombocytopenic purpura. Nature Med. 9, 1123–1124 (2003).
Delaflor-Weiss, E. & Mintz, P. D. The evaluation and management of platelet refractoriness and alloimmunization. Transfus. Med. Rev. 14, 180–196 (2000).
Landau, M. & Rosenberg, N. Molecular insight into human platelet antigens: structural and evolutionary conservation analyses offer new perspective to immunogenic disorders. Transfusion 51, 558–569 (2011).
Kaplan C. Neonatal alloimmune thrombocytopenia: a 50-year story. Immunohematology 23, 9–13 (2007).
Shtalrid, M. et al. Post-transfusion purpura: a challenging diagnosis. Isr. Med. Assoc. J. 8, 672–674 (2006).
Kao, K. J., Cook, D. J. & Scornik, J. C. Quantitative analysis of platelet surface HLA by W6/32 anti-HLA monoclonal antibody. Blood 68, 627–632 (1986).
Gouttefangeas, C. et al. Thrombocyte HLA molecules retain nonrenewable endogenous peptides of megakaryocyte lineage and do not stimulate direct allocytotoxicity in vitro. Blood 95, 3168–3175 (2000).
Aslam, R., Speck, E. R., Kim, M., Freedman, J. & Semple, J. W. Transfusion-related immunomodulation (TRIM) by platelets is dependent on their expression of MHC class I molecules and is independent of leukocytes. Transfusion 48, 1778–1786 (2008).
Chow, L. et al. A novel mouse model demonstrating both antibody- and T cell-mediated thrombocytopenia: differential response to therapy. Blood 115, 1247–1253 (2010).
Weyrich, A. S. & Zimmerman, G. A. Platelets: signaling cells in the immune continuum. Trends Immunol. 25, 489–495 (2004).
Acknowledgements
The authors thank A. H. Lazarus, H. Ni and V. Leytin for their helpful discussions and advice.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Glossary
- Megakaryocyte
-
A giant multinucleated cell of the bone marrow that gives rise to platelets.
- Thrombocytopenia
-
Any disorder in which there is an abnormally low number of platelets (below 150,000 platelets per microlitre of blood). Thrombocytopenia is caused either by increased breakdown of platelets (in the blood, spleen or liver) or by low production of platelets.
- Immune thrombocytopenia
-
An abnormal drop in platelet numbers caused by the presence of platelet-specific antibodies. Platelet production may be normal or impaired, as the disorder can also be caused by antibodies directed against megakaryocytes. The term includes autoimmune, alloimmune and certain drug-induced thrombocytopenias.
- Toll-like receptors
-
A family of pattern recognition receptors that have essential roles in innate immunity. They are a class of single-membrane-spanning receptors that have the ability to recognize structurally conserved molecules from bacteria. Engagement of Toll-like receptors activates the immune response.
- Tissue factor
-
The major cellular initiator of clot formation. It is essential for generating thrombin from the zymogen prothrombin.
- Neutrophil extracellular traps
-
Networks of extracellular fibres, produced by neutrophils, that catch and help kill pathogens. The extracellular fibril matrix is composed of decondensed chromatin.
Rights and permissions
About this article
Cite this article
Semple, J., Italiano, J. & Freedman, J. Platelets and the immune continuum. Nat Rev Immunol 11, 264–274 (2011). https://doi.org/10.1038/nri2956
Published:
Issue Date:
DOI: https://doi.org/10.1038/nri2956
This article is cited by
-
EGFR of platelet regulates macrophage activation and bacterial phagocytosis function
Journal of Inflammation (2024)
-
Comparative proteomics reveals different protein expression in platelets in patients with alcoholic liver cirrhosis
Proteome Science (2024)
-
Development and validation of a nomogram for predicting mortality in patients with acute severe traumatic brain injury: A retrospective analysis
Neurological Sciences (2024)
-
Associations Between Leisure-Time Physical Activity Level and Peripheral Immune Cell Populations in the US General Population, Analysis of the National Health and Nutrition Examination Survey Data, 1999–2018
Sports Medicine - Open (2023)
-
The critical role of platelet in cancer progression and metastasis
European Journal of Medical Research (2023)