Key Points
-
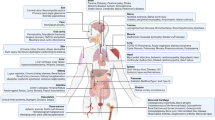
Bone-marrow-derived multipotent progenitor stromal cells — or mesenchymal stem cells (MSCs) — are the common predecessors of cells of the mesenchymal lineage, such as bone, cartilage and fat cells. They can also differentiate into cells from unrelated germline lineages (a process known as transdifferentiation).
-
Bone-marrow-derived stromal cells are the functional components of the haematopoietic stem cell (HSC) niche that support HSC homeostasis and they have anti-proliferative features that are in common with physiological stromal niches.
-
MSCs regulate many effector functions of innate immune cells, such as antigen presentation by dendritic cells (DCs), natural killer (NK)-cell cytotoxicity and the activation of neutrophils. However, activated NK cells can also kill MSCs.
-
MSCs inhibit T-cell proliferation through the induction of cell-division arrest, which is enhanced by interferon-γ released by activated T cells. MSCs also impair B-cell functions, such as antibody production.
-
After in vivo intravenous administration, MSCs can induce T-cell peripheral tolerance, home to inflamed tissues and exert a potent tissue-protective effect through the release of anti-inflammatory, anti-apoptotic and trophic molecules. In vivo studies indicate that the therapeutic effect of MSCs occurring through cell replacement is limited.
-
Clinical exploitation of in vitro-cultured MSCs has been safely carried out in some human conditions, which indicates that MSCs might be a new strategy for the treatment of immune-mediated diseases.
Abstract
Mesenchymal stem cells (MSCs) are a heterogeneous subset of stromal stem cells that can be isolated from many adult tissues. They can differentiate into cells of the mesodermal lineage, such as adipocytes, osteocytes and chondrocytes, as well as cells of other embryonic lineages. MSCs can interact with cells of both the innate and adaptive immune systems, leading to the modulation of several effector functions. After in vivo administration, MSCs induce peripheral tolerance and migrate to injured tissues, where they can inhibit the release of pro-inflammatory cytokines and promote the survival of damaged cells. This Review discusses the targets and mechanisms of MSC-mediated immunomodulation and the possible translation of MSCs to new therapeutic approaches.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Friedenstein, A. J., Chailakhyan, R. K., Latsinik, N. V., Panasyuk, A. F. & Keiliss-Borok, I. V. Stromal cells responsible for transferring the microenvironment of the hemopoietic tissues. Cloning in vitro and retransplantation in vivo. Transplantation 17, 331–340 (1974).
Pittenger, M. F. et al. Multilineage potential of adult human mesenchymal stem cells. Science 284, 143–147 (1999).
Kopen, G. C., Prockop, D. J. & Phinney, D. G. Marrow stromal cells migrate throughout forebrain and cerebellum, and they differentiate into astrocytes after injection into neonatal mouse brains. Proc. Natl Acad. Sci. USA 96, 10711–10716 (1999).
Caplan, A. I. Mesenchymal stem cells. J. Orthop. Res. 9, 641–650 (1991).
Sacchetti, B. et al. Self-renewing osteoprogenitors in bone marrow sinusoids can organize a hematopoietic microenvironment. Cell 131, 324–336 (2007). This study showed for the first time that only a limited subset of bone-marrow mesenchymal stromal cells in the sinusoidal niche can be considered stem cells in that they are self-renewing and can differentiate into all of the cells forming the haematopoietic microenvironment after transplantation.
Horwitz, E. et al. Clarification of the nomenclature for MSC: The International Society for Cellular Therapy position statement. Cytotherapy 7, 393–395 (2005).
Meirelles, L. da S., Chagastelles, P. C. & Nardi, N. B. Mesenchymal stem cells reside in virtually all post-natal organs and tissues. J. Cell Sci. 119, 2204–2213 (2006).
Muguruma, Y. et al. Reconstitution of the functional human hematopoietic microenvironment derived from human mesenchymal stem cells in the murine bone marrow compartment. Blood 107, 1878–1887 (2006).
Schofield, R. The relationship between the spleen colony-forming cell and the haemopoietic stem cell. Blood Cells 4, 7–25 (1978).
Wilson, A. & Trumpp, A. Bone-marrow haematopoietic-stem-cell niches. Nature Rev. Immunol. 6, 93–106 (2006).
Kiel, M. J. & Morrison, S. J. Uncertainty in the niches that maintain haematopoietic stem cells. Nature Rev. Immunol. 8, 290–301 (2008).
Bocelli-Tyndall, C. et al. Human articular chondrocytes suppress in vitro proliferation of anti-CD3 activated peripheral blood mononuclear cells. J. Cell Physiol. 209, 732–734 (2006).
Jones, S., Horwood, N., Cope, A. & Dazzi, F. The antiproliferative effect of mesenchymal stem cells is a fundamental property shared by all stromal cells. J. Immunol. 179, 2824–2831 (2007).
Haniffa, M. A. et al. Adult human fibroblasts are potent immunoregulatory cells and functionally equivalent to mesenchymal stem cells. J. Immunol. 179, 1595–1604 (2007).
Parsonage, G. et al. A stromal address code defined by fibroblasts. Trends Immunol. 26, 150–156 (2005).
Di Nicola, M. et al. Human bone marrow stromal cells suppress T-lymphocyte proliferation induced by cellular or nonspecific mitogenic stimuli. Blood 99, 3838–3843 (2002).
Bartholomew, A. et al. Mesenchymal stem cells suppress lymphocyte proliferation in vitro and prolong skin graft survival in vivo. Exp. Hematol. 30, 42–48 (2002).
Jiang, X. X. et al. Human mesenchymal stem cells inhibit differentiation and function of monocyte-derived dendritic cells. Blood 105, 4120–4126 (2005).
Nauta, A. J., Kruisselbrink, A. B., Lurvink, E., Willemze, R. & Fibbe, W. E. Mesenchymal stem cells inhibit generation and function of both CD34+-derived and monocyte-derived dendritic cells. J. Immunol. 177, 2080–2087 (2006). This study showed that MSCs inhibit the maturation and antigen-presentation functions of DCs from cord blood and monocytes through soluble factors, resulting in a tolerogenic DC phenotype.
Ramasamy, R. et al. Mesenchymal stem cells inhibit dendritic cell differentiation and function by preventing entry into the cell cycle. Transplantation 83, 71–76 (2007).
Li, Y. P. et al. Human mesenchymal stem cells license adult CD34+ hemopoietic progenitor cells to differentiate into regulatory dendritic cells through activation of the Notch pathway. J. Immunol. 180, 1598–1608 (2008).
Aggarwal, S. & Pittenger, M. F. Human mesenchymal stem cells modulate allogeneic immune cell responses. Blood 105, 1815–1822 (2005).
Beyth, S. et al. Human mesenchymal stem cells alter antigen-presenting cell maturation and induce T-cell unresponsiveness. Blood 105, 2214–2219 (2005).
Maccario, R. et al. Interaction of human mesenchymal stem cells with cells involved in alloantigen-specific immune response favors the differentiation of CD4+ T-cell subsets expressing a regulatory/suppressive phenotype. Haematologica 90, 516–525 (2005).
Moretta, A. Natural killer cells and dendritic cells: rendezvous in abused tissues. Nature Rev. Immunol. 2, 957–964 (2002).
Moretta, A. et al. Receptors for HLA class-I molecules in human natural killer cells. Annu. Rev. Immunol. 14, 619–648 (1996).
Moretta, A. et al. Activating receptors and coreceptors involved in human natural killer cell-mediated cytolysis. Annu. Rev. Immunol. 19, 197–223 (2001).
Spaggiari, G. M., Capobianco, A., Becchetti, S., Mingari, M. C. & Moretta, L. Mesenchymal stem cell–natural killer cell interactions: evidence that activated NK cells are capable of killing MSCs, whereas MSCs can inhibit IL-2-induced NK-cell proliferation. Blood 107, 1484–1490 (2006). This paper showed the mutual interaction between MSCs and NK cells. MSC-mediated inhibition of NK-cell cytotoxicity is related to the down-regulation of surface expression of NK-cell activating receptors. Conversely, cytokine-activated NK cells can kill MSCs efficiently in vitro.
Spaggiari, G. M. et al. Mesenchymal stem cells inhibit natural killer-cell proliferation, cytotoxicity, and cytokine production: role of indoleamine 2,3-dioxygenase and prostaglandin E2. Blood 111, 1327–1333 (2008).
Sotiropoulou, P. A., Perez, S. A., Gritzapis, A. D., Baxevanis, C. N. & Papamichail, M. Interactions between human mesenchymal stem cells and natural killer cells. Stem Cells 24, 74–85 (2006).
Poggi, A. et al. Interaction between human NK cells and bone marrow stromal cells induces NK cell triggering: role of NKp30 and NKG2D receptors. J. Immunol. 175, 6352–6360 (2005).
Selmani, Z. et al. Human leukocyte antigen-G5 secretion by human mesenchymal stem cells is required to suppress T lymphocyte and natural killer function and to induce CD4+CD25highFOXP3+ regulatory T cells. Stem Cells 26, 212–222 (2008).
Raffaghello, L. et al. Human mesenchymal stem cells inhibit neutrophil apoptosis: a model for neutrophil preservation in the bone marrow niche. Stem Cells 26, 151–162 (2008).
Fadeel, B., Ahlin, A., Henter, J. I., Orrenius, S. & Hampton, M. B. Involvement of caspases in neutrophil apoptosis: regulation by reactive oxygen species. Blood 92, 4808–4818 (1998).
Craddock, C. G. Jr, Perry, S., Ventzke, L. E. & Lawrence, J. S. Evaluation of marrow granulocytic reserves in normal and disease states. Blood 15, 840–855 (1960).
Tse, W. T., Pendleton, J. D., Beyer, W. M., Egalka, M. C. & Guinan, E. C. Suppression of allogeneic T-cell proliferation by human marrow stromal cells: implications in transplantation. Transplantation 75, 389–397 (2003).
Meisel, R. et al. Human bone marrow stromal cells inhibit allogeneic T-cell responses by indoleamine 2,3-dioxygenase mediated tryptophan degradation. Blood 103, 4619–4621 (2004).
Zappia, E. et al. Mesenchymal stem cells ameliorate experimental autoimmune encephalomyelitis inducing T-cell anergy. Blood 106, 1755–1761 (2005). This paper reported for the first time that intravenous injection of syngeneic MSCs in EAE-affected mice can be an effective treatment for autoimmune disease through the induction of peripheral T-cell tolerance.
Rasmusson, I., Ringden, O., Sundberg, B. & Le Blanc, K. Mesenchymal stem cells inhibit lymphocyte proliferation by mitogens and alloantigens by different mechanisms. Exp. Cell Res. 305, 33–41 (2005).
Krampera, M. et al. Bone marrow mesenchymal stem cells inhibit the response of naive and memory antigen-specific T cells to their cognate peptide. Blood 101, 3722–3729 (2003).
Glennie, S., Soeiro, I., Dyson, P. J., Lam, E. W. & Dazzi, F. Bone marrow mesenchymal stem cells induce division arrest anergy of activated T cells. Blood 105, 2821–2827 (2005). This study showed that MSC-mediated inhibition of cyclin D2 expression by stimulated T cells leads to cell-division arrest, an important mechanism that maintains the inhibition of T-cell proliferation.
Chabannes, D. et al. A role for heme oxygenase-1 in the immunosuppressive effect of adult rat and human mesenchymal stem cells. Blood 110, 3691–3694 (2007).
Ren, G. et al. Mesenchymal stem-cell-mediated immunosuppression occurs via concerted action of chemokines and nitric oxide. Cell Stem Cell 2, 141–150 (2008).
Sato, K. et al. Nitric oxide plays a critical role in suppression of T-cell proliferation by mesenchymal stem cells. Blood 109, 228–234 (2006).
Benvenuto, F. et al. Human mesenchymal stem cells promote survival of T cells in a quiescent state. Stem Cells 25, 1753–1760 (2007).
Rasmusson, I., Ringden, O., Sundberg, B. & Le Blanc, K. Mesenchymal stem cells inhibit the formation of cytotoxic T lymphocytes, but not activated cytotoxic T lymphocytes or natural killer cells. Transplantation 76, 1208–1213 (2003).
Rasmusson, I., Uhlin, M., Le Blanc, K. & Levitsky, V. Mesenchymal stem cells fail to trigger effector functions of cytotoxic T lymphocytes. J. Leukocyte Biol. 82, 887–893 (2007).
Morandi, F. et al. Immunogenicity of human mesenchymal stem cells in hla-class-i-restricted T-cell responses against viral or tumor-associated antigens. Stem Cells 26, 1275–1287 (2008).
Pevsner-Fischer, M. et al. Toll-like receptors and their ligands control mesenchymal stem cell functions. Blood 109, 1422–1432 (2007).
Tomchuck, S. L. et al. Toll-like receptors on human mesenchymal stem cells drive their migration and immunomodulating responses. Stem Cells 26, 99–107 (2008).
Hwa Cho, H., Bae, Y. C. & Jung, J. S. Role of Toll-like receptors on human adipose-derived stromal cells. Stem Cells 24, 2744–2752 (2006).
Liotta, F. et al. Toll-like receptors 3 and 4 are expressed by human bone marrow-derived mesenchymal stem cells and can inhibit their T-cell modulatory activity by impairing Notch signaling. Stem Cells 26, 279–289 (2008).
Svensson, M. & Kaye, P. M. Stromal-cell regulation of dendritic-cell differentiation and function. Trends Immunol. 27, 580–587 (2006).
Augello, A. et al. Bone marrow mesenchymal progenitor cells inhibit lymphocyte proliferation by activation of the programmed death 1 pathway. Eur. J. Immunol. 35, 1482–1490 (2005).
Krampera, M. et al. Role for interferon-γ in the immunomodulatory activity of human bone marrow mesenchymal stem cells. Stem Cells 24, 386–398 (2006). This paper gives important insights into the cross-talk between MSCs and immune cells, showing that MSC-mediated inhibition of T-cell and NK-cell functions is increased by IFN γ secreted by target cells, leading to the production of IDO by MSCs.
Corcione, A. et al. Human mesenchymal stem cells modulate B-cell functions. Blood 107, 367–372 (2006). In this paper, MSCs were shown to inhibit in vitro B-cell proliferation, differentiation to plasma cells and subsequent antibody secretion through paracrine mechanisms.
Traggiai, E. et al. Bone marrow-derived mesenchymal stem cells induce both polyclonal expansion and differentiation of b cells isolated from healthy donors and systemic lupus erythematosus patients. Stem Cells 26, 562–569 (2008).
Rasmusson, I., Le Blanc, K., Sundberg, B. & Ringden, O. Mesenchymal stem cells stimulate antibody secretion in human B cells. Scand. J. Immunol. 65, 336–343 (2007).
Gerdoni, E. et al. Mesenchymal stem cells effectively modulate pathogenic immune response in experimental autoimmune encephalomyelitis. Ann. Neurol. 61, 219–227 (2007).
Stagg, J., Pommey, S., Eliopoulos, N. & Galipeau, J. Interferon-γ-stimulated marrow stromal cells: a new type of nonhematopoietic antigen-presenting cell. Blood 107, 2570–2577 (2006).
Chan, J. L. et al. Antigen presenting property of mesenchymal stem cells occurs during a narrow window at low levels of interferon-γ. Blood 107, 4817–4824 (2006).
Feuerer, M. et al. Bone marrow as a priming site for T-cell responses to blood-borne antigen. Nature Med. 9, 1151–1157 (2003).
Ryan, J. M., Barry, F., Murphy, J. M. & Mahon, B. P. Interferon-γ does not break, but promotes the immunosuppressive capacity of adult human mesenchymal stem cells. Clin. Exp. Immunol. 149, 353–363 (2007).
Kogler, G. et al. Cytokine production and hematopoiesis supporting activity of cord blood-derived unrestricted somatic stem cells. Exp. Hematol. 33, 573–583 (2005).
Djouad, F. et al. Mesenchymal stem cells inhibit the differentiation of dendritic cells through an interleukin-6-dependent mechanism. Stem Cells 25, 2025–2032 (2007).
Almeida-Porada, G., Porada, C. D., Tran, N. & Zanjani, E. D. Cotransplantation of human stromal cell progenitors into preimmune fetal sheep results in early appearance of human donor cells in circulation and boosts cell levels in bone marrow at later time points after transplantation. Blood 95, 3620–3627 (2000).
Pereira, R. F. et al. Cultured adherent cells from marrow can serve as long-lasting precursor cells for bone, cartilage, and lung in irradiated mice. Proc. Natl Acad. Sci. USA 92, 4857–4861 (1995).
Orlic, D. et al. Bone marrow cells regenerate infarcted myocardium. Nature 410, 701–705 (2001).
Le Blanc, K. et al. Treatment of severe acute graft-versus-host disease with third party haploidentical mesenchymal stem cells. Lancet 363, 1439–1441 (2004). This paper reports the first successful use of infused MSCs to treat an immune-mediated human disease (GVHD).
Eliopoulos, N., Stagg, J., Lejeune, L., Pommey, S. & Galipeau, J. Allogeneic marrow stromal cells are immune rejected by MHC class I- and class II-mismatched recipient mice. Blood 106, 4057–4065 (2005).
Nauta, A. J. et al. Donor-derived mesenchymal stem cells are immunogenic in an allogeneic host and stimulate donor graft rejection in a non-myeloablative setting. Blood 108, 2114–2120 (2006).
Murphy, J. M. et al. Reduced chondrogenic and adipogenic activity of mesenchymal stem cells from patients with advanced osteoarthritis. Arthritis Rheum. 46, 704–713 (2002).
Papadaki, H. A. et al. Normal bone marrow hematopoietic stem cell reserves and normal stromal cell function support the use of autologous stem cell transplantation in patients with multiple sclerosis. Bone Marrow Transplant 36, 1053–1063 (2005).
Kastrinaki, M. C. et al. Functional, molecular and proteomic characterization of bone marrow mesenchymal stem cells in rheumatoid arthritis. Ann. Rheum. Dis. 67, 741–749 (2008).
Bacigalupo, A. et al. T-cell suppression mediated by mesenchymal stem cells is deficient in patients with severe aplastic anemia. Exp. Hematol. 33, 819–827 (2005).
Arnulf, B. et al. Phenotypic and functional characterization of bone marrow mesenchymal stem cells derived from patients with multiple myeloma. Leukemia 21, 158–163 (2007).
Devine, S. M., Cobbs, C., Jennings, M., Bartholomew, A. & Hoffman, R. Mesenchymal stem cells distribute to a wide range of tissues following systemic infusion into non-human primates. Blood 101, 2999–3001 (2003).
Li, Y. et al. Human marrow stromal cell therapy for stroke in rat: neurotrophins and functional recovery. Neurology 59, 514–523 (2002).
Ruster, B. et al. Mesenchymal stem cells display coordinated rolling and adhesion behavior on endothelial cells. Blood 108, 3938–3944 (2006).
Sordi, V. et al. Bone marrow mesenchymal stem cells express a restricted set of functionally active chemokine receptors capable of promoting migration to pancreatic islets. Blood 106, 419–427 (2005).
Son, B. R. et al. Migration of bone marrow and cord blood mesenchymal stem cells in vitro is regulated by stromal-derived factor-1–cxcr4 and hepatocyte growth factor–c-met axes and involves matrix metalloproteinases. Stem Cells 24, 1254–1264 (2006).
Uccelli, A., Pistoia, V. & Moretta, L. Mesenchymal stem cells: a new strategy for immunosuppression? Trends Immunol. 28, 219–226 (2007).
Munoz, J. R., Stoutenger, B. R., Robinson, A. P., Spees, J. L. & Prockop, D. J. Human stem/progenitor cells from bone marrow promote neurogenesis of endogenous neural stem cells in the hippocampus of mice. Proc. Natl Acad. Sci. USA 102, 18171–18176 (2005). MSCs implanted in the hippocampus of mice induced proliferation of endogenous neural stem cells expressing SOX2 and their migration and differentiation into both neural precursors and mature neural cells.
Rivera, F. J. et al. Mesenchymal stem cells instruct oligodendrogenic fate decision on adult neural stem cells. Stem Cells 24, 2209–2219 (2006).
Lee, R. H. et al. Multipotent stromal cells from human marrow home to and promote repair of pancreatic islets and renal glomeruli in diabetic NOD/scid mice. Proc. Natl Acad. Sci. USA 103, 17438–17443 (2006).
Urban, V. S. et al. Mesenchymal stem cells cooperate with bone marrow cells in therapy of diabetes. Stem Cells 26, 244–253 (2008).
Togel, F. et al. Administered mesenchymal stem cells protect against ischemic acute renal failure through differentiation-independent mechanisms. Am. J. Physiol. Renal Physiol. 289, F31–F42 (2005). In this paper, it is shown that MSCs rapidly home to the kidney following acute renal failure, where they release anti-inflammatory cytokines and protect renal cells from apoptosis without persistent engraftment or transdifferentiation.
Ortiz, L. A. et al. Interleukin 1 receptor antagonist mediates the antiinflammatory and antifibrotic effect of mesenchymal stem cells during lung injury. Proc. Natl Acad. Sci. USA 104, 11002–11007 (2007).
Mirotsou, M. et al. Secreted frizzled related protein 2 (Sfrp2) is the key Akt-mesenchymal stem cell-released paracrine factor mediating myocardial survival and repair. Proc. Natl Acad. Sci. USA 104, 1643–1648 (2007).
Ramasamy, R. et al. Mesenchymal stem cells inhibit proliferation and apoptosis of tumor cells: impact on in vivo tumor growth. Leukemia 21, 304–310 (2007).
Djouad, F. et al. Immunosuppressive effect of mesenchymal stem cells favors tumor growth in allogeneic animals. Blood 102, 3837–3844 (2003).
Ame-Thomas, P. et al. Human mesenchymal stem cells isolated from bone marrow and lymphoid organs support tumor B-cell growth: role of stromal cells in follicular lymphoma pathogenesis. Blood 109, 693–702 (2007).
Khakoo, A. Y. et al. Human mesenchymal stem cells exert potent antitumorigenic effects in a model of Kaposi's sarcoma. J. Exp. Med. 203, 1235–1247 (2006).
Karnoub, A. E. et al. Mesenchymal stem cells within tumour stroma promote breast cancer metastasis. Nature 449, 557–563 (2007).
Liotta, L. A. & Kohn, E. C. The microenvironment of the tumour–host interface. Nature 411, 375–379 (2001).
Horwitz, E. M. et al. Transplantability and therapeutic effects of bone marrow-derived mesenchymal cells in children with osteogenesis imperfecta. Nature Med. 5, 309–313 (1999). This paper reports the first use of MSCs for the treatment of a human disease based on their ability to differentiate to bone cells.
Koc, O. N. et al. Rapid hematopoietic recovery after coinfusion of autologous-blood stem cells and culture-expanded marrow mesenchymal stem cells in advanced breast cancer patients receiving high-dose chemotherapy. J. Clin. Oncol. 18, 307–316 (2000).
Ning, H. et al. The correlation between cotransplantation of mesenchymal stem cells and higher recurrence rate in hematologic malignancy patients: outcome of a pilot clinical study. Leukemia 22, 593–599 (2008).
Ball, L. M. et al. Cotransplantation of ex vivo expanded mesenchymal stem cells accelerates lymphocyte recovery and may reduce the risk of graft failure in haploidentical hematopoietic stem-cell transplantation. Blood 110, 2764–2767 (2007).
Okamoto, R. et al. Damaged epithelia regenerated by bone marrow-derived cells in the human gastrointestinal tract. Nature Med. 8, 1011–1017 (2002).
Prockop, D. J. & Olson, S. D. Clinical trials with adult stem/progenitor cells for tissue repair. Let's not overlook some essential precautions. Blood 109, 3147–3151 (2007).
Stier, S. et al. Osteopontin is a hematopoietic stem cell niche component that negatively regulates stem cell pool size. J. Exp. Med. 201, 1781–1791 (2005).
Arai, F. et al. Tie2/angiopoietin-1 signaling regulates hematopoietic stem cell quiescence in the bone marrow niche. Cell 118, 149–161 (2004).
Martino, G. & Pluchino, S. The therapeutic potential of neural stem cells. Nature Rev. Neurosci. 7, 395–406 (2006).
Augello, A., Tasso, R., Negrini, S. M., Cancedda, R. & Pennesi, G. Cell therapy using allogeneic bone marrow mesenchymal stem cells prevents tissue damage in collagen-induced arthritis. Arthritis Rheum. 56, 1175–1186 (2007).
Duncan, A. W. et al. Integration of Notch and Wnt signaling in hematopoietic stem cell maintenance. Nature Immunol. 6, 314–322 (2005).
Petersen, B. E. et al. Bone marrow as a potential source of hepatic oval cells. Science 284, 1168–1170 (1999).
Dennis, J. E. & Charbord, P. Origin and differentiation of human and murine stroma. Stem Cells 20, 205–214 (2002).
Takashima, Y. et al. Neuroepithelial cells supply an initial transient wave of MSC differentiation. Cell 129, 1377–1388 (2007).
Phinney, D. G. et al. Biological activities encoded by the murine mesenchymal stem cell transcriptome provide a basis for their developmental potential and broad therapeutic efficacy. Stem Cells 24, 186–198 (2006).
Pedemonte, E. et al. The molecular signature of therapeutic mesenchymal stem cells exposes the architecture of the hematopoietic stem cell niche synapse. BMC Genomics 8, 65 (2007).
Phinney, D. G. & Prockop, D. J. Mesenchymal stem/multi-potent stromal cells (mscs): the state of transdifferentiation and modes of tissue repair — current views. Stem Cells 25, 2896–2902 (2007).
Quarto, R. et al. Repair of large bone defects with the use of autologous bone marrow stromal cells. N. Engl. J. Med. 344, 385–386 (2001).
Rubio, D. et al. Spontaneous human adult stem cell transformation. Cancer Res. 65, 3035–3039 (2005).
Tolar, J. et al. Sarcoma derived from cultured mesenchymal stem cells. Stem Cells 25, 371–379 (2006).
Bernardo, M. E. et al. Human bone marrow derived mesenchymal stem cells do not undergo transformation after long-term in vitro culture and do not exhibit telomere maintenance mechanisms. Cancer Res. 67, 9142–9149 (2007).
Inoue, Y. et al. Subretinal transplantation of bone marrow mesenchymal stem cells delays retinal degeneration in the RCS rat model of retinal degeneration. Exp. Eye Res. 85, 234–241 (2007).
Gupta, N. et al. Intrapulmonary delivery of bone marrow-derived mesenchymal stem cells improves survival and attenuates endotoxin-induced acute lung injury in mice. J. Immunol. 179, 1855–1863 (2007).
Imberti, B. et al. Insulin-like growth factor-1 sustains stem cell mediated renal repair. J. Am. Soc. Nephrol. 18, 2921–2928 (2007).
Parekkadan, B. et al. Mesenchymal stem cell-derived molecules reverse fulminant hepatic failure. PLoS ONE 2, e941 (2007).
Acknowledgements
Some of the studies reported in this manuscript were supported by grants to A.U., L.M. and V.P. from the Fondazione Italiana Sclerosi Multipla (FISM), the Istituto Superiore di Sanità (ISS), the Italian Ministry of Health (Ricerca Finalizzata and Programma Integrato Ricerca Oncologica), the Italian Ministry of the University and Scientific Research (MIUR), the European Union FP6, the Fondazione Compagnia di San Paolo, Torino, Italy, the Fondazione CARIGE and the Fondazione CARIPLO.
Author information
Authors and Affiliations
Corresponding author
Glossary
- Stromal cells
-
Cells of non-lymphoid origin that form the framework of each organ. By expressing various molecules, these cells can support the adhesion, proliferation and survival of distinct cell subsets.
- Mesenchymal tissues
-
These are embryonic tissues of mesodermal origin, consisting of loosely packed, unspecialized cells set in a gelatinous ground substance, from which connective tissue, bone, cartilage and the circulatory and lymphatic systems develop.
- Transdifferentiation
-
The ability of a non-stem cell to transform into a different type of cell lineage, or when an already partially differentiated stem cell transforms into a different cell lineage or type.
- Stem cells
-
A subset of cells that has a self-renewing capacity and under appropriate conditions can give rise to several mature cell lineages.
- Mesodermal lineage
-
In animals with three tissue layers, the mesoderm is the middle layer of tissue, between the ectoderm and the endoderm. In vertebrates, the mesoderm forms the skeleton, muscles, heart, spleen and many other internal organs.
- HSC niche
-
The microenvironment inside the trabecular bone cavity, which is composed of a specialized population of cells that has an essential role in regulating the self-renewal and differentiation of haematopoietic stem cells (HSCs).
- Haematopoiesis
-
The commitment and differentiation processes that lead from a haematopoietic stem cell to the production of mature cells of all blood lineages — erythrocytes, myeloid cells (macrophages, mast cells, neutrophils and eosinophils), B and T cells, and natural killer cells.
- Sinusoids
-
Blood-filled spaces that lack the anatomy of a capillary. Sinusoids generally contain slow-flowing blood, which facilitates cellular interactions. Such vessels are found in the bone marrow and in the liver.
- Respiratory burst
-
A large increase in oxygen consumption and the generation of reactive oxygen species that accompanies the exposure of neutrophils to microorganisms and/or inflammatory mediators.
- Activation-induced cell death
-
A process by which activated, T-cell-receptor-restimulated T cells undergo cell death after engagement of cell-death receptors, such as CD95 or the tumour-necrosis factor receptor, or after exposure to reactive oxygen species.
- HLA-G
-
A non-classical MHC class Ib molecule that is involved in the establishment of immune tolerance at the maternal–fetal interface, the major soluble isoforms of which are HLA-G1 and HLA-G5.
- Notch signalling
-
A signalling system comprising highly conserved transmembrane receptors that regulate cell-fate choice in the development of many cell lineages, and so are crucial for the regulation of embryonic differentiation and development.
- Antibody-secreting cells
-
A term that denotes both proliferating plasmablasts and non-proliferating plasma cells. The term is used when both cell types might be present.
- Multiple sclerosis
-
A chronic inflammatory and demyelinating disease of the central nervous system. Multiple sclerosis involves an autoimmune response against components of myelin, which is thought to contribute to disease pathogenesis.
- T-cell anergy
-
A state of T-cell unresponsiveness to stimulation with antigen. It can be induced by stimulation with a large amount of specific antigen in the absence of the engagement of co-stimulatory molecules.
- WNT
-
WNT proteins are glycoproteins related to the Drosophila melanogaster protein Wingless, a ligand that regulates the temporal and spatial development of the embryo. WNT-mediated signalling has been shown to regulate cell-fate determination, proliferation, adhesion, migration and polarity during development. In addition to their crucial role in embryogenesis, WNT proteins and their downstream signalling molecules have been implicated in tumorigenesis and have causative roles in human colon cancers.
- Graft-versus-host disease
-
(GVHD). A disease that results from the immunological attack by donor allogeneic T cells that are transferred with the allograft (such as bone marrow, liver or gut) of target recipient organs or tissues (such as the skin or gut). GVHD occurs in graft recipients who cannot eliminate the host-reactive donor T cells, owing to immunosuppression, immunological immaturity or tolerance of the recipient.
- Histocompatibility antigens
-
Polymorphic peptides derived from normal cellular proteins that can be recognized in the context of MHC molecules. They get their name because they are responsible for the compatibility, or rather the lack of it, of the tissues of genetically different individuals. Immune responses specific for these polymorphic antigens can result in graft-versus-host reactions, graft rejection or beneficial anti-tumour responses.
- Graft-versus-leukaemia effect
-
An alloimmune attack against recipient haematopoietic neoplasms, which is mounted by donor immune cells in an allogeneic haematopoietic stem-cell transplant. With the exception of T-cell-depleted haploidentical allogeneic stem-cell transplants, wherein graft-versus-leukaemia effects can be mediated by alloreactive natural killer cells, the graft-versus-leukaemia response is mediated by αβ T cells in the donor allograft.
Rights and permissions
About this article
Cite this article
Uccelli, A., Moretta, L. & Pistoia, V. Mesenchymal stem cells in health and disease. Nat Rev Immunol 8, 726–736 (2008). https://doi.org/10.1038/nri2395
Issue Date:
DOI: https://doi.org/10.1038/nri2395
This article is cited by
-
Administration of adipose-derived stem cells extracellular vesicles in a murine model of spinal muscular atrophy: effects of a new potential therapeutic strategy
Stem Cell Research & Therapy (2024)
-
Mesenchymal stem cells promote ovarian reconstruction in mice
Stem Cell Research & Therapy (2024)
-
Human umbilical cord/placenta mesenchymal stem cell conditioned medium attenuates intestinal fibrosis in vivo and in vitro
Stem Cell Research & Therapy (2024)
-
LNP-RNA-engineered adipose stem cells for accelerated diabetic wound healing
Nature Communications (2024)
-
Epitranscriptomic modifications in mesenchymal stem cell differentiation: advances, mechanistic insights, and beyond
Cell Death & Differentiation (2024)