Key Points
-
The neonatal Fc receptor for IgG (FcRn) is responsible for the transfer of passive humoral immunity from the mother to the newborn in rodents and humans. Throughout life, FcRn contributes to effective humoral immunity by recycling IgG and extending its half-life in the circulation.
-
FcRn is expressed in various tissues in adults. The vascular endothelium is proposed to be the main site of IgG recycling, but myeloid-derived antigen-presenting cells also contribute significantly to extend the serum half-life of IgG.
-
FcRn is expressed in the epithelium of lungs and kidneys. IgG therapeutics delivered to the lungs can be absorbed via FcRn-mediated transcytosis.
-
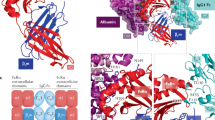
FcRn binds tightly to the Fc portion of IgG at acidic pH (pH 6.0) but not at physiological pH (pH 7.4). The hydrophobic interactions between FcRn and Fc are stabilized by salt-bridges that form between anionic residues on FcRn and titratable histidine residues of the IgG Fc region.
-
Mutagenesis of Fc-region residues at the FcRn–Fc interface can increase or decrease binding to FcRn. Improved pH-dependent binding to FcRn generally translates into increased half-life of IgG in the circulation.
-
The affinity of the FcRn–IgG interaction is highly dependent on the species and isotype of IgG. Consequently, transgenic mice expressing human FcRn transgenes are required for the initial evaluation of the pharmacokinetics of recombinant human IgG therapeutics before more focused testing in primate systems.
Abstract
The neonatal Fc receptor for IgG (FcRn) has been well characterized in the transfer of passive humoral immunity from a mother to her fetus. In addition, throughout life, FcRn protects IgG from degradation, thereby explaining the long half-life of this class of antibody in the serum. In recent years, it has become clear that FcRn is expressed in various sites in adults, where its potential function is now beginning to emerge. In addition, recent studies have examined the interaction between FcRn and the Fc portion of IgG with the aim of either improving the serum half-life of therapeutic monoclonal antibodies or reducing the half-life of pathogenic antibodies. This Review summarizes these two areas of FcRn biology.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Morphis, L. G. & Gitlin, D. Maturation of the maternofoetal transport system for human γ-globulin in the mouse. Nature 228, 573 (1970).
Brambell, F. W. The transmission of immunity from mother to young and the catabolism of immunoglobulins. Lancet 2, 1087–1093 (1966).
Erhlich, P. Über immunität durch vererbung und säugung. Z. Hyg. Infektionskr. 12, 183 (1892) (in German).
Simister, N. E. & Rees, A. R. Isolation and characterization of an Fc receptor from neonatal rat small intestine. Eur. J. Immunol. 15, 733–738 (1985).
Simister, N. E. & Mostov, K. E. An Fc receptor structurally related to MHC class I antigens. Nature 337, 184–187 (1989).
Jones, E. A. & Waldmann, T. A. The mechanism of intestinal uptake and transcellular transport of IgG in the neonatal rat. J. Clin. Invest. 51, 2916–2927 (1972).
Leach, J. L. et al. Isolation from human placenta of the IgG transporter, FcRn, and localization to the syncytiotrophoblast: implications for maternal–fetal antibody transport. J. Immunol. 157, 3317–3322 (1996).
Simister, N. E., Story, C. M., Chen, H. L. & Hunt, J. S. An IgG-transporting Fc receptor expressed in the syncytiotrophoblast of human placenta. Eur. J. Immunol. 26, 1527–1531 (1996).
Kristoffersen, E. K. Human placental Fc γ-binding proteins in the maternofetal transfer of IgG. APMIS Suppl. 64, 5–36 (1996).
Rodewald, R. pH-dependent binding of immunoglobulins to intestinal cells of the neonatal rat. J. Cell Biol. 71, 666–669 (1976).
Burmeister, W. P., Huber, A. H. & Bjorkman, P. J. Crystal structure of the complex of rat neonatal Fc receptor with Fc. Nature 372, 379–383 (1994).
Raghavan, M., Bonagura, V. R., Morrison, S. L. & Bjorkman, P. J. Analysis of the pH dependence of the neonatal Fc receptor/immunoglobulin G interaction using antibody and receptor variants. Biochemistry 34, 14649–14657 (1995).
Kim, J. K., Tsen, M. F., Ghetie, V. & Ward, E. S. Localization of the site of the murine IgG1 molecule that is involved in binding to the murine intestinal Fc receptor. Eur. J. Immunol. 24, 2429–2434 (1994).
Ober, R. J., Martinez, C., Vaccaro, C., Zhou, J. & Ward, E. S. Visualizing the site and dynamics of IgG salvage by the MHC class I-related receptor, FcRn. J. Immunol. 172, 2021–2029 (2004).
Ober, R. J., Martinez, C., Lai, X., Zhou, J. & Ward, E. S. Exocytosis of IgG as mediated by the receptor, FcRn: an analysis at the single-molecule level. Proc. Natl Acad. Sci. USA 101, 11076–11081 (2004).
Burmeister, W. P., Gastinel, L. N., Simister, N. E., Blum, M. L. & Bjorkman, P. J. Crystal structure at 2.2 Å resolution of the MHC-related neonatal Fc receptor. Nature 372, 336–343 (1994).
Ahouse, J. J. et al. Mouse MHC class I-like Fc receptor encoded outside the MHC. J. Immunol. 151, 6076–6088 (1993).
West, A. P. Jr., Herr, A. B. & Bjorkman, P. J. The chicken yolk sac IgY receptor, a functional equivalent of the mammalian MHC-related Fc receptor, is a phospholipase A2 receptor homolog. Immunity 20, 601–610 (2004).
Israel, E. J., Patel, V. K., Taylor, S. F., Marshak-Rothstein, A. & Simister, N. E. Requirement for a β2-microglobulin-associated Fc receptor for acquisition of maternal IgG by fetal and neonatal mice. J. Immunol. 154, 6246–6251 (1995).
Roopenian, D. C. et al. The MHC class I-like IgG receptor controls perinatal IgG transport, IgG homeostasis, and fate of IgG–Fc-coupled drugs. J. Immunol. 170, 3528–3533 (2003). A description of the FcRn-deficient mouse, which confirms the contribution of FcRn to the phenotypes of defective neonatal IgG transfer and shortened serum IgG half-life that were observed in β 2 m-deficient mice.
Junghans, R. P. & Anderson, C. L. The protection receptor for IgG catabolism is the β2-microglobulin-containing neonatal intestinal transport receptor. Proc. Natl Acad. Sci. USA 93, 5512–5516 (1996).
Ghetie, V. et al. Abnormally short serum half-lives of IgG in β2-microglobulin-deficient mice. Eur. J. Immunol. 26, 690–696 (1996).
Spriggs, M. K. et al. β2-microglobulin-, CD8+ T-cell-deficient mice survive inoculation with high doses of vaccinia virus and exhibit altered IgG responses. Proc. Natl Acad. Sci. USA 89, 6070–6074 (1992).
Chaudhury, C. et al. The major histocompatibility complex-related Fc receptor for IgG (FcRn) binds albumin and prolongs its lifespan. J. Exp. Med. 197, 315–322 (2003). The discovery that FcRn protects albumin.
Anderson, C. L. et al. Perspective—FcRn transports albumin: relevance to immunology and medicine. Trends Immunol. 27, 343–348 (2006).
Waldmann, T. A. & Strober, W. Metabolism of immunoglobulins. Prog. Allergy 13, 1–110 (1969).
Ward, E. S., Zhou, J., Ghetie, V. & Ober, R. J. Evidence to support the cellular mechanism involved in serum IgG homeostasis in humans. Int. Immunol. 15, 187–195 (2003). This study provides in vitro evidence that FcRn expressed in endothelial cells intercepts IgG and recycles it back to the circulation.
Borvak, J. et al. Functional expression of the MHC class I-related receptor, FcRn, in endothelial cells of mice. Int. Immunol. 10, 1289–1298 (1998).
Antohe, F., Radulescu, L., Gafencu, A., Ghetie, V. & Simionescu, M. Expression of functionally active FcRn and the differentiated bidirectional transport of IgG in human placental endothelial cells. Hum. Immunol. 62, 93–105 (2001).
Zhu, X. et al. MHC class I-related neonatal Fc receptor for IgG is functionally expressed in monocytes, intestinal macrophages, and dendritic cells. J. Immunol. 166, 3266–3276 (2001).
Bergtold, A., Desai, D. D., Gavhane, A. & Clynes, R. Cell surface recycling of internalized antigen permits dendritic cell priming of B cells. Immunity 23, 503–514 (2005).
Shah, U. et al. Distribution of the IgG Fc receptor, FcRn, in the human fetal intestine. Pediatr. Res. 53, 295–301 (2003).
Israel, E. J. et al. Expression of the neonatal Fc receptor, FcRn, on human intestinal epithelial cells. Immunology 92, 69–74 (1997).
Dickinson, B. L. et al. Bidirectional FcRn-dependent IgG transport in a polarized human intestinal epithelial cell line. J. Clin. Invest. 104, 903–911 (1999).
Yoshida, M. et al. IgG transport across mucosal barriers by neonatal Fc receptor for IgG and mucosal immunity. Springer Semin. Immunopathol. 28, 397–403 (2006).
Gill, R. K., Mahmood, S., Sodhi, C. P., Nagpaul, J. P. & Mahmood, A. IgG binding and expression of its receptor in rat intestine during postnatal development. Indian J. Biochem. Biophys. 36, 252–257 (1999).
Tiwari, B. & Junghans, R. P. Functional analysis of the mouse Fcgrt 5′ proximal promoter. Biochim. Biophys. Acta 1681, 88–98 (2005).
Mikulska, J. E. & Simister, N. E. Analysis of the promoter region of the human FcRn gene. Biochim. Biophys. Acta 1492, 180–184 (2000).
Yoshida, M. et al. Human neonatal Fc receptor mediates transport of IgG into luminal secretions for delivery of antigens to mucosal dendritic cells. Immunity 20, 769–783 (2004). This study provides evidence that FcRn expressed in the intestinal epithelium transports antigen-loaded immune complexes for uptake by mucosal DCs.
Yoshida, M. et al. Neonatal Fc receptor for IgG regulates mucosal immune responses to luminal bacteria. J. Clin. Invest. 116, 2142–2151 (2006).
Schlachetzki, F., Zhu, C. & Pardridge, W. M. Expression of the neonatal Fc receptor (FcRn) at the blood–brain barrier. J. Neurochem. 81, 203–206 (2002).
Ballabh, P., Braun, A. & Nedergaard, M. The blood–brain barrier: an overview: structure, regulation, and clinical implications. Neurobiol. Dis. 16, 1–13 (2004).
Zhang, Y. & Pardridge, W. M. Mediated efflux of IgG molecules from brain to blood across the blood–brain barrier. J. Neuroimmunol. 114, 168–172 (2001).
Deane, R. et al. IgG-assisted age-dependent clearance of Alzheimer's amyloid β peptide by the blood–brain barrier neonatal Fc receptor. J. Neurosci. 25, 11495–11503 (2005). This paper provides evidence that FcRn expressed at the blood−brain barrier exports IgG from the CNS and contributes to the efficacy of plaque-specific antibodies in the treatment of Alzheimer's disease.
Kacskovics, I. et al. FcRn mediates elongated serum half-life of human IgG in cattle. Int. Immunol. 18, 525–536 (2006).
Haymann, J. P. et al. Characterization and localization of the neonatal Fc receptor in adult human kidney. J. Am. Soc. Nephrol. 11, 632–639 (2000).
Kobayashi, N. et al. FcRn-mediated transcytosis of immunoglobulin G in human renal proximal tubular epithelial cells. Am. J. Physiol. Renal. Physiol. 282, F358–F365 (2002).
Mayer, B. et al. The neonatal Fc receptor (FcRn) is expressed in the bovine lung. Vet. Immunol. Immunopathol. 98, 85–89 (2004).
Spiekermann, G. M. et al. Receptor-mediated immunoglobulin G transport across mucosal barriers in adult life: functional expression of FcRn in the mammalian lung. J. Exp. Med. 196, 303–310 (2002).
Sakagami, M. et al. Expression and transport functionality of FcRn within rat alveolar epithelium: a study in primary cell culture and in the isolated perfused lung. Pharm. Res. 23, 270–279 (2006).
Kim, K. J. et al. Net absorption of IgG via FcRn-mediated transcytosis across rat alveolar epithelial cell monolayers. Am. J. Physiol. Lung Cell. Mol. Physiol. 287, L616–L622 (2004).
Bitonti, A. J. et al. Pulmonary delivery of an erythropoietin Fc fusion protein in non-human primates through an immunoglobulin transport pathway. Proc. Natl Acad. Sci. USA 101, 9763–9768 (2004).
Dumont, J. A. et al. Delivery of an erythropoietin–Fc fusion protein by inhalation in humans through an immunoglobulin transport pathway. J. Aerosol Med. 18, 294–303 (2005). This paper shows that FcRn expressed in the upper airway in humans can deliver pulmonary IgG therapeutics to the systemic circulation.
Low, S. C., Nunes, S. L., Bitonti, A. J. & Dumont, J. A. Oral and pulmonary delivery of FSH–Fc fusion proteins via neonatal Fc receptor-mediated transcytosis. Hum. Reprod. 20, 1805–1813 (2005).
Bitonti, A. J. & Dumont, J. A. Pulmonary administration of therapeutic proteins using an immunoglobulin transport pathway. Adv. Drug Deliv. Rev. 58, 1106–1118 (2006).
Dumont, J. A., Low, S. C., Peters, R. T. & Bitonti, A. J. Monomeric Fc fusions: impact on pharmacokinetic and biological activity of protein therapeutics. BioDrugs 20, 151–160 (2006).
Idusogie, E. E. et al. Mapping of the C1q binding site on rituxan, a chimeric antibody with a human IgG1 Fc. J. Immunol. 164, 4178–4184 (2000).
Martin, W. L., West, A. P. Jr., Gan, L. & Bjorkman, P. J. Crystal structure at 2.8 Å of an FcRn/heterodimeric Fc complex: mechanism of pH-dependent binding. Mol. Cell 7, 867–877 (2001). An excellent study detailing the molecular and structural interactions between FcRn and IgG-Fc.
Derrick, J. P. & Wigley, D. B. Crystal structure of a streptococcal protein G domain bound to an Fab fragment. Nature 359, 752–754 (1992).
Corper, A. L. et al. Structure of human IgM rheumatoid factor Fab bound to its autoantigen IgG Fc reveals a novel topology of antibody–antigen interaction. Nature Struct. Biol. 4, 374–381 (1997).
Deisenhofer, J. Crystallographic refinement and atomic models of a human Fc fragment and its complex with fragment B of protein A from Staphylococcus aureus at 2.9- and 2.8-Å resolution. Biochemistry 20, 2361–2370 (1981).
Vaughn, D. E. & Bjorkman, P. J. Structural basis of pH-dependent antibody binding by the neonatal Fc receptor. Structure 6, 63–73 (1998).
Medesan, C., Matesoi, D., Radu, C., Ghetie, V. & Ward, E. S. Delineation of the amino acid residues involved in transcytosis and catabolism of mouse IgG1. J. Immunol. 158, 2211–2217 (1997).
Kim, J. K. et al. Mapping the site on human IgG for binding of the MHC class I-related receptor, FcRn. Eur. J. Immunol. 29, 2819–2825 (1999).
Firan, M. et al. The MHC class I-related receptor, FcRn, plays an essential role in the maternofetal transfer of γ-globulin in humans. Int. Immunol. 13, 993–1002 (2001).
Shields, R. L. et al. High resolution mapping of the binding site on human IgG1 for FcγRI, FcγRII, FcγRIII, and FcRn and design of IgG1 variants with improved binding to the FcγR. J. Biol. Chem. 276, 6591–6604 (2001). A detailed study examining the effect of various point mutations on the FcRn–Fc interaction.
Petkova, S. B. et al. Enhanced half-life of genetically engineered human IgG1 antibodies in a humanized FcRn mouse model: potential application in humorally mediated autoimmune disease. Int. Immunol. 18, 1759–1769 (2006).
Vaughn, D. E. et al. Identification of critical IgG binding epitopes on the neonatal Fc receptor. J. Mol. Biol. 274, 597–607 (1997).
Ghetie, V. & Ward, E. S. Multiple roles for the major histocompatibility complex class I-related receptor FcRn. Annu. Rev. Immunol. 18, 739–766 (2000).
Martin, W. L. & Bjorkman, P. J. Characterization of the 2:1 complex between the class I MHC-related Fc receptor and its Fc ligand in solution. Biochemistry 38, 12639–12647 (1999).
Huber, A. H., Kelley, R. F., Gastinel, L. N. & Bjorkman, P. J. Crystallization and stoichiometry of binding of a complex between a rat intestinal Fc receptor and Fc. J. Mol. Biol. 230, 1077–1083 (1993).
Popov, S. et al. The stoichiometry and affinity of the interaction of murine Fc fragments with the MHC class I-related receptor, FcRn. Mol. Immunol. 33, 521–530 (1996).
Sánchez, L. M., Penny, D. M. & Bjorkman, P. J. Stoichiometry of the interaction between the major histocompatibility complex-related Fc receptor and its Fc ligand. Biochemistry 38, 9471–9476 (1999).
West, A. P. Jr. & Bjorkman, P. J. Crystal structure and immunoglobulin G binding properties of the human major histocompatibility complex-related Fc receptor. Biochemistry 39, 9698–9708 (2000).
Tesar, D. B., Tiangco, N. E. & Bjorkman, P. J. Ligand valency affects transcytosis, recycling and intracellular trafficking mediated by the neonatal Fc receptor. Traffic 7, 1127–1142 (2006).
Kim, J. K., Tsen, M. F., Ghetie, V. & Ward, E. S. Catabolism of the murine IgG1 molecule: evidence that both CH2–CH3 domain interfaces are required for persistence of IgG1 in the circulation of mice. Scand. J. Immunol. 40, 457–465 (1994).
Praetor, A., Jones, R. M., Wong, W. L. & Hunziker, W. Membrane-anchored human FcRn can oligomerize in the absence of IgG. J. Mol. Biol. 321, 277–284 (2002).
Hinton, P. R. et al. Engineered human IgG antibodies with longer serum half-lives in primates. J. Biol. Chem. 279, 6213–6216 (2004). The first study documenting improved pharmacokinetics in primates of human antibodies engineered to increase human FcRn binding.
Kamei, D. T. et al. Quantitative methods for developing Fc mutants with extended half-lives. Biotechnol. Bioeng. 92, 748–760 (2005).
Vaccaro, C., Zhou, J., Ober, R. J. & Ward, E. S. Engineering the Fc region of immunoglobulin G to modulate in vivo antibody levels. Nature Biotechnol. 23, 1283–1288 (2005).
Dall'Acqua, W. F., Kiener, P. A. & Wu, H. Properties of human IgG1s engineered for enhanced binding to the neonatal Fc receptor (FcRn). J. Biol. Chem. 281, 23514–23524 (2006).
Hinton, P. R. et al. An engineered human IgG1 antibody with longer serum half-life. J. Immunol. 176, 346–356 (2006).
Stern, M. & Herrmann, R. Overview of monoclonal antibodies in cancer therapy: present and promise. Crit. Rev. Oncol. Hematol. 54, 11–29 (2005).
Kenanova, V. et al. Tailoring the pharmacokinetics and positron emission tomography imaging properties of anti-carcinoembryonic antigen single-chain Fv-Fc antibody fragments. Cancer Res. 65, 622–631 (2005).
Dall'Acqua, W. F. et al. Increasing the affinity of a human IgG1 for the neonatal Fc receptor: biological consequences. J. Immunol. 169, 5171–5180 (2002).
Vaccaro, C., Bawdon, R., Wanjie, S., Ober, R. J. & Ward, E. S. Divergent activities of an engineered antibody in murine and human systems have implications for therapeutic antibodies. Proc. Natl Acad. Sci. USA 103, 18709–18714 (2006).
Brambell, F. W., Hemmings, W. A. & Morris, I. G. A Theoretical model of γ-globulin catabolism. Nature 203, 1352–1354 (1964). A seminal paper detailing the concentration–catabolism effect.
Humphrey, J. H. & Fahey, J. L. The metabolism of normal plasma proteins and γ-myeloma protein in mice bearing plasma-cell tumors. J. Clin. Invest. 40, 1696–1705 (1961).
Yu, Z. & Lennon, V. A. Mechanism of intravenous immune globulin therapy in antibody-mediated autoimmune diseases. N. Engl. J. Med. 340, 227–228 (1999).
Samuelsson, A., Towers, T. L. & Ravetch, J. V. Anti-inflammatory activity of IVIG mediated through the inhibitory Fc receptor. Science 291, 484–486 (2001).
Li, N. et al. Complete FcRn dependence for intravenous Ig therapy in autoimmune skin blistering diseases. J. Clin. Invest. 115, 3440–3450 (2005).
Akilesh, S. et al. The MHC class I-like Fc receptor promotes humorally mediated autoimmune disease. J. Clin. Invest. 113, 1328–1333 (2004). This study indicates that FcRn extends the serum half-life of pathogenic autoantibodies, thereby contributing to humoral autoimmune disease.
Hansen, R. J. & Balthasar, J. P. Intravenous immunoglobulin mediates an increase in anti-platelet antibody clearance via the FcRn receptor. Thromb. Haemost. 88, 898–899 (2002).
Raghavan, M., Chen, M. Y., Gastinel, L. N. & Bjorkman, P. J. Investigation of the interaction between the class I MHC-related Fc receptor and its immunoglobulin G ligand. Immunity 1, 303–315 (1994).
Getman, K. E. & Balthasar, J. P. Pharmacokinetic effects of 4C9, an anti-FcRn antibody, in rats: implications for the use of FcRn inhibitors for the treatment of humoral autoimmune and alloimmune conditions. J. Pharm. Sci. 94, 718–729 (2005).
Liu, L. et al. Amelioration of experimental autoimmune myasthenia gravis in rats by neonatal FcR blockade. J. Immunol. 178, 5390–5398 (2007).
DeLano, W. L., Ultsch, M. H., de Vos, A. M. & Wells, J. A. Convergent solutions to binding at a protein–protein interface. Science 287, 1279–1283 (2000).
Marino, M., Ruvo, M., De Falco, S. & Fassina, G. Prevention of systemic lupus erythematosus in MRL/lpr mice by administration of an immunoglobulin-binding peptide. Nature Biotechnol. 18, 735–739 (2000).
Ober, R. J., Radu, C. G., Ghetie, V. & Ward, E. S. Differences in promiscuity for antibody-FcRn interactions across species: implications for therapeutic antibodies. Int. Immunol. 13, 1551–1559 (2001).
Hansen, R. J. & Balthasar, J. P. Effects of intravenous immunoglobulin on platelet count and antiplatelet antibody disposition in a rat model of immune thrombocytopenia. Blood 100, 2087–2093 (2002).
Akilesh, S., Christianson, G. J., Roopenian, D. C. & Shaw, A. S. FcRn expression in bone marrow-derived cells functions to protect serum IgG from catabolism. J. Immunol. (in the press).
Acknowledgements
We thank the reviewers for their insightful comments and careful review. We apologize to authors whose work could not be cited due to space limitations.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The Jackson laboratory holds an issued patent on mice with a human FcRn transgene. Derry Roopenian receives royalties when these mice are licensed to pharmaceutical companies.
Related links
Glossary
- Syncytiotrophoblast
-
The outermost multinucleated syncytial cell layer of the trophoblast, which covers the chorionic villi. It is formed by fusion of the underlying layer of mononuclear trophoblast cells, and forms a barrier between the fetus and the mother.
- Transcytosis
-
The process of transport of material across a cell layer by uptake on one side of the cell into a coated vesicle. The vesicle might then be sorted through the trans-Golgi network and transported to the opposite side of the cell.
- Brush border
-
The surface layer of the normal small intestine that is comprised of small microvilli coated in a rich glycocalyx of mucus and other glycoproteins. The microvilli contain many of the digestive enzymes and transporter systems that are involved in the surface digestion and uptake of dietary materials. It provides a large surface area for absorption.
- MHC class I fold
-
The prototypic structure of MHC class I molecules and the related MHC class Ib molecules. The heavy chain polypeptide forms an α-helical sandwich that sits on top of an immunoglobulin domain. The heavy chain polypeptide pairs non-covalently with β2-microglobulin.
- Immune privilege
-
Immune-privileged sites are areas in the body with a decreased immune response to foreign antigens, including tissue grafts. These sites include the brain, eye, testis and placenta.
- Blood–brain barrier
-
A barrier formed by tight junctions between endothelial cells that markedly limits entry to the central nervous system by leukocytes and all large molecules, including to some extent immunoglobulins, cytokines and complement proteins.
- Amyloid plaques
-
Sites of amyloid-β accumulation and dystrophic neurites in the brains of mouse models and patients with Alzheimer's disease.
- Hinge region
-
A sequence of amino acids, which is often rich in cysteine and proline residues, that is present in the constant region of immunoglobulin heavy chains. It provides increased molecular flexibility. This region might be involved in the disulphide bonds that link adjacent immunoglobulin heavy chains.
- Staphylococcal protein A and streptococcal protein G
-
Proteins expressed on the cell surface of Staphylococcal or Streptococcal species. These proteins bind to the heavy chain of IgG antibodies from various species and therefore can be used in antibody isolation and purification.
- Rheumatoid factor
-
An antibody (usually IgM) that binds to the Fc region of IgG thereby forming immune complexes. Rheumatoid factors are sometimes found in patients with rheumatoid arthritis and other autoimmune diseases such as systemic lupus erythematosus.
- Humanized mice
-
Mice lacking certain genes of interest but transgenically expressing the human equivalent. Such mice provide an easy model system to study the biology of human genes.
Rights and permissions
About this article
Cite this article
Roopenian, D., Akilesh, S. FcRn: the neonatal Fc receptor comes of age. Nat Rev Immunol 7, 715–725 (2007). https://doi.org/10.1038/nri2155
Published:
Issue Date:
DOI: https://doi.org/10.1038/nri2155
This article is cited by
-
Engineered serum markers for non-invasive monitoring of gene expression in the brain
Nature Biotechnology (2024)
-
Antibody-drug conjugates in cancer therapy: innovations, challenges, and future directions
Archives of Pharmacal Research (2024)
-
Molecular Engineering of Interleukin-2 for Enhanced Therapeutic Activity in Autoimmune Diseases
BioDrugs (2024)
-
Efgartigimod in the treatment of Guillain–Barré syndrome
Journal of Neurology (2024)
-
Advances in antibody-based therapy in oncology
Nature Cancer (2023)