Abstract
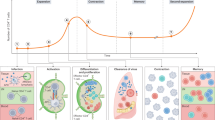
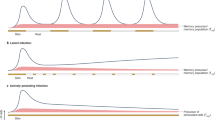
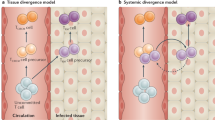
The immune system has evolved by continuously increasing its complexity to provide the host with an advantage over infectious agents. The development of immunological memory engenders long-lasting protection and lengthens the lifespan of the host. The generation of subsets of memory T cells with distinct homing and functional properties increases our defensive capabilities. However, the developmental relationship of memory T-cell subsets is a matter of debate. In this Opinion article, in light of recent developments, we suggest that it is probable that two distinct lineages comprise the memory CD8+ T-cell population generated in response to infection.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Reinach, T. Roi de Pont 283–285; 409–410 (Firmin-Didot, Paris, 1890) (in French).
Tripp, R. A., Hou, S. & Doherty, P. C. Temporal loss of the activated L-selectin-low phenotype for virus-specific CD8+ memory T cells. J. Immunol. 154, 5870–5875 (1995).
Hamann, D. et al. Phenotypic and functional separation of memory and effector human CD8+ T cells. J. Exp. Med. 186, 1407–1418 (1997).
Sallusto, F., Lenig, D., Forster, R., Lipp, M. & Lanzavecchia, A. Two subsets of memory T lymphocytes with distinct homing potentials and effector functions. Nature 401, 708–712 (1999).
Masopust, D., Vezys, V., Marzo, A. L. & Lefrançois, L. Preferential localization of effector memory cells in nonlymphoid tissue. Science 291, 2413–2417 (2001).
Reinhardt, R. L., Khoruts, A., Merica, R., Zell, T. & Jenkins, M. K. Visualizing the generation of memory CD4 T cells in the whole body. Nature 410, 101–105 (2001).
Unsoeld, H. & Pircher, H. Complex memory T-cell phenotypes revealed by coexpression of CD62L and CCR7. J. Virol. 79, 4510–4513 (2005).
Baars, P. A. et al. Properties of murine CD8+CD27− T cells. Eur. J. Immunol. 35, 3131–3141 (2005).
de Bree, G. J. et al. Selective accumulation of differentiated CD8+ T cells specific for respiratory viruses in the human lung. J. Exp. Med. 202, 1433–1442 (2005).
Masopust, D., Vezys, V., Wherry, E. J., Barber, D. L. & Ahmed, R. Cutting edge: gut microenvironment promotes differentiation of a unique memory CD8 T cell population. J. Immunol. 176, 2079–2083 (2006).
Fearon, D. T., Manders, P. & Wagner, S. D. Arrested differentiation, the self-renewing memory lymphocyte, and vaccination. Science 293, 248–250 (2001).
Wu, C. Y. et al. Distinct lineages of TH1 cells have differential capacities for memory cell generation in vivo. Nature Immunol. 3, 852–858 (2002).
Lefrançois, L., Marzo, A. & Williams, K. Sustained response initiation is required for T cell clonal expansion but not for effector or memory development in vivo. J. Immunol. 171, 2832–2839 (2003).
Seder, R. A. & Ahmed, R. Similarities and differences in CD4+ and CD8+ effector and memory T cell generation. Nature Immunol. 4, 835–842 (2003).
Lanzavecchia, A. & Sallusto, F. Progressive differentiation and selection of the fittest in the immune response. Nature Rev. Immunol. 2, 982–987 (2002).
Gett, A. V., Sallusto, F., Lanzavecchia, A. & Geginat, J. T cell fitness determined by signal strength. Nature Immunol. 4, 355–360 (2003).
Obar, J. J., Crist, S. G., Leung, E. K. & Usherwood, E. J. IL-15-independent proliferative renewal of memory CD8+ T cells in latent gammaherpesvirus infection. J. Immunol. 173, 2705–2714 (2004).
Wherry, E. J., Barber, D. L., Kaech, S. M., Blattman, J. N. & Ahmed, R. Antigen-independent memory CD8 T cells do not develop during chronic viral infection. Proc. Natl Acad. Sci. USA 101, 16004–16009 (2004).
Sierro, S., Rothkopf, R. & Klenerman, P. Evolution of diverse antiviral CD8+ T cell populations after murine cytomegalovirus infection. Eur. J. Immunol. 35, 1113–1123 (2005).
Clambey, E. T., van Dyk, L. F., Kappler, J. W. & Marrack, P. Non-malignant clonal expansions of CD8+ memory T cells in aged individuals. Immunol. Rev. 205, 170–189 (2005).
Wherry, E. J. et al. Lineage relationship and protective immunity of memory CD8 T cell subsets. Nature Immunol. 4, 225–234 (2003).
Marzo, A. L. et al. Initial T cell frequency dictates memory CD8+ T cell lineage commitment. Nature Immunol. 6, 793–799 (2005).
Bouneaud, C., Garcia, Z., Kourilsky, P. & Pannetier, C. Lineage relationships, homeostasis, and recall capacities of central- and effector-memory CD8 T cells in vivo. J. Exp. Med. 201, 579–590 (2005).
Bachmann, M. F., Wolint, P., Schwarz, K., Jager, P. & Oxenius, A. Functional properties and lineage relationship of CD8+ T cell subsets identified by expression of IL-7 receptor α and CD62L. J. Immunol. 175, 4686–4696 (2005).
Chao, C. C., Jensen, R. & Dailey, M. O. Mechanisms of L-selectin regulation by activated T cells. J. Immunol. 159, 1686–1694 (1997).
Galkina, E. et al. L-selectin shedding does not regulate constitutive T cell trafficking but controls the migration pathways of antigen-activated T lymphocytes. J. Exp. Med. 198, 1323–1335 (2003).
Smalley, D. M. & Ley, K. L-selectin: mechanisms and physiological significance of ectodomain cleavage. J. Cell. Mol. Med. 9, 255–266 (2005).
Blattman, J. N. et al. Estimating the precursor frequency of naive antigen-specific CD8 T cells. J. Exp. Med. 195, 657–664 (2002).
Baron, V. et al. The repertoires of circulating human CD8+ central and effector memory T cell subsets are largely distinct. Immunity 18, 193–204 (2003).
Chtanova, T. et al. Identification of T cell-restricted genes, and signatures for different T cell responses, using a comprehensive collection of microarray datasets. J. Immunol. 175, 7837–7847 (2005).
Williams, M. A. & Bevan, M. J. T cell memory: fixed or flexible? Nature Immunol. 6, 752–754 (2005).
Kaech, S. M. et al. Selective expression of the interleukin 7 receptor identifies effector CD8 T cells that give rise to long-lived memory cells. Nature Immunol. 4, 1191–1198 (2003).
Sprent, J. & Tough, D. F. T cell death and memory. Science 293, 245–248 (2001).
Miller, M. J., Hejazi, A. S., Wei, S. H., Cahalan, M. D. & Parker, I. T cell repertoire scanning is promoted by dynamic dendritic cell behavior and random T cell motility in the lymph node. Proc. Natl Acad. Sci. USA 101, 998–1003 (2004).
Mempel, T. R., Henrickson, S. E. & von Andrian, U. H. T-cell priming by dendritic cells in lymph nodes occurs in three distinct phases. Nature 427, 154–159 (2004).
Celli, S., Garcia, Z. & Bousso, P. CD4 T cells integrate signals delivered during successive DC encounters in vivo. J. Exp. Med. 202, 1271–1278 (2005).
Hogan, R. J. et al. Activated antigen-specific CD8+ T cells persist in the lungs following recovery from respiratory virus infections. J. Immunol. 166, 1813–1822 (2001).
Roberts, A. D., Ely, K. H. & Woodland, D. L. Differential contributions of central and effector memory T cells to recall responses. J. Exp. Med. 202, 123–133 (2005).
De Martinis, M. et al. CD50 and CD62L adhesion receptor expression on naive (CD45RA+) and memory (CD45RO+) T lymphocytes in the elderly. Pathobiology 68, 245–250 (2000).
Ginaldi, L. et al. Immunophenotypical changes of T lymphocytes in the elderly. Gerontology 46, 242–248 (2000).
Fagnoni, F. F. et al. Expansion of cytotoxic CD8+ CD28− T cells in healthy ageing people, including centenarians. Immunology 88, 501–507 (1996).
Saule, P. et al. Accumulation of memory T cells from childhood to old age: Central and effector memory cells in CD4+ versus effector memory and terminally differentiated memory cells in CD8+ compartment. Mech. Ageing Dev. 127, 274–281 (2006).
Hou, S., Hyland, L., Ryan, K. W., Portner, A. & Doherty, P. C. Virus specific CD8+ T-cell memory determined by clonal burst size. Nature 369, 652–654 (1994).
Selin, L. K., Vergilis, K., Welsh, R. M. & Nahill, S. R. Reduction of otherwise remarkably stable virus-specific cytotoxic T lymphocyte memory by heterologous viral infections. J. Exp. Med. 183, 2489–2499 (1996).
Asano, M. S. & Ahmed, R. CD8 T cell memory in B cell-deficient mice. J. Exp. Med. 183, 2165–2174 (1996).
Homann, D., Teyton, L. & Oldstone, M. B. Differential regulation of antiviral T-cell immunity results in stable CD8+ but declining CD4+ T-cell memory. Nature Med. 7, 913–919 (2001).
Schluns, K. S., Williams, K., Ma, A., Zheng, X. X. & Lefrançois, L. Requirement for IL-15 in the generation of primary and memory antigen-specific CD8 T cells. J. Immunol. 168, 4827–4831 (2002).
Klonowski, K. D. & Lefrancois, L. The CD8 memory T cell subsystem: integration of homeostatic signaling during migration. Semin. Immunol 17, 219–229 (2005).
Sallusto, F., Lenig, D., Mackay, C. R. & Lanzavecchia, A. Flexible programs of chemokine receptor expression on human polarized T helper 1 and 2 lymphocytes. J. Exp. Med. 187, 875–883 (1998).
Mackay, C. R. et al. Tissue-specific migration pathways by phenotypically distinct populations of memory T cells. Eur. J. Immunol. 22, 887–895 (1992).
Bromley, S. K., Thomas, S. Y. & Luster, A. D. Chemokine receptor CCR7 guides T cell exit from peripheral tissues and entry into afferent lymphatics. Nature Immunol. 6, 895–901 (2005).
Debes, G. F. et al. Chemokine receptor CCR7 required for T lymphocyte exit from peripheral tissues. Nature Immunol. 6, 889–894 (2005).
Lefrançois, L. et al. The role of β7 integrins in CD8 T cell trafficking during an anti-viral immune response. J. Exp. Med. 189, 1631–1638 (1999).
Klonowski, K. D. et al. Dynamics of blood-borne CD8 memory T cell migration in vivo. Immunity 20, 551–562 (2004).
Zammit, D. J., Cauley, L. S., Pham, Q.-M. & Lefrançois, L. Dendritic cells maximize the memory CD8 T cell response to infection. Immunity 22, 561–570 (2005).
Kaech, S. M., Hemby, S., Kersh, E. & Ahmed, R. Molecular and functional profiling of memory CD8 T cell differentiation. Cell 111, 837–851 (2002).
Zaph, C., Uzonna, J., Beverley, S. M. & Scott, P. Central memory T cells mediate long-term immunity to Leishmania major in the absence of persistent parasites. Nature Med. 10, 1104–1110 (2004).
Hogan, R. J. et al. Protection from respiratory virus infections can be mediated by antigen-specific CD4+ T cells that persist in the lungs. J. Exp. Med. 193, 981–986 (2001).
Roberts, A. D. & Woodland, D. L. Effector memory CD8+ T cells play a prominent role in recall responses to secondary viral infection in the lung. J. Immunol. 172, 6533–6537 (2004).
Acknowledgements
We would like to thank L. Puddington for critical review of the manuscript and all the members of the laboratory for intellectual and scientific contributions that led to the theories put forth in this paper. This work was supported by grants from the US National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Rights and permissions
About this article
Cite this article
Lefrançois, L., Marzo, A. The descent of memory T-cell subsets. Nat Rev Immunol 6, 618–623 (2006). https://doi.org/10.1038/nri1866
Issue Date:
DOI: https://doi.org/10.1038/nri1866
This article is cited by
-
Themis2 regulates natural killer cell memory function and formation
Nature Communications (2023)
-
Lymphatic endothelial cells prime naïve CD8+ T cells into memory cells under steady-state conditions
Nature Communications (2020)
-
Relevance of laboratory testing for the diagnosis of primary immunodeficiencies: a review of case-based examples of selected immunodeficiencies
Clinical and Molecular Allergy (2011)
-
Predicting CD62L expression during the CD8+ T‐cell response in vivo
Immunology & Cell Biology (2010)
-
Gene-modified tumor vaccine secreting a designer cytokine Hyper-Interleukin-6 is an effective therapy in mice bearing orthotopic renal cell cancer
Cancer Gene Therapy (2010)