Key Points
-
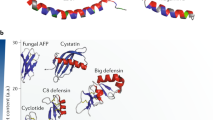
What are defensins? What are the structural characteristics of the three vertebrate defensin subclasses?
-
Which tissues and cells contain abundant defensins?
-
How are defensins synthesized and processed?
-
How is the synthesis and release of defensins regulated?
-
What are the effects of defensins on biological membranes and on microorganisms?
-
How do bacteria respond to limit the damage by defensins?
-
What are the biological functions of defensins in innate immunity?
Abstract
The production of natural antibiotic peptides has emerged as an important mechanism of innate immunity in plants and animals. Defensins are diverse members of a large family of antimicrobial peptides, contributing to the antimicrobial action of granulocytes, mucosal host defence in the small intestine and epithelial host defence in the skin and elsewhere. This review, inspired by a spate of recent studies of defensins in human diseases and animal models, focuses on the biological function of defensins.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Ganz, T. et al. Defensins. Natural peptide antibiotics of human neutrophils. J. Clin. Invest. 76, 1427–1435 (1985).
Selsted, M. E., Harwig, S. S., Ganz, T., Schilling, J. W. & Lehrer, R. I. Primary structures of three human neutrophil defensins. J. Clin. Invest. 76, 1436–1439 (1985). These two papers identified human defensins as members of a new antimicrobial peptide family.
Zanetti, M., Gennaro, R. & Romeo, D. Cathelicidins: a novel protein family with a common proregion and a variable C-terminal antimicrobial domain. FEBS Lett. 374, 1–5 (1995). A key conceptual review that proposed cathelicidins as the second main antimicrobial peptide family in mammals.
Lehrer, R. I. & Ganz, T. Cathelicidins: a family of endogenous antimicrobial peptides. Curr. Opin. Hematol. 9, 18–22 (2002).
Tsai, H. & Bobek, L. A. Human salivary histatins: promising anti-fungal therapeutic agents. Crit. Rev. Oral Biol. Med. 9, 480–497 (1998).
Schittek, B. et al. Dermcidin: a novel human antibiotic peptide secreted by sweat glands. Nature Immunol. 2, 1133–1137 (2001).
Brogden, K. A., Ackermann, M. & Huttner, K. M. Small, anionic, and charge-neutralizing propeptide fragments of zymogens are antimicrobial. Antimicrob. Agents Chemother. 41, 1615–1617 (1997).
Zeya, H. I. & Spitznagel, J. K. Antibacterial and enzymic basic proteins from leukocyte lysosomes: separation and identification. Science 142, 1085–1087 (1963).
Lehrer, R. I., Selsted, M. E., Szklarek, D. & Fleischmann, J. Antibacterial activity of microbicidal cationic proteins 1 and 2, natural peptide antibiotics of rabbit lung macrophages. Infect. Immun. 42, 10–14 (1983).
Selsted, M. E., Brown, D. M., DeLange, R. J. & Lehrer, R. I. Primary structures of MCP-1 and MCP-2, natural peptide antibiotics of rabbit lung macrophages. J. Biol. Chem. 258, 14485–14489 (1983).
Selsted, M. E., Szklarek, D. & Lehrer, R. I. Purification and antibacterial activity of antimicrobial peptides of rabbit granulocytes. Infect. Immun. 45, 150–154 (1984).
Ouellette, A. J. et al. Developmental regulation of cryptdin, a corticostatin/defensin precursor mRNA in mouse small intestinal crypt epithelium. J. Cell Biol. 108, 1687–1695 (1989). Defensin messenger RNA is abundantly expressed by the Paneth cells of mouse small intestine.
Diamond, G. et al. Tracheal antimicrobial peptide, a cysteine-rich peptide from mammalian tracheal mucosa: peptide isolation and cloning of a cDNA. Proc. Natl Acad. Sci. USA 88, 3952–3956 (1991). Another abundant defensin, the first member of the β-defensin subfamily, was isolated from cow tracheal epithelium.
Hill, C. P., Yee, J., Selsted, M. E. & Eisenberg, D. Crystal structure of defensin HNP-3, an amphiphilic dimer: mechanisms of membrane permeabilization. Science 251, 1481–1485 (1991). This is a key crystallographic study that defined the first defensin structure.
Pardi, A. et al. Solution structures of the rabbit neutrophil defensin NP-5. J. Mol. Biol. 201, 625–636 (1988).
Pardi, A., Zhang, X. L., Selsted, M. E., Skalicky, J. J. & Yip, P. F. NMR studies of defensin antimicrobial peptides. 2. Three-dimensional structures of rabbit NP-2 and human HNP-1. Biochemistry 31, 11357–11364 (1992).
Zhang, X. L., Selsted, M. E. & Pardi, A. NMR studies of defensin antimicrobial peptides. 1. Resonance assignment and secondary structure determination of rabbit NP-2 and human HNP-1. Biochemistry 31, 11348–11356 (1992).
Skalicky, J. J., Selsted, M. E. & Pardi, A. Structure and dynamics of the neutrophil defensins NP-2, NP-5, and HNP-1: NMR studies of amide hydrogen exchange kinetics. Proteins 20, 52–67 (1994).
Zimmermann, G. R., Legault, P., Selsted, M. E. & Pardi, A. Solution structure of bovine neutrophil β-defensin-12: the peptide fold of the β-defensins is identical to that of the classical defensins. Biochemistry 34, 13663–13671 (1995).
Hoover, D. M. et al. The structure of human β-defensin-2 shows evidence of higher-order oligomerization. J. Biol. Chem. 275, 32911–32918 (2000).
Sawai, M. V. et al. The NMR structure of human β-defensin-2 reveals a novel α-helical segment. Biochemistry 40, 3810–3816 (2001).
Tang, Y. Q. et al. A cyclic antimicrobial peptide produced in primate leukocytes by the ligation of two truncated α-defensins. Science 286, 498–502 (1999). The discovery of a circular defensin spliced together from two precursors by a new posttranslational process.
Cole, A. M. et al. Retrocyclin: a primate peptide that protects cells from infection by T- and M-tropic strains of HIV-1. Proc. Natl Acad. Sci. USA 99, 1813–1818 (2002).
Liu, L., Zhao, C., Heng, H. H. Q. & Ganz, T. The human β-defensin-1 and α-defensins are encoded by adjacent genes: two peptide families with differing disulfide topology share a common ancestry. Genomics 43, 316–320 (1997). Despite conjectures to the contrary, α- and β-defensins did not evolve independently, but evolved from a common ancestral molecule.
Harwig, S. S. et al. Gallinacins: cysteine-rich antimicrobial peptides of chicken leukocytes. FEBS Lett. 342, 281–285 (1994).
Zhao, C. et al. Gallinacin-3, an inducible epithelial β-defensin in the chicken. Infect. Immun. 69, 2684–2691 (2001).
Evans, E. W., Beach, G. G., Wunderlich, J. & Harmon, B. G. Isolation of antimicrobial peptides from avian heterophils. J. Leukoc. Biol. 56, 661–665 (1994).
Brockus, C. W., Jackwood, M. W. & Harmon, B. G. Characterization of β-defensin prepropeptide mRNA from chicken and turkey bone marrow. Anim. Genet. 29, 283–289 (1998).
Marquardt, H., Todaro, G. J. & Twardzik, D. R. Snake venom growth arresting peptide. US Patent 4774318-A 3 (1988).
Ganz, T. Extracellular release of antimicrobial defensins by human polymorphonuclear leukocytes. Infect. Immun. 55, 568–571 (1987).
Ayabe, T. et al. Secretion of microbicidal α-defensins by intestinal Paneth cells in response to bacteria. Nature Immunol. 1, 113–118 (2000).
Valore, E. V. et al. Human β-defensin-1: an antimicrobial peptide of urogenital tissues. J. Clin. Invest. 101, 1633–1642 (1998).
Harder, J., Bartels, J., Christophers, E. & Schroeder, J. -M. A peptide antibiotic from human skin. Nature 387, 861–862 (1997).
Shi, J. et al. Porcine epithelial β-defensin 1 is expressed in the dorsal tongue at antimicrobial concentrations. Infect. Immun. 67, 3121–3127 (1999).
Eisenhauer, P. B. & Lehrer, R. I. Mouse neutrophils lack defensins. Infect. Immun. 60, 3446–3447 (1992). An important study that raised doubts about the use of mice as a faithful model of human innate immunity.
Eisenhauer, P. B. et al. Purification and antimicrobial properties of three defensins from rat neutrophils. Infect. Immun. 57, 2021–2027 (1989).
Kokryakov, V. N. et al. Protegrins: leukocyte antimicrobial peptides that combine features of corticostatic defensins and tachyplesins. FEBS Lett. 327, 231–236 (1993).
Yount, N. Y. et al. Cloning and expression of bovine neutrophil β-defensins. Biosynthetic profile during neutrophilic maturation and localization of mature peptide to novel cytoplasmic dense granules. J. Biol. Chem. 274, 26249–26258 (1999).
Schonwetter, B. S., Stolzenberg, E. D. & Zasloff, M. A. Epithelial antibiotics induced at sites of inflammation. Science 267, 1645–1648 (1995).
Tarver, A. P. et al. Enteric β-defensin: molecular cloning and characterization of a gene with inducible intestinal epithelial cell expression associated with Cryptosporidium parvum infection. Infect. Immun. 66, 1045–1056 (1998).
Ganz, T. et al. The structure of the rabbit macrophage defensin genes and their organ-specific expression. J. Immunol. 143, 1358–1365 (1989).
Ryan, L. K., Rhodes, J., Bhat, M. & Diamond, G. Expression of β-defensin genes in bovine alveolar macrophages. Infect. Immun. 66, 878–881 (1998).
Duits, L. A. et al. Inhibition of hBD-3, but not hBD-1 and hBD-2, mRNA expression by corticosteroids. Biochem. Biophys. Res. Commun. 280, 522–525 (2001).
Agerberth, B. et al. The human antimicrobial and chemotactic peptides LL-37 and α-defensins are expressed by specific lymphocyte and monocyte populations. Blood 96, 3086–3093 (2000).
Sparkes, R. S. et al. Assignment of defensin gene(s) to human chromosome 8p23. Genomics 5, 240–244 (1989).
Harder, J. et al. Mapping of the gene encoding human β-defensin-2 (DEFB2) to chromosome region 8p22-p23.1. Genomics 46, 472–475 (1997).
Liu, L. et al. Structure and mapping of the human β-defensin HBD-2 gene and its expression at sites of inflammation. Gene 222, 237–244 (1998).
Linzmeier, R., Ho, C. H., Hoang, B. V. & Ganz, T. A 450-kb contig of defensin genes on human chromosome 8p23. Gene 233, 205–211 (1999).
Mars, W. M. et al. Inheritance of unequal numbers of the genes encoding the human neutrophil defensins HP-1 and HP-3. J. Biol. Chem. 270, 30371–30376 (1995).
Schutte, B. C. et al. Discovery of five conserved β-defensin gene clusters using a computational search strategy. Proc. Natl Acad. Sci. USA 99, 2129–2133 (2002). There are many more members of the defensin gene family than has previously been suspected. What do all these defensins do?
Parmley, R. T., Doran, T., Boyd, R. L. & Gilbert, C. Unmasking and redistribution of lysosomal sulfated glycoconjugates in phagocytic polymorphonuclear leukocytes. J. Histochem. Cytochem. 34, 1701–1707 (1986).
Spicer, S. S., Staley, M. W., Wetzel, M. G. & Wetzel, B. K. Acid mucosubstance and basic protein in mouse Paneth cells. J. Histochem. Cytochem. 15, 225–242 (1967).
Kostoulas, G., Horler, D., Naggi, A., Casu, B. & Baici, A. Electrostatic interactions between human leukocyte elastase and sulfated glycosaminoglycans: physiological implications. Biol. Chem. 378, 1481–1489 (1997).
Fromm, J. R., Hileman, R. E., Caldwell, E. E., Weiler, J. M. & Linhardt, R. J. Differences in the interaction of heparin with arginine and lysine and the importance of these basic amino acids in the binding of heparin to acidic fibroblast growth factor. Arch. Biochem. Biophys. 323, 279–287 (1995).
Hileman, R. E., Fromm, J. R., Weiler, J. M. & Linhardt, R. J. Glycosaminoglycan–protein interactions: definition of consensus sites in glycosaminoglycan binding proteins. Bioessays 20, 156–167 (1998).
Daher, K. A., Lehrer, R. I., Ganz, T. & Kronenberg, M. Isolation and characterization of human defensin cDNA clones. Proc. Natl Acad. Sci. USA 85, 7327–7331 (1988).
Valore, E. V. & Ganz, T. Posttranslational processing of defensins in immature human myeloid cells. Blood 79, 1538–1544 (1992).
Michaelson, D., Rayner, J., Couto, M. & Ganz, T. Cationic defensins arise from charge-neutralized propeptides: a mechanism for avoiding leukocyte autocytotoxicity? J. Leukoc. Biol. 51, 634–639 (1992).
Valore, E. V., Martin, E., Harwig, S. S. & Ganz, T. Intramolecular inhibition of human defensin HNP-1 by its propiece. J. Clin. Invest. 97, 1624–1629 (1996).
Liu, L. & Ganz, T. The pro region of human neutrophil defensin contains a motif that is essential for normal subcellular sorting. Blood 85, 1095–1103 (1995).
Yount, N. Y. et al. Rat neutrophil defensins. Precursor structures and expression during neutrophilic myelopoiesis. J. Immunol. 155, 4476–4484 (1995).
Arnljots, K., Sorensen, O., Lollike, K. & Borregaard, N. Timing, targeting and sorting of azurophil granule proteins in human myeloid cells. Leukemia 12, 1789–1795 (1998).
Cowland, J. B. & Borregaard, N. The individual regulation of granule protein mRNA levels during neutrophil maturation explains the heterogeneity of neutrophil granules. J. Leukoc. Biol. 66, 989–995 (1999).
Harwig, S. S., Park, A. S. & Lehrer, R. I. Characterization of defensin precursors in mature human neutrophils. Blood 79, 1532–1537 (1992).
Ghosh, D. et al. Paneth cell trypsin is the processing enzyme for human defensin-5. Nature Immunol. 3, 583–590 (2002). The processing of human defensin 5 (HD5) by trypsin after release from Paneth cells granules is different from the pre-storage processing of mouse intestinal defensins.
Oren, A., Ganz, T., Liu, L. & Meerloo, T. In human epidermis, β-defensin 2 is packaged in lamellar bodies. Exp. Mol. Pathol. 74, 180–182 (2003).
Rice, W. G. et al. Defensin-rich dense granules of human neutrophils. Blood 70, 757–765 (1987).
Joiner, K. A., Ganz, T., Albert, J. & Rotrosen, D. The opsonizing ligand on Salmonella typhimurium influences incorporation of specific, but not azurophil, granule constituents into neutrophil phagosomes. J. Cell Biol. 109, 2771–2782 (1989).
Mallow, E. B. et al. Human enteric defensins. Gene structure and developmental expression. J. Biol. Chem. 271, 4038–4045 (1996).
Ma, Y., Su, Q. & Tempst, P. Differentiation-stimulated activity binds an ETS-like, essential regulatory element in the human promyelocytic defensin-1 promoter. J. Biol. Chem. 273, 8727–8740 (1998).
Liu, L., Roberts, A. A. & Ganz, T. By IL-1 signaling, monocyte-derived cells dramatically enhance the epidermal antimicrobial response to lipopolysaccharide. J. Immunol. 170, 575–580 (2003). The induction of defensin synthesis in the infected or inflamed epidermis depends on the intermediate production of interleukin-1 by myeloid cells.
Tsutsumi-Ishii, Y. & Nagaoka, I. Modulation of human β-defensin-2 transcription in pulmonary epithelial cells by lipopolysaccharide-stimulated mononuclear phagocytes via proinflammatory cytokine production. J. Immunol. 170, 4226–4236 (2003).
Sorensen, O. E. et al. Wound healing and expression of antimicrobial peptides/polypeptides in human keratinocytes, a consequence of common growth factors. J. Immunol. 170, 5583–5589 (2003).
Garcia, J. R. et al. Human β-defensin 4: a novel inducible peptide with a specific salt-sensitive spectrum of antimicrobial activity. FASEB J. 15, 1819–1821 (2001).
Diamond, G., Kaiser, V., Rhodes, J., Russell, J. P. & Bevins, C. L. Transcriptional regulation of β-defensin gene expression in tracheal epithelial cells. Infect. Immun. 68, 113–119 (2000).
Stolzenberg, E. D., Anderson, G. M., Ackermann, M. R., Whitlock, R. H. & Zasloff, M. Epithelial antibiotic induced in states of disease. Proc. Natl Acad. Sci. USA 94, 8686–8690 (1997).
Ganz, T., Sherman, M. P., Selsted, M. E. & Lehrer, R. I. Newborn rabbit alveolar macrophages are deficient in two microbicidal cationic peptides, MCP-1 and MCP-2. Am. Rev. Respir. Dis. 132, 901–904 (1985).
Selsted, M. E., Szklarek, D., Ganz, T. & Lehrer, R. I. Activity of rabbit leukocyte peptides against Candida albicans. Infect. Immun. 49, 202–206 (1985).
Lehrer, R. I., Ganz, T., Szklarek, D. & Selsted, M. E. Modulation of the in vitro candidacidal activity of human neutrophil defensins by target cell metabolism and divalent cations. J. Clin. Invest. 81, 1829–1835 (1988).
Lehrer, R. I., Daher, K., Ganz, T. & Selsted, M. E. Direct inactivation of viruses by MCP-1 and MCP-2, natural peptide antibiotics from rabbit leukocytes. J. Virol. 54, 467–472 (1985).
Daher, K. A., Selsted, M. E. & Lehrer, R. I. Direct inactivation of viruses by human granulocyte defensins. J. Virol. 60, 1068–1074 (1986).
Zhang, L. et al. Contribution of human α-defensin 1, 2, and 3 to the anti-HIV-1 activity of CD8 antiviral factor. Science 298, 995–1000 (2002). References 80–82 show that some defensins have interesting antiviral activity.
Lichtenstein, A., Ganz, T., Selsted, M. E. & Lehrer, R. I. In vitro tumor cell cytolysis mediated by peptide defensins of human and rabbit granulocytes. Blood 68, 1407–1410 (1986).
Lichtenstein, A. K., Ganz, T., Nguyen, T. M., Selsted, M. E. & Lehrer, R. I. Mechanism of target cytolysis by peptide defensins. Target cell metabolic activities, possibly involving endocytosis, are crucial for expression of cytotoxicity. J. Immunol. 140, 2686–2694 (1988).
Lichtenstein, A. Mechanism of mammalian cell lysis mediated by peptide defensins. Evidence for an initial alteration of the plasma membrane. J. Clin. Invest. 88, 93–100 (1991).
Van Wetering, S. et al. Effect of defensins on interleukin-8 synthesis in airway epithelial cells. Am. J. Physiol. 272, L888–L896 (1997).
Zhang, H. et al. Neutrophil defensins mediate acute inflammatory response and lung dysfunction in dose-related fashion. Am. J. Physiol. Lung Cell. Mol. Physiol. 280, L947–L954 (2001).
Lehrer, R. I. et al. Interaction of human defensins with Escherichia coli. Mechanism of bactericidal activity. J. Clin. Invest. 84, 553–561 (1989). An important paper that showed the serial permeabilization of outer and inner membranes of Escherichia. coli by defensins.
Kagan, B. L., Selsted, M. E., Ganz, T. & Lehrer, R. I. Antimicrobial defensin peptides form voltage-dependent ion-permeable channels in planar lipid bilayer membranes. Proc. Natl Acad. Sci. USA 87, 210–214 (1990).
Fujii, G., Selsted, M. E. & Eisenberg, D. Defensins promote fusion and lysis of negatively charged membranes. Protein Sci. 2, 1301–1312 (1993).
Wimley, W. C., Selsted, M. E. & White, S. H. Interactions between human defensins and lipid bilayers: evidence for formation of multimeric pores. Protein Sci. 3, 1362–1373 (1994).
Lohner, K., Latal, A., Lehrer, R. I. & Ganz, T. Differential scanning microcalorimetry indicates that human defensin, HNP-2, interacts specifically with biomembrane mimetic systems. Biochemistry 36, 1525–1531 (1997).
Wang, W., Cole, A. M., Hong, T., Waring, A. J. & Lehrer, R. I. Retrocyclin, an antiretroviral θ-defensin, is a lectin. J. Immunol. 170, 4708–4716 (2003).
Huang, H. W. Action of antimicrobial peptides: two-state model. Biochemistry 39, 8347–8352 (2000).
Matsuzaki, K. Why and how are peptide–lipid interactions utilized for self defence? Biochem. Soc. Trans. 29, 598–601 (2001).
Shai, Y. Mechanism of the binding, insertion and destabilization of phospholipid bilayer membranes by α-helical antimicrobial and cell non-selective membrane-lytic peptides. Biochim. Biophys. Acta 1462, 55–70 (1999).
Wilson, C. L. et al. Regulation of intestinal α-defensin activation by the metalloproteinase matrilysin in innate host defense. Science 286, 113–117 (1999). Ablation of Paneth cell defensin processing predisposed mice to lethal enteric infection with gram-negative bacteria.
Salzman, N. H., Ghosh, D., Huttner, K. M., Paterson, Y. & Bevins, C. L. Protection against enteric salmonellosis in transgenic mice expressing a human intestinal defensin. Nature 422, 522–526 (2003). The transgenic expression of human intestinal defensin at physiological levels protected mice against lethal intestinal infection with Salmonella typhimurium. The onset of protection was rapid — within a few hours — was confined to the oral route of infection, indicating that it was due to the direct antimicrobial effect of transgenic defensin.
Morrison, G., Kilanowski, F., Davidson, D. & Dorin, J. Characterization of the mouse β-defensin 1, Defb1, mutant mouse model. Infect. Immun. 70, 3053–3060 (2002).
Moser, C. et al. β-Defensin 1 contributes to pulmonary innate immunity in mice. Infect. Immun. 70, 3068–3072 (2002).
Nizet, V. et al. Innate antimicrobial peptide protects the skin from invasive bacterial infection. Nature 414, 454–457 (2001).
Groisman, E. A., Chiao, E., Lipps, C. J. & Heffron, F. Salmonella typhimurium phoP virulence gene is a transcriptional regulator. Proc. Natl Acad. Sci. USA 86, 7077–7081 (1989).
Miller, S. I., Pulkkinen, W. S., Selsted, M. E. & Mekalanos, J. J. Characterization of defensin resistance phenotypes associated with mutations in the phoP virulence regulon of Salmonella typhimurium. Infect. Immun. 58, 3706–3710 (1990).
Miller, S. I. PhoP/PhoQ: macrophage-specific modulators of Salmonella virulence? Mol. Microbiol. 5, 2073–2078 (1991).
Groisman, E. A., Heffron, F. & Solomon, F. Molecular genetic analysis of the Escherichia coli phoP locus. J. Bacteriol. 174, 486–491 (1992).
Guo, L. et al. Lipid A acylation and bacterial resistance against vertebrate antimicrobial peptides. Cell 95, 189–198 (1998).
Guina, T., Yi, E. C., Wang, H., Hackett, M. & Miller, S. I. A PhoP-regulated outer membrane protease of Salmonella enterica serovar typhimurium promotes resistance to α-helical antimicrobial peptides. J. Bacteriol. 182, 4077–4086 (2000).
Shafer, W. M., Qu, X., Waring, A. J. & Lehrer, R. I. Modulation of Neisseria gonorrhoeae susceptibility to vertebrate antibacterial peptides due to a member of the resistance/nodulation/division efflux pump family. Proc. Natl Acad. Sci. USA 95, 1829–1833 (1998).
Peschel, A. et al. Inactivation of the dlt operon in Staphylococcus aureus confers sensitivity to defensins, protegrins, and other antimicrobial peptides. J. Biol. Chem. 274, 8405–8410 (1999).
Peschel, A. et al. Staphylococcus aureus resistance to human defensins and evasion of neutrophil killing via the novel virulence factor MprF is based on modification of membrane lipids with l-lysine. J. Exp. Med. 193, 1067–1076 (2001).
Territo, M. C., Ganz, T., Selsted, M. E. & Lehrer, R. Monocyte-chemotactic activity of defensins from human neutrophils. J. Clin. Invest. 84, 2017–2020 (1989).
Chertov, O. et al. Identification of defensin-1, defensin-2, and CAP37/azurocidin as T-cell chemoattractant proteins released from interleukin-8-stimulated neutrophils. J. Biol. Chem. 271, 2935–2940 (1996).
Yang, D. et al. β-defensins: linking innate and adaptive immunity through dendritic and T cell CCR6. Science 286, 525–528 (1999). A key paper arguing that defensins also function as signals to adaptive immunity through their interactions with the CC-chemokine receptor 6 of dendritic cells and lymphocytes.
Yang, D., Chen, Q., Chertov, O. & Oppenheim, J. J. Human neutrophil defensins selectively chemoattract naive T and immature dendritic cells. J. Leukoc. Biol 68, 9–14 (2000).
Biragyn, A. et al. Toll-like receptor 4-dependent activation of dendritic cells by β-defensin 2. Science 298, 1025–1029 (2002).
Zhu, Q. Z., Singh, A. V., Bateman, A., Esch, F. & Solomon, S. The corticostatic (anti-ACTH) and cytotoxic activity of peptides isolated from fetal, adult and tumor-bearing lung. J. Steroid. Biochem. 27, 1017–1022 (1987).
Zhu, Q., Bateman, A., Singh, A. & Solomon, S. Isolation and biological activity of corticostatic peptides (anti-ACTH). Endocr. Res. 15, 129–149 (1989).
Solomon, S. et al. Corticostatic peptides. J. Steroid Biochem. Mol. Biol. 40, 391–398 (1991).
Tominaga, T. et al. Effects of corticostatin-I on rat adrenal cells in vitro. J. Endocrinol. 125, 287–292 (1990).
MacLeod, R. J. et al. Corticostatic peptides cause nifedipine-sensitive volume reduction in jejunal villus enterocytes. Proc. Natl Acad. Sci. USA 88, 552–556 (1991).
Bateman, A. et al. The isolation and characterization of a novel corticostatin/defensin-like peptide from the kidney. J. Biol. Chem. 271, 10654–10659 (1996).
Lencer, W. I. et al. Induction of epithelial chloride secretion by channel-forming cryptdins 2 and 3. Proc. Natl Acad. Sci. USA 94, 8585–8589 (1997).
Merlin, D., Yue, G., Lencer, W. I., Selsted, M. E. & Madara, J. L. Cryptdin-3 induces novel apical conductance(s) in Cl–secretory, including cystic fibrosis, epithelia. Am. J. Physiol. Cell Physiol. 280, C296–C302 (2001).
Smith, J. J., Travis, S. M., Greenberg, E. P. & Welsh, M. J. Cystic fibrosis airway epithelia fail to kill bacteria because of abnormal airway surface fluid. Cell 85, 229–236 (1996).
Matsui, H. et al. Evidence for periciliary liquid layer depletion, not abnormal ion composition, in the pathogenesis of cystic fibrosis airways disease. Cell 95, 1005–1015 (1998).
Wine, J. J. The genesis of cystic fibrosis lung disease. J. Clin. Invest 103, 309–312 (1999).
Verkman, A. S., Song, Y. & Thiagarajah, J. R. Role of airway surface liquid and submucosal glands in cystic fibrosis lung disease. Am. J. Physiol. Cell Physiol. 284, C2–C15 (2003).
Matsushita, I. et al. Genetic variants of human β-defensin-1 and chronic obstructive pulmonary disease. Biochem. Biophys. Res. Commun. 291, 17–22 (2002).
Jurevic, R. J., Bai, M., Chadwick, R. B., White, T. C. & Dale, B. A. Single-nucleotide polymorphisms (SNPs) in human β-defensin 1: high-throughput SNP assays and association with candida carriage in type I diabetics and nondiabetic controls. J. Clin. Microbiol. 41, 90–96 (2003).
Selsted, M. E. & Harwig, S. S. Determination of the disulfide array in the human defensin HNP-2. A covalently cyclized peptide. J. Biol. Chem. 264, 4003–4007 (1989).
Tang, Y. Q. & Selsted, M. E. Characterization of the disulfide motif in BNBD-12, an antimicrobial β-defensin peptide from bovine neutrophils. J. Biol. Chem. 268, 6649–6653 (1993).
Cornet, B. et al. Refined three-dimensional solution structure of insect defensin A. Structure. 3, 435–448 (1995).
Faurschou, M., Sorensen, O. E., Johnsen, A. H., Askaa, J. & Borregaard, N. Defensin-rich granules of human neutrophils: characterization of secretory properties. Biochim. Biophys. Acta 1591, 29–35 (2002).
Wilde, C. G., Griffith, J. E., Marra, M. N., Snable, J. L. & Scott, R. W. Purification and characterization of human neutrophil peptide 4, a novel member of the defensin family. J. Biol. Chem. 264, 11200–11203 (1989).
Jones, D. E. & Bevins, C. L. Defensin-6 mRNA in human Paneth cells: implications for antimicrobial peptides in host defense of the human bowel. FEBS Lett. 315, 187–192 (1993).
Jones, D. E. & Bevins, C. L. Paneth cells of the human small intestine express an antimicrobial peptide gene. J. Biol. Chem. 267, 23216–23225 (1992).
Bensch, K. W., Raida, M., Magert, H. J., Schulz-Knappe, P. & Forssmann, W. G. hBD-1: a novel β-defensin from human plasma. FEBS Lett. 368, 331–335 (1995).
Harder, J., Bartels, J., Christophers, E. & Schroder, J. M. Isolation and characterization of human β-defensin-3, a novel human inducible peptide antibiotic. J. Biol Chem. 276, 5707–5713 (2001).
Frye, M. et al. Expression of human α-defensin 5 (HD5) mRNA in nasal and bronchial epithelial cells. J. Clin. Pathol. 53, 770–773 (2000).
Quayle, A. J. et al. Gene expression, immunolocalization, and secretion of human defensin-5 in human female reproductive tract. Am. J. Pathol. 152, 1247–1258 (1998).
Com, E. et al. Expression of antimicrobial defensins in the male reproductive tract of rats, mice, and humans. Biol. Reprod. 68, 95–104 (2003).
Tang, Y. Q., Yuan, J., Miller, C. J. & Selsted, M. E. Isolation, characterization, cDNA cloning, and antimicrobial properties of two distinct subfamilies of α-defensins from rhesus macaque leukocytes. Infect. Immun. 67, 6139–6144 (1999).
Bals, R. et al. Rhesus monkey (Macaca mulatta) mucosal antimicrobial peptides are close homologues of human molecules. Clin. Diagn. Lab Immunol. 8, 370–375 (2001).
Boniotto, M., Tossi, A. & Crovella, S. β-defensin 2 in the rhesus monkey (Macaca mulatta) and the long-tailed macaque (M. fascicularis). Clin. Diagn. Lab Immunol. 9, 503–504 (2002).
Bals, R., Goldman, M. J. & Wilson, J. M. Mouse β-defensin 1 is a salt-sensitive antimicrobial peptide present in epithelia of the lung and urogenital tract. Infect. Immun. 66, 1225–1232 (1998).
Bals, R. et al. Mouse β-defensin 3 is an inducible antimicrobial peptide expressed in the epithelia of multiple organs. Infect. Immun. 67, 3542–3547 (1999).
Burd, R. S., Furrer, J. L., Sullivan, J. & Smith, A. L. Murine β-defensin-3 is an inducible peptide with limited tissue expression and broad-spectrum antimicrobial activity. Shock 18, 461–464 (2002).
Huttner, K. M., Kozak, C. A. & Bevins, C. L. The mouse genome encodes a single homolog of the antimicrobial peptide human β-defensin 1. FEBS Lett 413, 45–49 (1997).
Jia, H. P. et al. A novel murine β-defensin expressed in tongue, esophagus, and trachea. J. Biol Chem. 275, 33314–33320 (2000).
Morrison, G. M. et al. Mouse β-defensin-1 is a functional homolog of human β-defensin-1. Mamm. Genome 9, 453–457 (1998).
Morrison, G. M., Davidson, D. J. & Dorin, J. R. A novel mouse β-defensin, Defb2, which is upregulated in the airways by lipopolysaccharide. FEBS Lett. 442, 112–116 (1999).
Yamamoto, M. & Matsui, Y. Testis-specific expression of a novel mouse defensin-like gene, Tdl. Mech. Dev. 116, 217–221 (2002).
Shirafuji, Y., Oono, T., Kanzaki, H., Hirakawa, S. & Arata, J. Detection of cryptdin in mouse skin. Clin. Diagn. Lab Immunol. 6, 336–340 (1999).
Qu, X. D., Lloyd, K. C., Walsh, J. H. & Lehrer, R. I. Secretion of type II phospholipase A2 and cryptdin by rat small intestinal Paneth cells. Infect. Immun. 64, 5161–5165 (1996).
Jia, H. P. et al. Molecular cloning and characterization of rat genes encoding homologues of human β-defensins. Infect. Immun. 67, 4827–4833 (1999).
Zhang, G. et al. Molecular cloning and tissue expression of porcine β-defensin-1. FEBS Lett 424, 37–40 (1998).
Selsted, M. E. et al. Purification, primary structures, and antibacterial activities of β-defensins, a new family of antimicrobial peptides from bovine neutrophils. J. Biol. Chem. 268, 6641–6648 (1993).
Selsted, M. E. et al. Purification, primary structures, and antibacterial activities of β-defensins, a new family of antimicrobial peptides from bovine neutrophils. J. Biol. Chem. 271, 16430 (1996).
Harwig, S. S., Swiderek, K. M., Kokryakov, V. N., Lee, T. D. & Lehrer, R. I. in Techniques in Protein Chemistry V (ed. Crabb, J. W.) 81–88 (Academic Press, San Diego, 1994).
Porter, E. M. et al. Isolation of human intestinal defensins from ileal neobladder urine. FEBS Lett. 434, 272–276 (1998).
Satoh, Y., Ishikawa, K., Oomori, Y., Takeda, S. & Ono, K. Bethanechol and a G-protein activator, NaF/AlCl3, induce secretory response in Paneth cells of mouse intestine. Cell Tissue Res. 269, 213–220 (1992).
Diamond, G., Russell, J. P. & Bevins, C. L. Inducible expression of an antibiotic peptide gene in lipopolysaccharide-challenged tracheal epithelial cells. Proc. Natl Acad. Sci. USA 93, 5156–5160 (1996).
Acknowledgements
I would like to acknowledge many influential discussions with R. Lehrer and C. Bevins. This work would not have been possible without the consistent financial support from the National Institutes of Health and the Will Rogers Fund.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ganz, T. Defensins: antimicrobial peptides of innate immunity. Nat Rev Immunol 3, 710–720 (2003). https://doi.org/10.1038/nri1180
Issue Date:
DOI: https://doi.org/10.1038/nri1180
This article is cited by
-
Effects of topical application of resveratrol on tight junction barrier and antimicrobial compound production in lactating goat mammary glands
Veterinary Research (2024)
-
Bacillus subtilis Induces Human Beta Defensin-2 Through its Lipoproteins in Human Intestinal Epithelial Cells
Probiotics and Antimicrobial Proteins (2024)
-
Influence of gut microbiome on health and development of penaeid shrimps
Aquatic Sciences (2024)
-
Antimicrobial Peptides Targeting Streptococcus mutans: Current Research on Design, Screening and Efficacy
Current Microbiology (2024)
-
Exploring the potential role of defensins in differential vector competence of body and head lice for Bartonella quintana
Parasites & Vectors (2023)