Abstract
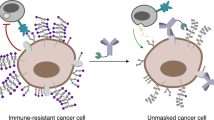
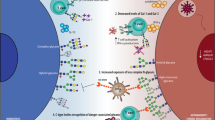
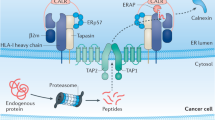
Tumour growth is accompanied by tumour evasion of the immune system, a process that is facilitated by immune checkpoint molecules such as programmed cell death protein 1 (PD1). However, the role of tumour glycosylation in immune evasion has mostly been overlooked, despite the fact that aberrant tumour glycosylation alters how the immune system perceives the tumour and can also induce immunosuppressive signalling through glycan-binding receptors. As such, specific glycan signatures found on tumour cells can be considered as a novel type of immune checkpoint. In parallel, glycosylation of tumour proteins generates neo-antigens that can serve as targets for tumour-specific T cells. In this Opinion article, we highlight how the tumour 'glyco-code' modifies immunity and suggest that targeting glycans could offer new therapeutic opportunities.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Dunn, G. P., Bruce, A. T., Ikeda, H., Old, L. J. & Schreiber, R. D. Cancer immunoediting: from immunosurveillance to tumor escape. Nat. Immunol. 3, 991–998 (2002).
McGranahan, N. & Swanton, C. Clonal heterogeneity and tumor evolution: past, present, and the future. Cell 168, 613–628 (2017).
Blank, C. U., Haanen, J. B., Ribas, A. & Schumacher, T. N. The “cancer immunogram”. Science 352, 658–660 (2016).
Chen, D. S. & Mellman, I. Oncology meets immunology: the cancer-immunity cycle. Immunity 39, 1–10 (2013).
Sharma, P. & Allison, J. P. The future of immune checkpoint therapy. Science 348, 56–61 (2015).
Varki, A. Since there are PAMPs and DAMPs, there must be SAMPs? Glycan “self-associated molecular patterns” dampen innate immunity, but pathogens can mimic them. Glycobiology 21, 1121–1124 (2011).
van Kooyk, Y. & Rabinovich, G. A. Protein-glycan interactions in the control of innate and adaptive immune responses. Nat. Immunol. 9, 593–601 (2008).
Cagnoni, A. J., Perez Saez, J. M., Rabinovich, G. A. & Marino, K. V. Turning-off signaling by siglecs, selectins, and galectins: chemical inhibition of glycan-dependent interactions in cancer. Front. Oncol. 6, 109 (2016).
Pinho, S. S. & Reis, C. A. Glycosylation in cancer: mechanisms and clinical implications. Nat. Rev. Cancer 15, 540–555 (2015).
Koike, T. et al. Hypoxia induces adhesion molecules on cancer cells: a missing link between Warburg effect and induction of selectin-ligand carbohydrates. Proc. Natl Acad. Sci. USA 101, 8132–8137 (2004).
Radhakrishnan, P. et al. Immature truncated O-glycophenotype of cancer directly induces oncogenic features. Proc. Natl Acad. Sci. USA 111, E4066–E4075 (2014).
Ju, T. & Cummings, R. D. Protein glycosylation: chaperone mutation in Tn syndrome. Nature 437, 1252 (2005).
Gill, D. J. et al. Initiation of GalNAc-type O-glycosylation in the endoplasmic reticulum promotes cancer cell invasiveness. Proc. Natl Acad. Sci. USA 110, E3152–E3161 (2013).
Nguyen, A. T. et al. Organelle specific O-glycosylation drives MMP14 activation, tumor growth, and metastasis. Cancer Cell 32, 639–653.e6 (2017).
van Gisbergen, K. P., Aarnoudse, C. A., Meijer, G. A., Geijtenbeek, T. B. & van Kooyk, Y. Dendritic cells recognize tumor-specific glycosylation of carcinoembryonic antigen on colorectal cancer cells through dendritic cell-specific intercellular adhesion molecule-3-grabbing nonintegrin. Cancer Res. 65, 5935–5944 (2005).
Gringhuis, S. I. et al. Fucose-based PAMPs prime dendritic cells for follicular T helper cell polarization via DC-SIGN-dependent IL-27 production. Nat. Commun. 5, 5074 (2014).
Garcia-Vallejo, J. J. et al. CNS myelin induces regulatory functions of DC-SIGN-expressing, antigen-presenting cells via cognate interaction with MOG. J. Exp. Med. 211, 1465–1483 (2014).
van Vliet, S. J. et al. MGL signaling augments TLR2-mediated responses for enhanced IL-10 and TNF-alpha secretion. J. Leukoc. Biol. 94, 315–323 (2013).
van Vliet, S. J., Gringhuis, S. I., Geijtenbeek, T. B. & van Kooyk, Y. Regulation of effector T cells by antigen-presenting cells via interaction of the C-type lectin MGL with CD45. Nat. Immunol. 7, 1200–1208 (2006).
Lenos, K. et al. MGL ligand expression is correlated to BRAF mutation and associated with poor survival of stage III colon cancer patients. Oncotarget 6, 26278–26290 (2015).
Macauley, M. S., Crocker, P. R. & Paulson, J. C. Siglec-mediated regulation of immune cell function in disease. Nat. Rev. Immunol. 14, 653–666 (2014).
Perdicchio, M. et al. Tumor sialylation impedes T cell mediated anti-tumor responses while promoting tumor associated-regulatory T cells. Oncotarget 7, 8771–8782 (2016).
Perdicchio, M. et al. Sialic acid-modified antigens impose tolerance via inhibition of T-cell proliferation and de novo induction of regulatory T cells. Proc. Natl Acad. Sci. USA 113, 3329–3334 (2016).
Julien, S., Videira, P. A. & Delannoy, P. Sialyl-tn in cancer: (how) did we miss the target? Biomolecules 2, 435–466 (2012).
Carrascal, M. A. et al. Sialyl Tn-expressing bladder cancer cells induce a tolerogenic phenotype in innate and adaptive immune cells. Mol. Oncol. 8, 753–765 (2014).
Beatson, R. et al. The mucin MUC1 modulates the tumor immunological microenvironment through engagement of the lectin Siglec-9. Nat. Immunol. 17, 1273–1281 (2016).
Jandus, C. et al. Interactions between Siglec-7/9 receptors and ligands influence NK cell-dependent tumor immunosurveillance. J. Clin. Invest. 124, 1810–1820 (2014).
Li, C. W. et al. Glycosylation and stabilization of programmed death ligand-1 suppresses T-cell activity. Nat. Commun. 7, 12632 (2016).
Mendez-Huergo, S. P., Blidner, A. G. & Rabinovich, G. A. Galectins: emerging regulatory checkpoints linking tumor immunity and angiogenesis. Curr. Opin. Immunol. 45, 8–15 (2017).
Toscano, M. A. et al. Differential glycosylation of TH1, TH2 and TH-17 effector cells selectively regulates susceptibility to cell death. Nat. Immunol. 8, 825–834 (2007).
Ilarregui, J. M. et al. Tolerogenic signals delivered by dendritic cells to T cells through a galectin-1-driven immunoregulatory circuit involving interleukin 27 and interleukin 10. Nat. Immunol. 10, 981–991 (2009).
Rubinstein, N. et al. Targeted inhibition of galectin-1 gene expression in tumor cells results in heightened T cell-mediated rejection; a potential mechanism of tumor-immune privilege. Cancer Cell 5, 241–251 (2004).
Tsuboi, S. et al. A novel strategy for evasion of NK cell immunity by tumours expressing core2 O-glycans. EMBO J. 30, 3173–3185 (2011).
Dardalhon, V. et al. Tim-3/galectin-9 pathway: regulation of Th1 immunity through promotion of CD11b+Ly-6G+ myeloid cells. J. Immunol. 185, 1383–1392 (2010).
Vlad, A. M. et al. Complex carbohydrates are not removed during processing of glycoproteins by dendritic cells: processing of tumor antigen MUC1 glycopeptides for presentation to major histocompatibility complex class II-restricted T cells. J. Exp. Med. 196, 1435–1446 (2002).
Apostolopoulos, V. et al. A glycopeptide in complex with MHC class I uses the GalNAc residue as an anchor. Proc. Natl Acad. Sci. USA 100, 15029–15034 (2003).
Vogt, G. et al. Gains of glycosylation comprise an unexpectedly large group of pathogenic mutations. Nat. Genet. 37, 692–700 (2005).
Malaker, S. A. et al. Identification of glycopeptides as posttranslationally modified neoantigens in leukemia. Cancer Immunol. Res. 5, 376–384 (2017).
Amin, R. et al. DC-SIGN-expressing macrophages trigger activation of mannosylated IgM B-cell receptor in follicular lymphoma. Blood 126, 1911–1920 (2015).
Hollander, N. & Haimovich, J. Altered N-linked glycosylation in follicular lymphoma and chronic lymphocytic leukemia: involvement in pathogenesis and potential therapeutic targeting. Front. Immunol. 8, 912 (2017).
Kaur, S., Kumar, S., Momi, N., Sasson, A. R. & Batra, S. K. Mucins in pancreatic cancer and its microenvironment. Nat. Rev. Gastroenterol. Hepatol. 10, 607–620 (2013).
Drake, R. R. Glycosylation and cancer: moving glycomics to the forefront. Adv. Cancer Res. 126, 1–10 (2015).
Kailemia, M. J. et al. Recent advances in the mass spectrometry methods for glycomics and cancer. Anal. Chem. 90, 208–224 (2018).
Drake, R. R. et al. MALDI mass spectrometry imaging of N-linked glycans in cancer tissues. Adv. Cancer Res. 134, 85–116 (2017).
Kunzke, T. et al. Native glycan fragments detected by MALDI-FT-ICR mass spectrometry imaging impact gastric cancer biology and patient outcome. Oncotarget 8, 68012–68025 (2017).
Stadlmann, J. et al. Comparative glycoproteomics of stem cells identifies new players in ricin toxicity. Nature 549, 538–542 (2017).
Cummings, R. D. The haystack is full of needles: technology rescues sugars! Mol. Cell 68, 827–829 (2017).
Kim, J. et al. Detection of early pancreatic ductal adenocarcinoma with thrombospondin-2 and CA19-9 blood markers. Sci. Transl Med. 9, eaah5583 (2017).
Gilgunn, S., Conroy, P. J., Saldova, R., Rudd, P. M. & O'Kennedy, R. J. Aberrant PSA glycosylation — a sweet predictor of prostate cancer. Nat. Rev. Urol. 10, 99–107 (2013).
Yang, A. P. et al. CA72-4 combined with CEA, CA125 and CAl9-9 improves the sensitivity for the early diagnosis of gastric cancer. Clin. Chim. Acta 437, 183–186 (2014).
Tiernan, J. P. et al. Carcinoembryonic antigen is the preferred biomarker for in vivo colorectal cancer targeting. Br. J. Cancer 108, 662–667 (2013).
Campos, D. et al. Probing the O-glycoproteome of gastric cancer cell lines for biomarker discovery. Mol. Cell Proteomics 14, 1616–1629 (2015).
Syed, P. et al. Role of lectin microarrays in cancer diagnosis. Proteomics 16, 1257–1265 (2016).
Gill, D. J., Chia, J., Senewiratne, J. & Bard, F. Regulation of O-glycosylation through Golgi-to-ER relocation of initiation enzymes. J. Cell Biol. 189, 843–858 (2010).
Ashkani, J. & Naidoo, K. J. Glycosyltransferase gene expression profiles classify cancer types and propose prognostic subtypes. Sci. Rep. 6, 26451 (2016).
Agrawal, P. et al. A systems biology approach identifies FUT8 as a driver of melanoma metastasis. Cancer Cell 31, 804–819.e7 (2017).
Dube, D. H. & Bertozzi, C. R. Glycans in cancer and inflammation — potential for therapeutics and diagnostics. Nat. Rev. Drug Discov. 4, 477–488 (2005).
Miles, D. & Papazisis, K. Rationale for the clinical development of STn-KLH (Theratope) and anti-MUC-1 vaccines in breast cancer. Clin. Breast Cancer 3 (Suppl. 4), S134–S138 (2003).
Miles, D. et al. Phase III multicenter clinical trial of the sialyl-TN (STn)-keyhole limpet hemocyanin (KLH) vaccine for metastatic breast cancer. Oncologist 16, 1092–1100 (2011).
Xiao, H., Woods, E. C., Vukojicic, P. & Bertozzi, C. R. Precision glycocalyx editing as a strategy for cancer immunotherapy. Proc. Natl Acad. Sci. USA 113, 10304–10309 (2016).
Bull, C. et al. Targeting aberrant sialylation in cancer cells using a fluorinated sialic acid analog impairs adhesion, migration, and in vivo tumor growth. Mol. Cancer Ther. 12, 1935–1946 (2013).
Jennewein, M. F. & Alter, G. Immunoregulatory roles of antibody glycosylation. Trends Immunol. 38, 358–372 (2017).
Posey, A. D. Jr. et al. Engineered CAR T cells targeting the cancer-associated Tn-glycoform of the membrane mucin MUC1 control adenocarcinoma. Immunity 44, 1444–1454 (2016).
Tacken, P. J., de Vries, I. J., Torensma, R. & Figdor, C. G. Dendritic-cell immunotherapy: from ex vivo loading to in vivo targeting. Nat. Rev. Immunol. 7, 790–802 (2007).
Unger, W. W. et al. Antigen targeting to dendritic cells combined with transient regulatory T cell inhibition results in long-term tumor regression. Oncoimmunology 4, e970462 (2015).
Chen, D. S. & Mellman, I. Elements of cancer immunity and the cancer-immune set point. Nature 541, 321–330 (2017).
Lisacek, F. et al. Databases and Associated Tools for Glycomics and Glycoproteomics. Methods Mol. Biol. 1503, 235–264 (2017).
Varki, A. et al. Essentials of Glycobiology (Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY, 2009).
Gould, S. E., Junttila, M. R. & de Sauvage, F. J. Translational value of mouse models in oncology drug development. Nat. Med. 21, 431–439 (2015).
Day, C. P., Merlino, G. & Van Dyke, T. Preclinical mouse cancer models: a maze of opportunities and challenges. Cell 163, 39–53 (2015).
Garcia-Vallejo, J. J. & van Kooyk, Y. The physiological role of DC-SIGN: a tale of mice and men. Trends Immunol. 34, 482–486 (2013).
Costache, M. et al. Evolution of fucosyltransferase genes in vertebrates. J. Biol. Chem. 272, 29721–29728 (1997).
Varki, A. Uniquely human evolution of sialic acid genetic and biology. Proc. Natl ACad. Sci. USA 107 (Suppl. 2), 8939–8946 (2010).
Uhlen, M. et al. Proteomics. Tissue-based map of the human proteome. Science 347, 1260419 (2015).
Acknowledgements
The authors acknowledge support from the European Research Council (ERC-339977-Glycotreat; Y.v.K. and S.T.T.S.) and the European Union Horizon 2020 (Marie Skłodowska-Curie, Grant agreement No. 642870, and the European Training Network IMMUNOSHAPE (E.R.)). The authors thank the fruitful discussions with S. van Vliet, J. J. Garcia Vallejo and the contributions of our group-members that work on the immuno-glyco-code of cancer.
Author information
Authors and Affiliations
Contributions
Y.v.K. and E.R. researched data for the article, made substantial contributions to the discussion of content and wrote, reviewed and edited the manuscript before submission. S.T.T.S. researched data for the article and made substantial contributions to the discussion of content and the writing of the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary S1 (movie)
Supplementary Video hosted on Brightcove/Video Cloud nri.2018.3-S1.mp4 https://studio.brightcove.com/products/videocloud/media/videos/5716519085001 (MP4 161928 kb)
Rights and permissions
About this article
Cite this article
RodrÍguez, E., Schetters, S. & van Kooyk, Y. The tumour glyco-code as a novel immune checkpoint for immunotherapy. Nat Rev Immunol 18, 204–211 (2018). https://doi.org/10.1038/nri.2018.3
Published:
Issue Date:
DOI: https://doi.org/10.1038/nri.2018.3
This article is cited by
-
Therapeutic and immunomodulatory potentials of mesenchymal stromal/stem cells and immune checkpoints related molecules
Biomarker Research (2024)
-
FUT8-mediated aberrant N-glycosylation of SEMA7A promotes head and neck squamous cell carcinoma progression
International Journal of Oral Science (2024)
-
Integrative proteomics and n-glycoproteomics reveal the synergistic anti-tumor effects of aspirin- and gemcitabine-based chemotherapy on pancreatic cancer cells
Cellular Oncology (2024)
-
Pancreatic cancer-associated fibroblasts modulate macrophage differentiation via sialic acid-Siglec interactions
Communications Biology (2024)
-
A pan-cancer analysis revealing the role of LFNG, MFNG and RFNG in tumor prognosis and microenvironment
BMC Cancer (2023)