Key Points
-
The regulator of G-protein signalling (RGS)-protein family, which has more than 30 members, is an intriguing set of drug targets for central nervous system (CNS) therapeutics.
-
In addition to the conserved RGS-box, they carry a diverse range of other signal-transduction modulatory and scaffolding domains (for example, RhoGEF (Rho guanine-nucleotide-exchange factor), PDZ (PSD95, Dlg and Z0-1/2) and PTB (phosphotyrosine binding) domains).
-
They show differential expression in many brain regions and are dramatically up- or down-regulated by pharmacological stimuli and pathophysiological processes.
-
They strongly modulate G-protein-coupled receptor (GPCR) signalling, and their inhibition should mimic or enhance the action of classical GPCR-agonist drugs.
-
RGS inhibitors could create a new pharmacological class, with properties such as 'specificity enhancement' and 'desensitization reduction', for existing GPCR agonists.
Abstract
G-protein-coupled receptors (GPCRs) are major targets for drug discovery. The regulator of G-protein signalling (RGS)-protein family has important roles in GPCR signal transduction. RGS proteins contain a conserved RGS-box, which is often accompanied by other signalling regulatory elements. RGS proteins accelerate the deactivation of G proteins to reduce GPCR signalling; however, some also have an effector function and transmit signals. Combining GPCR agonists with RGS inhibitors should potentiate responses, and could markedly increase the agonist's regional specificity. The diversity of RGS proteins with highly localized and dynamically regulated distributions in brain makes them attractive targets for pharmacotherapy of central nervous system disorders.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Gilman, A. G. G proteins: transducers of receptor-generated signals. Annu. Rev. Biochem. 56, 615–649 (1987).
Arshavsky, V. Y. & Pugh, E. N. Jr. Lifetime regulation of G protein-effector complex: emerging importance of RGS proteins. Neuron 20, 11–14 (1998).
Chan, R. K. & Otte, C. A. Isolation and genetic analysis of Saccharomyces cerevisiae mutants supersensitive to G1 arrest by a factor and α-factor pheromones. Mol. Cell. Biol. 2, 11–20 (1982).
Koelle, M. R. & Horvitz, H. R. EGL-10 regulates G protein signalling in the C. elegans nervous system and shares a conserved domain with many mammalian proteins. Cell 84, 115–125 (1996).
Wu, H. K. et al. Differential expression of a basic helix–loop–helix phosphoprotein gene, G0S8, in acute leukemia and localization to human chromosome 1q31. Leukemia 9, 1291–1298 (1995).
De Vries, L., Mousli, M., Wurmser, A. & Farquhar, M. G. GAIP, a protein that specifically interacts with the trimeric G protein Gαi3, is a member of a protein family with a highly conserved core domain. Proc. Natl Acad. Sci. USA 92, 11916–11920 (1995).
Siderovski, D. P., Hessel, A., Chung, S., Mak, T. W. & Tyers, M. A new family of regulators of G-protein-coupled receptors? Curr. Biol. 6, 211–212 (1996).
Druey, K. M., Blumer, K. J., Kang, V. H. & Kehrl, J. H. Inhibition of G-protein-mediated MAP kinase activation by a new mammalian gene family. Nature 379, 742–746 (1996).
Berman, D. M., Wilkie, T. M. & Gilman, A. G. GAIP and RGS4 are GTPase-activating proteins for the Gi subfamily of G protein α subunits. Cell 86, 445–452 (1996).
Hunt, T. W., Fields, T. A., Casey, P. J. & Peralta, E. G. RGS10 is a selective activator of Gαi GTPase activity. Nature 383, 175–177 (1996).
Watson, N., Linder, M. E., Druey, K. M., Kehrl, J. H. & Blumer, K. J. RGS family members: GTPase-activating proteins for heterotrimeric G-protein α-subunits. Nature 383, 172–175 (1996).
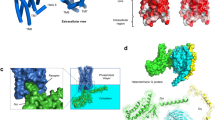
Tesmer, J. J., Berman, D. M., Gilman, A. G. & Sprang, S. R. Structure of RGS4 bound to AlF4-activated G(iα1): stabilization of the transition state for GTP hydrolysis. Cell 89, 251–261 (1997).This first crystal structure of an RGS protein established the mechanism of GAP activity to be stabilization of the transition state for GTP hydrolysis. Furthermore, the complex of RGS4 and G i α 1 (along with the NMR structure of free RGS4 in reference 59 ) provides molecular details of the target site for RGS-box inhibitors.
De Vries, L. & Gist Farquhar, M. RGS proteins: more than just GAPs for heterotrimeric G proteins. Trends Cell Biol. 9, 138–144 (1999).
Siderovski, D. P., Strockbine, B. & Behe, C. I. Whither goest the RGS proteins? Crit. Rev. Biochem. Mol. Biol. 34, 215–251 (1999).
Hart, M. J. et al. Direct stimulation of the guanine nucleotide exchange activity of p115 RhoGEF by Gα13. Science 280, 2112–2114 (1998).This paper presents the first evidence for an RGS-domain-containing protein acting as a GAP at Gα 12 or Gα 13 . In addition, they established a direct molecular link between Gα 13 and Rho, proving that an RGS protein can serve as an effector as well as a negative regulator of G-protein signalling.
Fukuhara, S., Murga, C., Zohar, M., Igishi, T. & Gutkind, J. S. A novel PDZ domain containing guanine nucleotide exchange factor links heterotrimeric G proteins to Rho. J. Biol. Chem. 274, 5868–5879 (1999).
Kourlas, P. J. et al. Identification of a gene at 11q23 encoding a guanine nucleotide exchange factor: evidence for its fusion with MLL in acute myeloid leukemia. Proc. Natl Acad. Sci. USA 97, 2145–2150 (2000).
Reuther, G. W. et al. Leukemia-associated Rho guanine nucleotide exchange factor, a Dbl family protein found mutated in leukemia, causes transformation by activation of RhoA. J. Biol. Chem. 276, 27145–27151 (2001).
De Vries, L., Zheng, B., Fischer, T., Elenko, E. & Farquhar, M. G. The regulator of G protein signalling family. Annu. Rev. Pharmacol. Toxicol. 40, 235–271 (2000).
Ross, E. M. & Wilkie, T. M. GTPase-activating proteins for heterotrimeric G proteins: regulators of G protein signalling (RGS) and RGS-like proteins. Annu. Rev. Biochem. 69, 795–827 (2000).
Zhong, H. & Neubig, R. R. Regulator of G protein signalling proteins: novel multifunctional drug targets. J. Pharmacol. Exp. Ther. 297, 837–845 (2001).
DiBello, P. R. et al. Selective uncoupling of RGS action by a single point mutation in the G protein α-subunit. J. Biol. Chem. 273, 5780–5784 (1998).
Lan, K. L. et al. A point mutation in Gαo and Gαi1 blocks interaction with regulator of G protein signalling proteins. J. Biol. Chem. 273, 12794–12797 (1998).References 22 and 23 showed the selective loss of RGS activity on Gα subunits with a glycine-to-serine mutation in the switch 1 region.
Lan, K. L. Mechanism and specificity of G protein regulation by RGS proteins. Thesis, Univ. Michigan (1999).
Jeong, S. W. & Ikeda, S. R. Endogenous regulator of G-protein signalling proteins modify N-type calcium channel modulation in rat sympathetic neurons. J. Neurosci. 20, 4489–4496 (2000).With the mutation that renders G o α subunits insensitive to RGS-box GAP activity, the authors show a role for endogenous RGS in rat sympathetic neuron proteins to accelerate the kinetics of noradrenaline signalling to N-type Ca2+-channel inhibition. They also show the substantial enhancement of agonist potency that is achievable by eliminating RGS action on G proteins.
Jeong, S. W. & Ikeda, S. R. Differential regulation of G protein-gated inwardly rectifying K(+) channel kinetics by distinct domains of RGS8. J. Physiol. 535, 335–347 (2001).
Mark, M. D. & Herlitze, S. G-protein mediated gating of inward-rectifier K+ channels. Eur. J. Biochem. 267, 5830–5836 (2000).
Chen, H. & Lambert, N. A. Endogenous regulators of G protein signalling proteins regulate presynaptic inhibition at rat hippocampal synapses. Proc. Natl Acad. Sci. USA 97, 12810–12815 (2000).
Howland, D. S. et al. Transgenic expression of an RGS-resistant Gqa mutant (G188S) in rat brain. Soc. Neurosci. Abstr. 26, 239.1 (2000).
Rosenzweig-Lipson, S. et al. Potentiation of 5-HT2C and 5-HT2A mediated effects in transgenic RGS-insensitive Gq (G188S) mutant rats. Soc. Neurosci. Abstr. 26, 239.3 (2000).
Grauer, S. M. et al. Behavioral parallels between transgenic RGS-insensitive Gqα mutant (G188S) rats and those treated with lithium chloride. Soc. Neurosci. Abstr. 27, 959.2 (2001).
Nahorski, S. R., Ragan, C. I. & Challiss, R. A. Lithium and the phosphoinositide cycle: an example of uncompetitive inhibition and its pharmacological consequences. Trends Pharmacol. Sci. 12, 297–303 (1991).
Melliti, K., Meza, U. & Adams, B. Muscarinic stimulation of α1E Ca channels is selectively blocked by the effector antagonist function of RGS2 and phospholipase C-β1. J. Neurosci. 20, 7167–7173 (2000).
Shi, C. S., Sinnarajah, S., Cho, H., Kozasa, T. & Kehrl, J. H. G13α-mediated PYK2 activation. PYK2 is a mediator of G13α-induced serum response element-dependent transcription. J. Biol. Chem. 275, 24470–24476 (2000).
Snow, B. E. et al. GTPase activating specificity of RGS12 and binding specificity of an alternatively spliced PDZ (PSD-95/Dlg/ZO-1) domain. J. Biol. Chem. 273, 17749–17755 (1998).
Xu, X. et al. RGS proteins determine signalling specificity of Gq-coupled receptors. J. Biol. Chem. 274, 3549–3456 (1999).
Zeng, W. et al. The N-terminal domain of RGS4 confers receptor selective inhibition of G protein signalling. J. Biol. Chem. 273, 34687–34691 (1998).
Diversé-Pierluissi, M. A. et al. Regulators of G protein signalling proteins as determinants of the rate of desensitization of presynaptic calcium channels. J. Biol. Chem. 274, 14490–14494 (1999).
Schiff, M. L. et al. Tyrosine-kinase-dependent recruitment of RGS12 to the N-type calcium channel. Nature 408, 723–727 (2000).References 38 and 39 establish specific and non-overlapping roles for RGS4, GAIP and RGS12 in regulating desensitization of neurotransmitter signalling to the inhibition of presynaptic N-type calcium channels in dorsal root ganglia.
Snow, B. E., Brothers, G. M. & Siderovski, D. P. Molecular cloning of regulators of G-protein signalling family members and characterization of binding specificity of RGS12 PDZ domain. Methods Enzymol. 344, 740–761 (2002).
Gold, S. J., Ni, Y. G., Dohlman, H. G. & Nestler, E. J. Regulators of G-protein signalling (RGS) proteins: region-specific expression of nine subtypes in rat brain. J. Neurosci. 17, 8024–8037 (1997).The remarkable region-specific expression of different RGS proteins shown in this in situ hybridization study provides the basis for tissue-specific selectivity enhancement by RGS protein inhibitors.
Ingi, T. et al. Dynamic regulation of RGS2 suggests a novel mechanism in G-protein signalling and neuronal plasticity. J. Neurosci. 18, 7178–7188 (1998).
Ni, Y. G. et al. Region-specific regulation of RGS4 (regulator of G-protein-signalling protein type 4) in brain by stress and glucocorticoids: in vivo and in vitro studies. J. Neurosci. 19, 3674–3680 (1999).
Mirnics, K., Middleton, F. A., Stanwood, G. D., Lewis, D. A. & Levitt, P. Disease-specific changes in regulator of G-protein signalling 4 (RGS4) expression in schizophrenia. Mol. Psychiatry 6, 293–301 (2001).
Burchett, S. A., Bannon, M. J. & Granneman, J. G. RGS mRNA expression in rat striatum: modulation by dopamine receptors and effects of repeated amphetamine administration. J. Neurochem. 72, 1529–1533 (1999).
Robinet, E. A., Geurts, M., Maloteaux, J. M. & Pauwels, P. J. Chronic treatment with certain antipsychotic drugs preserves upregulation of regulator of G-protein signalling 2 mRNA in rat striatum as opposed to c-Fos mRNA. Neurosci. Lett. 307, 45–48 (2001).
Burchett, S. A., Volk, M. L., Bannon, M. J. & Granneman, J. G. Regulators of G protein signalling: rapid changes in mRNA abundance in response to amphetamine. J. Neurochem. 70, 2216–2219 (1998).
Oliveira-Dos-Santos, A. J. et al. Regulation of T cell activation, anxiety, and male aggression by RGS2. Proc. Natl Acad. Sci. USA 97, 12272–12277 (2000).The authors show that genetic ablation of Rgs2 from the mouse genome has an anxiogenic effect, indicating a central role for RGS2 in the CNS circuitry that controls behaviours of fear and aggression.
Heximer, S. P., Watson, N., Linder, M. E., Blumer, K. J. & Hepler, J. R. RGS2/G0S8 is a selective inhibitor of Gqα function. Proc. Natl Acad. Sci. USA 94, 14389–14393 (1997).
Sinnarajah, S. et al. RGS2 regulates signal transduction in olfactory neurons by attenuating activation of adenylyl cyclase III. Nature 409, 1051–1055 (2001).
He, W., Cowan, C. W. & Wensel, T. G. RGS9, a GTPase accelerator for phototransduction. Neuron 20, 95–102 (1998).
Granneman, J. G. et al. Molecular characterization of human and rat RGS9L, a novel splice variant enriched in dopamine target regions, and chromosomal localization of the RGS9 gene. Mol. Pharmacol. 54, 687–694 (1998).
Rahman, Z. et al. Cloning and characterization of RGS9-2: a striatal-enriched alternatively spliced product of the RGS9 gene. J. Neurosci. 19, 2016–2026 (1999).
Chen, C. K. et al. Slowed recovery of rod photoresponse in mice lacking the GTPase accelerating protein RGS9-1. Nature 403, 557–560 (2000).
Lyubarsky, A. L. et al. RGS9-1 is required for normal inactivation of mouse cone phototransduction. Mol. Vis. 7, 71–78 (2001).
Rahman, Z. et al. Functional role of RGS9-2 in striatum. Soc. Neurosci. Abstr. 27, 221.5 (2001).
Tekumalla, P. K. et al. Elevated levels of ΔFosB and RGS9 in striatum in Parkinson's disease. Biol. Psychiatry 50, 813–816 (2001).
De Alba, E., De Vries, L., Farquhar, M. G. & Tjandra, N. Solution structure of human GAIP (Gα interacting protein): a regulator of G protein signalling. J. Mol. Biol. 291, 927–939 (1999).
Moy, F. J. et al. NMR structure of free RGS4 reveals an induced conformational change upon binding Gα. Biochemistry 39, 7063–7073 (2000).
Slep, K. C. et al. Structural determinants for regulation of phosphodiesterase by a G protein at 2.0 A. Nature 409, 1071–1077 (2001).
Zhang, J. et al. Modulation of RGS (regulators of G-protein signalling) proteins. Soc. Neurosci. Abstr. 27, 43.9 (2001).
Grafstein-Dunn, E., Wang, Y., Lee, Y. T., Wang, Q. & Young, K. H. Identification of peptides that modulate regulator of G-protein signalling 4 (RGS4) function. Soc. Neurosci. Abstr. 27, 43.10 (2001).
Longenecker, K. L., Lewis, M. E., Chikumi, H., Gutkind, J. S. & Derewenda, Z. S. Structure of the RGS-like domain from PDZ-RhoGEF: linking heterotrimeric G protein-coupled signalling to Rho GTPases. Structure (Camb.) 9, 559–569 (2001).
Chen, Z., Wells, C. D., Sternweis, P. C. & Sprang, S. R. Structure of the rgRGS domain of p115RhoGEF. Nature Struct. Biol. 8, 805–809 (2001).
Spink, K. E., Polakis, P. & Weis, W. I. Structural basis of the axin–adenomatous polyposis coli interaction. EMBO J. 19, 2270–2279 (2000).
Popov, S. G., Krishna, U. M., Falck, J. R. & Wilkie, T. M. Ca2+/Calmodulin reverses phosphatidylinositol 3,4,5-trisphosphate-dependent inhibition of regulators of G protein-signalling GTPase-activating protein activity. J. Biol. Chem. 275, 18962–18968 (2000).This paper shows that the negative regulation of RGS4 GAP activity by InsP 3 and its reversal by calmodulin provide a mechanistic basis for the development of RGS agonists as well as inhibitors.
Ishii, M. et al. Ca(2+) elevation evoked by membrane depolarization regulates G protein cycle via RGS proteins in the heart. Circ. Res. 89, 1045–1050 (2001).
Kimple, R. A. et al. RGS12 and RGS14 GoLoco motifs are Gαi interaction sites with guanine nucleotide dissociation inhibitor activity. J. Biol. Chem. 276, 29275–29281 (2001).
Shakespeare, W. et al. Structure-based design of an osteoclast-selective, nonpeptide src homology 2 inhibitor with in vivo antiresorptive activity. Proc. Natl Acad. Sci. USA 97, 9373–9378 (2000).
Garnier, M. et al. RGS4 upregulation in conditions of neuropathic pain and morphine tolerance. Soc. Neurosci. Abstr. 27, 282.10 (2001).
Bohn, L. M., Gainetdinov, R. R., Lin, F. T., Lefkowitz, R. J. & Caron, M. G. μ-Opioid receptor desensitization by β-arrestin-2 determines morphine tolerance but not dependence. Nature 408, 720–723 (2000).
Schuler, V. et al. Epilepsy, hyperalgesia, impaired memory, and loss of pre- and postsynaptic GABA(B) responses in mice lacking GABA(B(1)). Neuron 31, 47–58 (2001).
Zheng, B., De Vries, L. & Gist Farquhar, M. Divergence of RGS proteins: evidence for the existence of six mammalian RGS subfamilies. Trends Biochem. Sci. 24, 411–414 (1999).
De Vries, L., Elenko, E., Hubler, L., Jones, T. L. & Farquhar, M. G. GAIP is membrane-anchored by palmitoylation and interacts with the activated (GTP-bound) form of Gαi subunits. Proc. Natl Acad. Sci. USA 93, 15203–15208 (1996).
Snow, B. E. et al. A G protein γ subunit-like domain shared between RGS11 and other RGS proteins specifies binding to Gβ5 subunits. Proc. Natl Acad. Sci. USA 95, 13307–13312 (1998).
Carman, C. V. et al. Selective regulation of Gα (q/11) by an RGS domain in the G protein-coupled receptor kinase, GRK2. J. Biol. Chem. 274, 34483–34492 (1999).
Zheng, B. et al. RGS-PX1, a GAP for Gαs and sorting nexin in vesicular trafficking. Science 294, 1939–1942 (2001).The authors identify SNX13, a sorting nexin protein, as containing an RGS-box with G · α-specific GAP activity. The authors have renamed SNX13 as RGS-PX1. With this finding, RGS proteins have now been discovered for all subclasses of mammalian Gα subunits.
Wang, L. et al. Cloning and mitochondrial localization of full-length D-AKAP2, a protein kinase A anchoring protein. Proc. Natl Acad. Sci. USA 98, 3220–3225 (2001).
Mishima, K. et al. The scopolamine-induced impairment of spatial cognition parallels the acetylcholine release in the ventral hippocampus in rats. Jpn. J. Pharmacol. 84, 163–173 (2000).
Wu, M., Shanabrough, M., Leranth, C. & Alreja, M. Cholinergic excitation of septohippocampal GABA but not cholinergic neurons: implications for learning and memory. J. Neurosci. 20, 3900–3908 (2000).
Hepler, J. R., 81. Berman, D. M., Gilman, A. G. & Kozasa, T. RGS4 and GAIP are GTPase-activating proteins for Gqα and block activation of phospholipase Cβ by γ-thio-GTP-Gqα. Proc. Natl Acad. Sci. USA 94, 428–432 (1997).
Jordan, J. D., Carey, K. D., Stork, P. J. & Iyengar, R. Modulation of Rap activity by direct interaction of Gα(o) with Rap1 GTPase-activating protein. J. Biol. Chem. 274, 21507–21510 (1999).
Wang, J. et al. RGSZ1, a Gz-selective RGS protein in brain. J. Biol. Chem. 273, 26014–26025 (1998).
Glick, J. L., Meigs, T. E., Miron, A. & Casey, P. J. RGSZ1, a Gz-selective regulator of G protein signalling whose action is sensitive to the phosphorylation state of Gz-alpha. J. Biol. Chem. 273, 26008–26013 (1998).
Berman, D. M., Kozasa, T. & Gilman, A. G. The GTPase-activating protein RGS4 stabilizes the transition state for nucleotide hydrolysis. J. Biol. Chem. 271, 27209–27212 (1996).
Scheschonka, A. et al. RGS3 is a GTPase-activating protein for g(iα) and g(qα) and a potent inhibitor of signalling by GTPase-deficient forms of G(qα) and G(11α). Mol. Pharmacol. 58, 719–728 (2000).
Zhou, J. et al. Characterization of RGS5 in regulation of G protein-coupled receptor signalling. Life Sci. 68, 1457–1469 (2001).
Saitoh, O., Kubo, Y., Miyatani, Y., Asano, T. & Nakata, H. RGS8 accelerates G-protein-mediated modulation of K+ currents. Nature 390, 525–529 (1997).
Druey, K. M. et al. Amino-terminal cysteine residues of RGS16 are required for palmitoylation and modulation of Gi- and Gq-mediated signalling. J. Biol. Chem. 274, 18836–18842 (1999).
Park, I. K. et al. Molecular cloning and characterization of a novel regulator of G-protein signalling from mouse hematopoietic stem cells. J. Biol. Chem. 276, 915–923 (2001).
Posner, B. A., Gilman, A. G. & Harris, B. A. Regulators of G protein signalling 6 and 7. Purification of complexes with Gβ5 and assessment of their effects on G protein-mediated signalling pathways. J. Biol. Chem. 274, 31087–31093 (1999).
Lan, K. L., Zhong, H., Nanamori, M. & Neubig, R. R. Rapid kinetics of regulator of G-protein signalling (RGS)-mediated Gαi and Gαo deactivation. Gα specificity of RGS4 and RGS7. J. Biol. Chem. 275, 33497–33503 (2000).
Hollinger, S., Taylor, J. B., Goldman, E. H. & Hepler, J. R. RGS14 is a bifunctional regulator of Gα(i/o) activity that exists in multiple populations in brain. J. Neurochem. 79, 941–949 (2001).
Fukuhara, S., Chikumi, H. & Gutkind, J. S. Leukemia-associated Rho guanine nucleotide exchange factor (LARG) links heterotrimeric G proteins of the G(12) family to Rho. FEBS Lett. 485, 183–188 (2000).
Grafstein-Dunn, E., Young, K. H., Cockett, M. I. & Khawaja, X. Z. Regional distribution of regulators of G-protein signalling (RGS) 1, 2, 13, 14, 16, and GAIP messenger ribonucleic acids by in situ hybridization in rat brain. Brain Res. Mol. Brain Res. 88, 113–123 (2001).
Kirsch, T., Wellner, M., Luft, F. C., Haller, H. & Lippoldt, A. Altered gene expression in cerebral capillaries of stroke-prone spontaneously hypertensive rats. Brain Res. 910, 106–115 (2001).
Benzing, T. et al. Upregulation of RGS7 may contribute to tumor necrosis factor-induced changes in central nervous function. Nature Med. 5, 913–918 (1999).
Acknowledgements
We would like to thank R. Kimple and H. Zhong for assistance with the figures. R.R.N. acknowledges support from the National Institutes of Health. D.P.S. is a Year 2000 Scholar of The EJLB Foundation, a recipient of the Burroughs–Wellcome Fund New Investigator Award in the Basic Pharmacological Sciences, and acknowledges further grant support from the National Institutes of Health.
Author information
Authors and Affiliations
Related links
Related links
DATABASES
M1 muscarinic acetylcholine receptor
M3 muscarinic acetylcholine receptor
M5 muscarinic acetylcholine receptor
FURTHER INFORMATION
Glossary
- Gα-TRANSDUCIN
-
The G protein in the retina that transduces light signals from the photoreceptor GPCR rhodopsin.
- PDZ DOMAIN
-
(PSD, 95/Dlg and ZO-1/2). Protein–protein interaction domain that binds, in particular, to carboxy-terminal polypeptides.
- PLACE-PREFERENCE CONDITIONING:
-
An often-used behavioural test for the rewarding and/or aversive consequences of drugs by associating these consequences with particular environmental cues.
- SH2 DOMAIN
-
(Src homology domain 2). A domain that interacts with phosphotyrosine-containing polypeptides.
- CHEMORECEPTOR TRIGGERZONE
-
(CTZ). The 'nausea centre' of the brain, which is located within the brainstem in the area postrema. The CTZ is poorly protected by the blood–brain barrier, and is therefore responsive to blood-borne agents perceived as 'toxins'.
Rights and permissions
About this article
Cite this article
Neubig, R., Siderovski, D. Regulators of G-Protein signalling as new central nervous system drug targets. Nat Rev Drug Discov 1, 187–197 (2002). https://doi.org/10.1038/nrd747
Issue Date:
DOI: https://doi.org/10.1038/nrd747
This article is cited by
-
Orthosteric ligand selectivity and allosteric probe dependence at Hydroxycarboxylic acid receptor HCAR2
Signal Transduction and Targeted Therapy (2023)
-
MicroRNA-204-5p reduction in rat hippocampus contributes to stress-induced pathology via targeting RGS12 signaling pathway
Journal of Neuroinflammation (2021)
-
Rgs4 is a regulator of mTOR activity required for motoneuron axon outgrowth and neuronal development in zebrafish
Scientific Reports (2021)
-
Residue-level determinants of RGS R4 subfamily GAP activity and specificity towards the Gi subfamily
Cellular and Molecular Life Sciences (2021)
-
Ligand-triggered de-repression of Arabidopsis heterotrimeric G proteins coupled to immune receptor kinases
Cell Research (2018)