Key Points
-
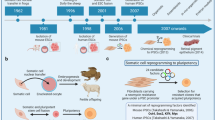
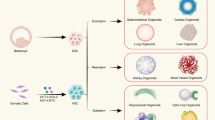
Human pluripotent stem cells (PSCs) can give rise to any cell type in the body. PSC derivatives are being developed for therapeutic use in diseases affecting organs and organ systems such as the eye, nervous system, heart, pancreas, liver, intestines, lung, circulatory system and skin, as well as multi-organ and systemic diseases.
-
Human embryonic stem cells (hESCs) and induced PSCs (iPSCs) are the two main sources of PSCs that are being used for therapeutic endeavours. The destruction of human embryos, which occurs during the hESC derivation process, led to the advent of iPSCs, which are derived from the reprogramming of somatic cells back to an embryonic-like pluripotent state.
-
More than ten clinical trials testing PSC-based therapies are currently being conducted around the world, with eye disease (macular degeneration) being the most common indication. Other indications currently being tested in clinical trials with PSC-based therapies are spinal cord injury, severe heart disease and type 1 diabetes.
-
The next generation of PSC-based clinical trials will probably include a PSC-derived dopaminergic neuron therapy for Parkinson disease, and improvements in differentiation protocols are advancing the development of other PSC-based therapies for conditions such as diabetes, liver disease and skin disorders.
-
Unlike hESCs, iPSCs can be generated in a patient-specific manner, thus enabling autologous and presumably non-immunogenic PSC-based therapies to be developed. iPSCs are also enabling the establishment of 'disease in a dish' cell-based models to study mechanisms of disease pathogenesis and for use in drug screening platforms.
-
iPSCs are being used with gene editing approaches to correct disease-causing genetic mutations in cells derived from patients afflicted with various haematological diseases, among others. The combination of these two technologies is providing the foundation for the development of gene-corrected cell-replacement strategies.
-
Safety is paramount in developing any new PSC-based therapy and regulations are in place to mitigate health and safety risks. However, preliminary data from ongoing first-in-man clinical trials are providing evidence that PSC-therapies can be used safely in humans.
Abstract
Pluripotent stem cells (PSCs) hold great promise for drug discovery and regenerative medicine owing to their ability to differentiate into any cell type in the body. After more than three decades of research, including delays due to the potential tumorigenicity of PSCs and inefficiencies in differentiation methods, the field is at a turning point, with a number of clinical trials across the globe now testing PSC-derived products in humans. Ocular diseases dominate these first-in-man trials, and Phase l/ll results are showing promising safety data as well as possible efficacy. In addition, the advent of induced PSC (iPSC) technology is enabling the development of a wide range of cell-based disease models from genetically predisposed patients, thereby facilitating drug discovery. In this Review, we discuss the recent progress and remaining challenges for the use of PSCs in regenerative medicine and drug development.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Thomson, J. A. et al. Embryonic stem cell lines derived from human blastocysts. Science 282, 1145–1147 (1998). This landmark study was the first to demonstrate the isolation and derivation of PSCs from the inner cell mass of developing human embryos. This method has become the most commonly used approach for the derivation of hESC lines, but it has also created controversy given the destruction of human embryos that occurs during the derivation process.
de Wert, G. & Mummery, C. Human embryonic stem cells: research, ethics and policy. Hum. Reprod. 18, 672–682 (2003).
National Institutes of Health. Federal policy on human embryonic stem cells. NIH Stem Cell Information [online], (2001).
National Institutes of Health. Human embryonic stem cell policy under former President Bush (Aug. 9, 2001–Mar. 9, 2009). NIH Stem Cell Information [online], (2001).
Obama, B. Executive Order 13505 of March 9, 2009: Removing barriers to responsible scientific research invovlving human stem cells. Government Publishing Office [online], (2009).
National Institutes of Health. Research using these lines is eligible for NIH funding. NIH human embryonic stem cell registry [online], (2015).
National Institutes of Health. Submitted hESC lines pending review. NIH human embryonic stem cell registry [online], (2015).
Takahashi, K. & Yamanaka, S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 126, 663–676 (2006).
Takahashi, K. et al. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 131, 861–872 (2007).
Yu, J. et al. Induced pluripotent stem cell lines derived from human somatic cells. Science 318, 1917–1920 (2007). References 9 and 10, both published at the end of 2007, were the first to show that the reprogramming of human somatic cells can give rise to iPSCs.
Nobel Media AB. The Nobel Prize in Physiology or Medicine 2012: John B. Gurdon and Shinya Yamanaka. Nobel Prize [online], (2012).
Warren, L. et al. Highly efficient reprogramming to pluripotency and directed differentiation of human cells with synthetic modified mRNA. Cell Stem Cell 7, 618–630 (2010).
Yu, J. et al. Human induced pluripotent stem cells free of vector and transgene sequences. Science 324, 797–801 (2009).
Okita, K. et al. A more efficient method to generate integration-free human iPS cells. Nat. Methods 8, 409–412 (2011).
Jia, F. et al. A nonviral minicircle vector for deriving human iPS cells. Nat. Methods 7, 197–199 (2010).
Woltjen, K. et al. piggyBac transposition reprograms fibroblasts to induced pluripotent stem cells. Nature 458, 766–770 (2009).
Kim, D. et al. Generation of human induced pluripotent stem cells by direct delivery of reprogramming proteins. Cell Stem Cell 4, 472–476 (2009).
Fusaki, N., Ban, H., Nishiyama, A., Saeki, K. & Hasegawa, M. Efficient induction of transgene-free human pluripotent stem cells using a vector based on Sendai virus, an RNA virus that does not integrate into the host genome. Proc. Jpn Acad. Ser. B Phys. Biol. Sci. 85, 348–362 (2009).
Lu, B. et al. Long-term safety and function of RPE from human embryonic stem cells in preclinical models of macular degeneration. Stem Cells 27, 2126–2135 (2009).
Kamao, H. et al. Characterization of human induced pluripotent stem cell-derived retinal pigment epithelium cell sheets aiming for clinical application. Stem Cell Rep. 2, 205–218 (2014).
Reardon, S. & Cyranoski, D. Japan stem-cell trial stirs envy: researchers elsewhere can't wait to test iPS cells in humans. Nature 513, 278–288 (2014).
Coghlan, A. Mutation alert halts stem-cell trial to cure blindness. New Scientist Daily News [online] (2015).
Schwartz, S. D. et al. Embryonic stem cell trials for macular degeneration: a preliminary report. Lancet 379, 713–720 (2012).
Schwartz, S. D. et al. Human embryonic stem cell-derived retinal pigment epithelium in patients with age-related macular degeneration and Stargardt's macular dystrophy: follow-up of two open-label Phase 1/2 studies. Lancet 385, 7–13 (2015). References 23 and 24 are the first published reports on the safety and tolerability of any PSC-based therapy in humans.
Song, W. K. et al. Treatment of macular degeneration using embryonic stem cell-derived retinal pigment epithelium: preliminary results in Asian patients. Stem Cell Rep. 4, 860–872 (2015).
Buchholz, D. E. et al. Rapid and efficient directed differentiation of human pluripotent stem cells into retinal pigmented epithelium. Stem Cells Translat. Med. 2, 384–393 (2013).
Caras, I. W., Littman, N. & Abo, A. Proceedings: debilitating eye diseases. Stem Cells Translat. Med. 3, 1393–1397 (2014).
Lamba, D. A., Karl, M. O., Ware, C. B. & Reh, T. A. Efficient generation of retinal progenitor cells from human embryonic stem cells. Proc. Natl Acad. Sci. USA 103, 12769–12774 (2006).
Ikeda, H. et al. Generation of Rx+/Pax6+ neural retinal precursors from embryonic stem cells. Proc. Natl Acad. Sci. USA 102, 11331–11336 (2005).
Jagatha, B. et al. In vitro differentiation of retinal ganglion-like cells from embryonic stem cell derived neural progenitors. Biochem. Biophys. Res. Commun. 380, 230–235 (2009).
Riazifar, H., Jia, Y., Chen, J., Lynch, G. & Huang, T. Chemically induced specification of retinal ganglion cells from human embryonic and induced pluripotent stem cells. Stem Cells Translat. Med. 3, 424–432 (2014).
Tanaka, T. et al. Generation of retinal ganglion cells with functional axons from human induced pluripotent stem cells. Scientif. Rep. 5, 8344 (2015).
Tucker, B. A. et al. Duplication of TBK1 stimulates autophagy in iPSC-derived retinal cells from a patient with normal tension glaucoma. J. Stem Cell Res. Ther. 3, 161 (2014).
Fahim, A. T., Daiger, S. P. & Weleber, R. G. Retinitis pigmentosa overview. GeneReviews [online], (2013).
Yoshida, T. et al. The use of induced pluripotent stem cells to reveal pathogenic gene mutations and explore treatments for retinitis pigmentosa. Mol. Brain 7, 45 (2014).
Welsbie, D. S. et al. Functional genomic screening identifies dual leucine zipper kinase as a key mediator of retinal ganglion cell death. Proc. Natl Acad. Sci. USA 110, 4045–4050 (2013).
Zhong, X. et al. Generation of three-dimensional retinal tissue with functional photoreceptors from human iPSCs. Nat. Commun. 5, 4047 (2014).
Ferrer, M. et al. A multiplex high-throughput gene expression assay to simultaneously detect disease and functional markers in induced pluripotent stem cell-derived retinal pigment epithelium. Stem Cells Translat. Med. 3, 911–922 (2014).
Menasche, P. et al. Towards a clinical use of human embryonic stem cell-derived cardiac progenitors: a translational experience. Eur. Heart J. 36, 743–750 (2015).
Bellamy, V. et al. Long-term functional benefits of human embryonic stem cell-derived cardiac progenitors embedded into a fibrin scaffold. J. Heart Lung Transplant. 34, 1198–1207 (2015). This article presents preclinical evidence supporting the efficacy of a PSC-derived cardiac progenitor cell-based therapy, which is now in clinical trials for severe heart disease.
Chong, J. J. et al. Human embryonic-stem-cell-derived cardiomyocytes regenerate non-human primate hearts. Nature 510, 273–277 (2014).
Ye, L. et al. Cardiac repair in a porcine model of acute myocardial infarction with human induced pluripotent stem cell-derived cardiovascular cells. Cell Stem Cell 15, 750–761 (2014).
Navarrete, E. G. et al. Screening drug-induced arrhythmia [corrected] using human induced pluripotent stem cell-derived cardiomyocytes and low-impedance microelectrode arrays. Circulation 128, S3–S13 (2013).
Liang, P. et al. Drug screening using a library of human induced pluripotent stem cell-derived cardiomyocytes reveals disease-specific patterns of cardiotoxicity. Circulation 127, 1677–1691 (2013).
Drawnel, F. M. et al. Disease modeling and phenotypic drug screening for diabetic cardiomyopathy using human induced pluripotent stem cells. Cell Rep. 9, 810–821 (2014).
Kim, C. et al. Studying arrhythmogenic right ventricular dysplasia with patient-specific iPSCs. Nature 494, 105–110 (2013).
Ebert, A. D. et al. Characterization of the molecular mechanisms underlying increased ischemic damage in the aldehyde dehydrogenase 2 genetic polymorphism using a human induced pluripotent stem cell model system. Sci. Transl. Med. 6, 255ra130 (2014).
Aggarwal, P. et al. RNA expression profiling of human iPSC-derived cardiomyocytes in a cardiac hypertrophy model. PloS One 9, e108051 (2014).
Sheng, X. et al. Human pluripotent stem cell-derived cardiomyocytes: response to TTX and lidocain reveals strong cell to cell variability. PloS One 7, e45963 (2012).
Foldes, G. et al. Aberrant α-adrenergic hypertrophic response in cardiomyocytes from human induced pluripotent cells. Stem Cell Rep. 3, 905–914 (2014).
Kelly, O. G. et al. Cell-surface markers for the isolation of pancreatic cell types derived from human embryonic stem cells. Nat. Biotech. 29, 750–756 (2011).
D'Amour, K. A. et al. Production of pancreatic hormone-expressing endocrine cells from human embryonic stem cells. Nat. Biotech. 24, 1392–1401 (2006).
Kroon, E. et al. Pancreatic endoderm derived from human embryonic stem cells generates glucose-responsive insulin-secreting cells in vivo. Nat. Biotech. 26, 443–452 (2008).
Schulz, T. C. et al. A scalable system for production of functional pancreatic progenitors from human embryonic stem cells. PloS One 7, e37004 (2012). References 51–54 describe the development (including the identification, generation, preclinical proof of concept, and scalability) of hESC-derived pancreatic endoderm therapy for type 1 diabetes, developed by Viacyte Inc., which is now in clinical trials.
Pagliuca, F. W. et al. Generation of functional human pancreatic β cells in vitro. Cell 159, 428–439 (2014).
Spence, J. R. et al. Directed differentiation of human pluripotent stem cells into intestinal tissue in vitro. Nature 470, 105–109 (2011).
Watson, C. L. et al. An in vivo model of human small intestine using pluripotent stem cells. Nat. Med. 20, 1310–1314 (2014).
Clayton, D. F. & Darnell, J. E. Jr. Changes in liver-specific compared to common gene transcription during primary culture of mouse hepatocytes. Mol. Cell. Biol. 3, 1552–1561 (1983).
Rambhatla, L., Chiu, C. P., Kundu, P., Peng, Y. & Carpenter, M. K. Generation of hepatocyte-like cells from human embryonic stem cells. Cell Transplant. 12, 1–11 (2003).
Cai, J. et al. Directed differentiation of human embryonic stem cells into functional hepatic cells. Hepatology 45, 1229–1239 (2007).
Agarwal, S., Holton, K. L. & Lanza, R. Efficient differentiation of functional hepatocytes from human embryonic stem cells. Stem Cells 26, 1117–1127 (2008).
Basma, H. et al. Differentiation and transplantation of human embryonic stem cell-derived hepatocytes. Gastroenterology 136, 990–999 (2009).
Takayama, K. et al. Efficient generation of functional hepatocytes from human embryonic stem cells and induced pluripotent stem cells by HNF4α transduction. Mol. Ther. 20, 127–137 (2012).
Song, Z. et al. Efficient generation of hepatocyte-like cells from human induced pluripotent stem cells. Cell Res. 19, 1233–1242 (2009).
Touboul, T., Vallier, L. & Weber, A. Robust differentiation of fetal hepatocytes from human embryonic stem cells and iPS. Med. Sci. (Paris) 26, 1061–1066 (in French) (2010).
Ma, X. et al. Highly efficient differentiation of functional hepatocytes from human induced pluripotent stem cells. Stem Cells Translat. Med. 2, 409–419 (2013).
Gieseck, R. L. 3rd et al. Maturation of induced pluripotent stem cell derived hepatocytes by 3D-culture. PloS One 9, e86372 (2014).
Takebe, T. et al. Vascularized and functional human liver from an iPSC-derived organ bud transplant. Nature 499, 481–484 (2013). This paper describes the generation and engraftment of iPSC-derived liver buds — self-organizing 3D structures that are a stepping stone to the generation of solid organs using iPSCs.
Eggenschwiler, R. et al. Sustained knockdown of a disease-causing gene in patient-specific induced pluripotent stem cells using lentiviral vector-based gene therapy. Stem Cells Translat. Med. 2, 641–654 (2013).
Varughese, A. Abandoning the stem cell clinical trial ship. Biotechniques [online], (2011).
McCormack, K. Treatment for spinal cord injury to start clinical trial funded by California's stem cell agency. California Institute for Regenerative Medicine [online], (2014).
Bjorklund, L. M. Stem cell therapy for Parkinson's disease. Dialogues Clin. Neurosci. 6, 303–311 (2004).
Kim, J. H. et al. Dopamine neurons derived from embryonic stem cells function in an animal model of Parkinson's disease. Nature 418, 50–56 (2002).
Biomics, M. Stem cell crusader offers hope in battle against neurological diseases. Scripps Research Institute [online], (2014).
Kirkeby, A. et al. Generation of regionally specified neural progenitors and functional neurons from human embryonic stem cells under defined conditions. Cell Rep. 1, 703–714 (2012).
Kriks, S. et al. Dopamine neurons derived from human ES cells efficiently engraft in animal models of Parkinson's disease. Nature 480, 547–551 (2011).
Grealish, S. et al. Human ESC-derived dopamine neurons show similar preclinical efficacy and potency to fetal neurons when grafted in a rat model of Parkinson's disease. Cell Stem Cell 15, 653–665 (2014).
Doi, D. et al. Isolation of human induced pluripotent stem cell-derived dopaminergic progenitors by cell sorting for successful transplantation. Stem Cell Rep. 2, 337–350 (2014). References 76–78 speak to the tremendous progress being made in developing PSC-based therapies for Parkinson Disease.
Kim, S. U., Lee, H. J. & Kim, Y. B. Neural stem cell-based treatment for neurodegenerative diseases. Neuropathol. 33, 491–504 (2013).
Takamatsu, K. et al. Degradation of amyloid beta by human induced pluripotent stem cell-derived macrophages expressing Neprilysin-2. Stem Cell Res. 13, 442–453 (2014).
Nizzardo, M. et al. Minimally invasive transplantation of iPSC-derived ALDHhiSSCloVLA4+ neural stem cells effectively improves the phenotype of an amyotrophic lateral sclerosis model. Hum. Mol. Genet. 23, 342–354 (2014).
HD iPSC Consortium. Induced pluripotent stem cells from patients with Huntington's disease show CAG-repeat-expansion-associated phenotypes. Cell Stem Cell 11, 264–278 (2012). Published by a consortium of 8 research institutes, this paper describes the generation of 14 Huntington disease-specific iPSC lines, representing a range of disease phenotypes and backgrounds of patients with Huntington disease. The lines are a shared resource enabling greater consistency and synergy in the research being conducted by participating groups.
Byers, B., Lee, H. L. & Reijo Pera, R. Modeling Parkinson's disease using induced pluripotent stem cells. Curr. Neurol. Neurosci. Rep. 12, 237–242 (2012).
Devine, M. J. et al. Parkinson's disease induced pluripotent stem cells with triplication of the α-synuclein locus. Nat. Commun. 2, 440 (2011).
Nguyen, H. N. et al. LRRK2 mutant iPSC-derived DA neurons demonstrate increased susceptibility to oxidative stress. Cell Stem Cell 8, 267–280 (2011).
Sanchez-Danes, A. et al. Disease-specific phenotypes in dopamine neurons from human iPS-based models of genetic and sporadic Parkinson's disease. EMBO Mol. Med. 4, 380–395 (2012).
Schondorf, D. C. et al. iPSC-derived neurons from GBA1-associated Parkinson's disease patients show autophagic defects and impaired calcium homeostasis. Nat. Commun. 5, 4028 (2014).
Israel, M. A. et al. Probing sporadic and familial Alzheimer's disease using induced pluripotent stem cells. Nature 482, 216–220 (2012).
Yagi, T. et al. Modeling familial Alzheimer's disease with induced pluripotent stem cells. Hum. Mol. Genet. 20, 4530–4539 (2011).
Dimos, J. T. et al. Induced pluripotent stem cells generated from patients with ALS can be differentiated into motor neurons. Science 321, 1218–1221 (2008).
Burkhardt, M. F. et al. A cellular model for sporadic ALS using patient-derived induced pluripotent stem cells. Mol. Cell. Neurosciences 56, 355–364 (2013).
Chen, H. et al. Modeling ALS with iPSCs reveals that mutant SOD1 misregulates neurofilament balance in motor neurons. Cell Stem Cell 14, 796–809 (2014).
Sareen, D. et al. Targeting RNA foci in iPSC-derived motor neurons from ALS patients with a C9ORF72 repeat expansion. Sci. Transl. Med. 5, 208ra149 (2013).
Ebert, A. D. et al. Induced pluripotent stem cells from a spinal muscular atrophy patient. Nature 457, 277–280 (2009).
Chang, T. et al. Brief report: phenotypic rescue of induced pluripotent stem cell-derived motoneurons of a spinal muscular atrophy patient. Stem Cells 29, 2090–2093 (2011).
Garbes, L. et al. VPA response in SMA is suppressed by the fatty acid translocase CD36. Hum. Mol. Genet. 22, 398–407 (2013).
Marchetto, M. C. et al. A model for neural development and treatment of Rett syndrome using human induced pluripotent stem cells. Cell 143, 527–539 (2010).
Marchetto, M. C. et al. Non-cell-autonomous effect of human SOD1 G37R astrocytes on motor neurons derived from human embryonic stem cells. Cell Stem Cell 3, 649–657 (2008).
Egawa, N. et al. Drug screening for ALS using patient-specific induced pluripotent stem cells. Sci. Transl. Med. 5, 145ra104 (2012).
Yang, Y. M. et al. A small molecule screen in stem-cell-derived motor neurons identifies a kinase inhibitor as a candidate therapeutic for ALS. Cell Stem Cell 12, 713–726 (2013).
Yahata, N. et al. Anti-Aβ drug screening platform using human iPS cell-derived neurons for the treatment of Alzheimer's disease. PloS One 6, e25788 (2011).
Liu, Q. et al. Effect of potent γ-secretase modulator in human neurons derived from multiple presenilin 1-induced pluripotent stem cell mutant carriers. JAMA Neurol. 71, 1481–1489 (2014).
Koch, P. et al. Presenilin-1 L166P mutant human pluripotent stem cell-derived neurons exhibit partial loss of γ-secretase activity in endogenous amyloid-β generation. Am. J. Pathol. 180, 2404–2416 (2012).
Lee, G. et al. Large-scale screening using familial dysautonomia induced pluripotent stem cells identifies compounds that rescue IKBKAP expression. Nat. Biotech. 30, 1244–1248 (2012). This is one of the first examples of iPSC derivatives being used in large-scale drug discovery screening which led to the identification of a novel therapeutic target for familial dysautonomia.
Funato, K., Major, T., Lewis, P. W., Allis, C. D. & Tabar, V. Use of human embryonic stem cells to model pediatric gliomas with H3.3K27M histone mutation. Science 346, 1529–1533 (2014).
Olivier, E. N., Qiu, C., Velho, M., Hirsch, R. E. & Bouhassira, E. E. Large-scale production of embryonic red blood cells from human embryonic stem cells. Exp. Hematol. 34, 1635–1642 (2006).
Lu, S. J. et al. Biologic properties and enucleation of red blood cells from human embryonic stem cells. Blood 112, 4475–4484 (2008).
Lapillonne, H. et al. Red blood cell generation from human induced pluripotent stem cells: perspectives for transfusion medicine. Haematologica 95, 1651–1659 (2010).
Dias, J. et al. Generation of red blood cells from human induced pluripotent stem cells. Stem Cells Dev. 20, 1639–1637 (2011).
Takayama, N. et al. Generation of functional platelets from human embryonic stem cells in vitro via ES-sacs, VEGF-promoted structures that concentrate hematopoietic progenitors. Blood 111, 5298–5306 (2008).
Lu, S. J. et al. Platelets generated from human embryonic stem cells are functional in vitro and in the microcirculation of living mice. Cell Res. 21, 530–545 (2011).
Takayama, N. et al. Transient activation of c-MYC expression is critical for efficient platelet generation from human induced pluripotent stem cells. J. Exp. Med. 207, 2817–2830 (2010).
Feng, Q. et al. Scalable generation of universal platelets from human induced pluripotent stem cells. Stem Cell Rep. 3, 817–831 (2014).
Kaufman, R. M., Airo, R., Pollack, S. & Crosby, W. H. Circulating megakaryocytes and platelet release in the lung. Blood 26, 720–731 (1965).
Long, M. W. Megakaryocyte differentiation events. Semin. Hematol. 35, 192–199 (1998).
Thon, J. N. et al. Platelet bioreactor-on-a-chip. Blood 124, 1857–1867 (2014).
Su, Z., Frye, C., Bae, K. M., Kelley, V. & Vieweg, J. Differentiation of human embryonic stem cells into immunostimulatory dendritic cells under feeder-free culture conditions. Clin. Cancer Res. 14, 6207–6217 (2008).
Bandi, S. & Akkina, R. Human embryonic stem cell (hES) derived dendritic cells are functionally normal and are susceptible to HIV-1 infection. AIDS Res. Ther. 5, 1 (2008).
Tseng, S. Y. et al. Generation of immunogenic dendritic cells from human embryonic stem cells without serum and feeder cells. Regenerative Med. 4, 513–526 (2009).
Slukvin, I. I., Vodyanik, M. A., Thomson, J. A., Gumenyuk, M. E. & Choi, K. D. Directed differentiation of human embryonic stem cells into functional dendritic cells through the myeloid pathway. J. Immunol. 176, 2924–2932 (2006).
Timmermans, F. et al. Generation of T cells from human embryonic stem cell-derived hematopoietic zones. J. Immunol. 182, 6879–6888 (2009).
Galic, Z. et al. T lineage differentiation from human embryonic stem cells. Proc. Natl Acad. Sci. USA 103, 11742–11747 (2006).
Woll, P. S., Martin, C. H., Miller, J. S. & Kaufman, D. S. Human embryonic stem cell-derived NK cells acquire functional receptors and cytolytic activity. J. Immunol. 175, 5095–5103 (2005).
Woll, P. S. et al. Human embryonic stem cells differentiate into a homogeneous population of natural killer cells with potent in vivo antitumor activity. Blood 113, 6094–6101 (2009).
Ni, Z. et al. Human pluripotent stem cells produce natural killer cells that mediate anti-HIV-1 activity by utilizing diverse cellular mechanisms. J. Virol. 85, 43–50 (2011).
Vodyanik, M. A., Bork, J. A., Thomson, J. A. & Slukvin, I. I. Human embryonic stem cell-derived CD34+ cells: efficient production in the coculture with OP9 stromal cells and analysis of lymphohematopoietic potential. Blood 105, 617–626 (2005).
Kaufman, D. S., Hanson, E. T., Lewis, R. L., Auerbach, R. & Thomson, J. A. Hematopoietic colony-forming cells derived from human embryonic stem cells. Proc. Natl Acad. Sci. USA 98, 10716–10721 (2001).
Levenberg, S., Golub, J. S., Amit, M., Itskovitz-Eldor, J. & Langer, R. Endothelial cells derived from human embryonic stem cells. Proc. Natl Acad. Sci. USA 99, 4391–4396 (2002).
Wang, Z. Z. et al. Endothelial cells derived from human embryonic stem cells form durable blood vessels in vivo. Nat. Biotech. 25, 317–318 (2007).
Kane, N. M. et al. Derivation of endothelial cells from human embryonic stem cells by directed differentiation: analysis of microRNA and angiogenesis in vitro and in vivo. Arterioscler. Thromb. Vasc. Biol. 30, 1389–1397 (2010).
Nourse, M. B. et al. VEGF induces differentiation of functional endothelium from human embryonic stem cells: implications for tissue engineering. Arterioscler. Thromb. Vasc. Biol. 30, 80–89 (2010).
Ferreira, L. S. et al. Vascular progenitor cells isolated from human embryonic stem cells give rise to endothelial and smooth muscle like cells and form vascular networks in vivo. Circul. Res. 101, 286–294 (2007).
Hill, K. L. et al. Human embryonic stem cell-derived vascular progenitor cells capable of endothelial and smooth muscle cell function. Exp. Hematol. 38, 246–257 (2010).
Oberlin, E., Tavian, M., Blazsek, I. & Peault, B. Blood-forming potential of vascular endothelium in the human embryo. Development 129, 4147–4157 (2002).
Uenishi, G. et al. Tenascin C promotes hematoendothelial development and T lymphoid commitment from human pluripotent stem cells in chemically defined conditions. Stem Cell Rep. 3, 1073–1084 (2014).
French, A., Yang, C. T., Taylor, S., Watt, S. M. & Carpenter, L. Human induced pluripotent stem cell-derived B lymphocytes can express sIgM and can be generated via a hemogenic endothelium intermediate. Stem Cells Dev., http://dx.doi.org/10.1089/scd.2014.0318 (2014).
Xie, F. et al. Seamless gene correction of beta-thalassemia mutations in patient-specific iPSCs using CRISPR/Cas9 and piggyBac. Genome Res. 24, 1526–1533 (2014).
Garcon, L. et al. Ribosomal and hematopoietic defects in induced pluripotent stem cells derived from Diamond Blackfan anemia patients. Blood 122, 912–921 (2013).
Rio, P. et al. Targeted gene therapy and cell reprogramming in Fanconi anemia. EMBO Mol. Med. 6, 835–848 (2014). References 137–139 demonstrate how the combination of iPSC technology and gene editing can correct disease-causing mutations in hematologic disorders, facilitating the study of their underlying pathologies and identifying novel approaches to treating these diseases.
Tebas, P. et al. Gene editing of CCR5 in autologous CD4 T cells of persons infected with HIV. New Engl. J. Med. 370, 901–910 (2014).
Snoeck, H. W. Modeling human lung development and disease using pluripotent stem cells. Development 142, 13–16 (2015).
Wang, D., Haviland, D. L., Burns, A. R., Zsigmond, E. & Wetsel, R. A. A pure population of lung alveolar epithelial type II cells derived from human embryonic stem cells. Proc. Natl Acad. Sci. USA 104, 4449–4454 (2007).
Ghaedi, M. et al. Human iPS cell-derived alveolar epithelium repopulates lung extracellular matrix. J. Clin. Invest. 123, 4950–4962 (2013).
Wong, A. P. et al. Directed differentiation of human pluripotent stem cells into mature airway epithelia expressing functional CFTR protein. Nat. Biotech. 30, 876–882 (2012).
Soh, B. S. et al. CD166(pos) subpopulation from differentiated human ES and iPS cells support repair of acute lung injury. Mol. Ther. 20, 2335–2346 (2012).
Huang, S. X. et al. Efficient generation of lung and airway epithelial cells from human pluripotent stem cells. Nat. Biotech. 32, 84–91 (2014).
Gazdhar, A. et al. The secretome of induced pluripotent stem cells reduces lung fibrosis in part by hepatocyte growth factor. Stem Cell Res. Ther. 5, 123 (2014).
Banerjee, E. R. et al. Human embryonic stem cells differentiated to lung lineage-specific cells ameliorate pulmonary fibrosis in a xenograft transplant mouse model. PloS One 7, e33165 (2012).
Somers, A. et al. Generation of transgene-free lung disease-specific human induced pluripotent stem cells using a single excisable lentiviral stem cell cassette. Stem Cells 28, 1728–1740 (2010).
Green, H., Easley, K. & Iuchi, S. Marker succession during the development of keratinocytes from cultured human embryonic stem cells. Proc. Natl Acad. Sci. USA 100, 15625–15630 (2003).
Kidwai, F. K. et al. Differentiation of human embryonic stem cells into clinically amenable keratinocytes in an autogenic environment. J. Investigative Dermatol. 133, 618–628 (2013).
Petrova, A. et al. 3D in vitro model of a functional epidermal permeability barrier from human embryonic stem cells and induced pluripotent stem cells. Stem Cell Rep. 2, 675–689 (2014).
Sebastiano, V. et al. Human COL7A1-corrected induced pluripotent stem cells for the treatment of recessive dystrophic epidermolysis bullosa. Sci. Transl. Med. 6, 264ra163 (2014). This paper illustrates how the combination of iPSC technology and gene editing can be used to create healthy autologous skin grafts for a debilitating skin-blistering disease.
Umegaki-Arao, N. et al. Induced pluripotent stem cells from human revertant keratinocytes for the treatment of epidermolysis bullosa. Sci. Transl. Med. 6, 264ra164 (2014).
Gnedeva, K. et al. Derivation of hair-inducing cell from human pluripotent stem cells. PloS One 10, e0116892 (2015).
Ding, S. L. & Shen, C. Y. Model of human aging: recent findings on Werner's and Hutchinson–Gilford progeria syndromes. Clin. Intervent. Aging 3, 431–444 (2008).
Nissan, X., Blondel, S. & Peschanski, M. In vitro pathological modelling using patient-specific induced pluripotent stem cells: the case of progeria. Biochem. Soc. Trans. 39, 1775–1779 (2011).
Liu, G. H. et al. Recapitulation of premature ageing with iPSCs from Hutchinson–Gilford progeria syndrome. Nature 472, 221–225 (2011).
Zhang, J. et al. A human iPSC model of Hutchinson Gilford progeria reveals vascular smooth muscle and mesenchymal stem cell defects. Cell Stem Cell 8, 31–45 (2011).
Cheung, H. H. et al. Telomerase protects werner syndrome lineage-specific stem cells from premature aging. Stem Cell Rep. 2, 534–546 (2014).
Nissan, X. et al. Unique preservation of neural cells in Hutchinson–Gilford progeria syndrome is due to the expression of the neural-specific miR-9 microRNA. Cell Rep. 2, 1–9 (2012). This paper shows how the differentiation of iPSCs into various lineages can help to unravel the complexities of systemic diseases. It identified the underlying reasons why a mutated lamin gene in Hutchinson–Gilford progeria syndrome causes premature aging in many different organ systems but spares neurological function.
Blondel, S. et al. Induced pluripotent stem cells reveal functional differences between drugs currently investigated in patients with hutchinson-gilford progeria syndrome. Stem Cells Translat. Med. 3, 510–519 (2014).
Newman, R. E., Yoo, D., LeRoux, M. A. & Danilkovitch-Miagkova, A. Treatment of inflammatory diseases with mesenchymal stem cells. Inflamm. Allergy Drug Targets 8, 110–123 (2009).
Mendicino, M., Bailey, A. M., Wonnacott, K., Puri, R. K. & Bauer, S. R. MSC-based product characterization for clinical trials: an FDA perspective. Cell Stem Cell 14, 141–145 (2014).
Galipeau, J. The mesenchymal stromal cells dilemma — does a negative Phase III trial of random donor mesenchymal stromal cells in steroid-resistant graft-versus-host disease represent a death knell or a bump in the road? Cytotherapy 15, 2–8 (2013).
Wagner, W. et al. Replicative senescence of mesenchymal stem cells: a continuous and organized process. PloS One 3, e2213 (2008).
Maijenburg, M. W., van der Schoot, C. E. & Voermans, C. Mesenchymal stromal cell migration: possibilities to improve cellular therapy. Stem Cells Dev. 21, 19–29 (2012).
Sanchez, L. et al. Enrichment of human ESC-derived multipotent mesenchymal stem cells with immunosuppressive and anti-inflammatory properties capable to protect against experimental inflammatory bowel disease. Stem Cells 29, 251–262 (2011).
Kimbrel, E. A. et al. Mesenchymal stem cell population derived from human pluripotent stem cells displays potent immunomodulatory and therapeutic properties. Stem Cells Dev. 23, 1611–1624 (2014).
Wang, X. et al. Human ESC-derived MSCs outperform bone marrow MSCs in the treatment of an EAE model of multiple sclerosis. Stem Cell Rep. 3, 115–130 (2014).
Zhang, J. et al. Exosomes released from human induced pluripotent stem cells-derived MSCs facilitate cutaneous wound healing by promoting collagen synthesis and angiogenesis. J. Translat. Med. 13, 49 (2015).
Jung, Y., Bauer, G. & Nolta, J. A. Concise review: Induced pluripotent stem cell-derived mesenchymal stem cells: progress toward safe clinical products. Stem Cells 30, 42–47 (2012).
Bailey, A. M. Balancing tissue and tumor formation in regenerative medicine. Sci. Transl. Med. 6, 147fs128 (2012).
Frey-Vasconcells, J., Whittlesey, K. J., Baum, E. & Feigal, E. G. Translation of stem cell research: points to consider in designing preclinical animal studies. Stem Cells Translat. Med. 1, 353–358 (2012).
Carpenter, M. K., Frey-Vasconcells, J. & Rao, M. S. Developing safe therapies from human pluripotent stem cells. Nat. Biotech. 27, 606–613 (2009). This paper discusses key safety points that should be addressed when developing any PSC-based therapy. It also contains links to useful FDA regulatory documents that provide guidance for investigators wishing to advance their therapies to clinical trials.
Rao, M. S. & Malik, N. Assessing iPSC reprogramming methods for their suitability in translational medicine. J. Cell. Biochem. 113, 3061–3068 (2012).
Lowry, W. E. & Quan, W. L. Roadblocks en route to the clinical application of induced pluripotent stem cells. J. Cell Sci. 123, 643–651 (2010).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
E.A.K. and R.L. are employees of Ocata Therapeutics, a clinical stage biotechnology company focused on stem cell-based therapeutics and regenerative ophthalmology.
Related links
FURTHER INFORMATION
Glossary
- Pluripotent stem cells
-
(PSCs). Cells that have the ability to self-renew indefinitely and to differentiate into endoderm, ectoderm and mesoderm, the three germ cell layers that give rise to all cell types in the body.
- Human embryonic stem cells
-
(hESCs). Pluripotent cells that are derived from early stage human embryos.
- Induced pluripotent stem cell
-
(iPSC). A somatic cell that has been induced to become pluripotent by adding factors to the cell to change the state of its nuclear material.
- Reprogramming
-
The process by which a somatic cell is induced to become pluripotent. It involves the addition of specific factors to the cell which alter the state of nuclear material so that it resembles that of a pluripotent embryonic cell rather than that of a differentiated somatic cell.
- Age-related macular degeneration
-
(AMD). A progressive form of vision loss that is associated with aging and is due to the deterioration of photoreceptors and retinal pigment epithelium within the macula, the area of the retina responsible for central vision. There are two forms of AMD: dry and wet. The dry form involves the accumulation of debris between the retina and choroid cell layers and accounts for ~90% of AMD cases. The wet form can arise after the dry form and involves the abnormal growth of blood vessels beneath the retina that can leak fluid. Wet AMD accounts for ~10% of AMD cases.
- Stargardt disease
-
An inherited disorder that results in juvenile onset of macular degeneration and often leads to blindness.
- Retinal pigment epithelium
-
(RPE). A single layer of hexagonal cells at the outermost edge of the retina, anchored to the choroid through the extracellular-matrix-containing Bruch's membrane. The RPE keeps the photoreceptor layer healthy by providing nutrients, secreting growth factors, exchanging ions and phagocytosing old photoreceptor parts.
- Investigational new drug
-
(IND). A designation used to describe a drug that has permission from the US Food and Drug Administration (FDA) to be shipped across state lines, thus allowing it to be tested in human clinical trials. IND applications are reviewed by the FDA to ensure that testing of the drug in humans does not pose excessive risk to the patient.
- Pancreatic endoderm
-
Multipotent progenitor cells that can give rise to various cell types within the pancreas, including glucose-sensing, insulin-producing β-cells, the mature cell type that is destroyed by a person's own immune system in type 1 diabetes.
- α1 antitrypsin
-
A protease inhibitor that is produced in the liver and secreted into the circulation. It protects tissue from being digested by enzymes such as neutrophil elastase and is crucial in maintaining lung function. Deficiency of α1 antitrypsin, a genetic disorder characterized by critically low levels of circulating wild type α1 antitrypsin and deposition of abnormal α1 antitrypsin in the liver, can lead to liver cirrhosis as well as emphysema and chronic obstructive pulmonary disease.
- Mesenchymal stem cells
-
(MSCs). A type of multipotent adult stem cell found in the stroma, or connective tissue, of a variety of different organs. In addition to multipotent differentiation capabilities, they have immunomodulatory properties that may be beneficial in treating autoimmune, degenerative and inflammatory disorders.
- Dopaminergic neuron
-
A neuron that is responsible for the synthesis and release of the neurotransmitter dopamine. Through dopamine release, dopaminergic neurons in the middle part of the brain are responsible for regulating neuromuscular motor control and reward-driven behaviour and are the cell population that deteriorates in Parkinson disease.
- Gene editing
-
The alteration of a specific genetic sequence, often done to correct disease-causing mutations. Approaches include the use of site-specific nucleases (for example, zinc finger, TALEN and CRISPR–Cas9 nucleases) to cut DNA at a precise location and the use of viruses that stimulate endogenous homologous recombination, thereby allowing the sequence of a gene to be altered or repaired.
- Multipotent
-
The ability to differentiate into several different cell types (but not all cell types of the body, as is the case for pluripotent cells). Multipotency is more limited in scope than pluripotency and often used to describe adult or tissue-specific stem cells or progenitors.
- Adult stem cell
-
A rare somatic cell that resides in different tissues of the body and can self-renew. Adult stem cells can also repopulate or replenish mature cells in the local tissue or organ environment through the process of differentiation. Examples include hematopoietic stem cells, intestinal stem cells, neural stem cells and testicular stem cells.
Rights and permissions
About this article
Cite this article
Kimbrel, E., Lanza, R. Current status of pluripotent stem cells: moving the first therapies to the clinic. Nat Rev Drug Discov 14, 681–692 (2015). https://doi.org/10.1038/nrd4738
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrd4738
This article is cited by
-
LNP-RNA-engineered adipose stem cells for accelerated diabetic wound healing
Nature Communications (2024)
-
Long noncoding RNA ZBTB40-IT1 regulates bone mass by directing the differentiation of human bone marrow mesenchymal stromal cells via the microRNA-514a-3p/FOXO4 axis
Human Cell (2022)
-
Cell-derived extracellular vesicles and membranes for tissue repair
Journal of Nanobiotechnology (2021)