Key Points
-
Tumours actively inhibit the antitumour immune response.
-
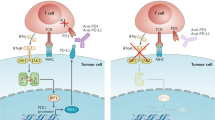
The programmed cell death protein 1–programmed cell death 1 ligand 1 (PD1–PDL1) pathway has a natural role in regulating peripheral tolerance and restraining over-exuberant immune responses.
-
PDL1 is a critical dominant immunoinhibitor in many tumour types, leading to immune evasion by the tumour. PD1 pathway blockade unleashes a previously exhausted immune response that is focused on tumour neoantigens.
-
Immunotherapies that increase the antitumour immune response, such as interleukin-2, or blockade of cytotoxic T lymphocyte antigen 4 (CTLA4), PD1 or PDL1 can benefit a moderate number of patients with cancer, with a durable clinical benefit in some.
-
Many patients fail to develop tumour shrinkage when blocking only one immune checkpoint, and increasing response rates is a major and achievable goal.
-
Given the tolerability and efficacy of PD1 pathway blockade in clinical trials it is a good foundation for combination therapies seeking to increase response rates.
-
Once PD1 or CTLA4 are blocked, many other therapies can augment their efficacy, including blockade of other immunoinhibitory pathways, stimulation by activating pathways such as tumour necrosis factor receptor superfamily members, some chemotherapies, radiation, epigenetic modifiers, targeted therapies, angiogenesis blockade, augmentation of natural killer cell activity, chimeric antigen receptor T cell therapies, and vaccines.
-
Therapeutics may work by blocking immunoinhibitory targets or stimulating immunoactivating targets not only on lymphocytes but also on macrophage, natural killer and stromal cells to overcome failure to respond to single checkpoint blockade.
Abstract
Targeting immune checkpoints such as programmed cell death protein 1 (PD1), programmed cell death 1 ligand 1 (PDL1) and cytotoxic T lymphocyte antigen 4 (CTLA4) has achieved noteworthy benefit in multiple cancers by blocking immunoinhibitory signals and enabling patients to produce an effective antitumour response. Inhibitors of CTLA4, PD1 or PDL1 administered as single agents have resulted in durable tumour regression in some patients, and combinations of PD1 and CTLA4 inhibitors may enhance antitumour benefit. Numerous additional immunomodulatory pathways as well as inhibitory factors expressed or secreted by myeloid and stromal cells in the tumour microenvironment are potential targets for synergizing with immune checkpoint blockade. Given the breadth of potential targets in the immune system, critical questions to address include which combinations should move forward in development and which patients will benefit from these treatments. This Review discusses the leading drug targets that are expressed on tumour cells and in the tumour microenvironment that allow enhancement of the antitumour immune response.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Schreiber, R. D., Old, L. J. & Smyth, M. J. Cancer immunoediting: integrating immunity's roles in cancer suppression and promotion. Science 331, 1565–1570 (2011).
Fritsch, E. F. et al. HLA-binding properties of tumor neoepitopes in humans. Cancer Immunol. Res. 2, 522–529 (2014).
Gubin, M. M. et al. Checkpoint blockade cancer immunotherapy targets tumour-specific mutant antigens. Nature 515, 577–581 (2014). Identifies tumour-specific mutant proteins (neoantigens) as the major T cell rejection targets following anti-PD1 and/or anti-CTLA4 tumour immunotherapy in mice. Vaccines incorporating these neoantigens can induce tumour rejection.
Freeman, G. J. et al. Engagement of the PD-1 immunoinhibitory receptor by a novel B7 family member leads to negative regulation of lymphocyte activation. J. Exp. Med. 192, 1027–1034 (2000). Shows that PDL1 is a ligand for PD1 and inhibits T cell activation. It also shows that PDL1 expression is upregulated by IFN γ.
Blackburn, S. D. et al. Coregulation of CD8+ T cell exhaustion by multiple inhibitory receptors during chronic viral infection. Nat. Immunol. 10, 29–37 (2009). Shows that expression of multiple inhibitory receptors in addition to PD1 is associated with greater T cell exhaustion in a chronic viral infection model.
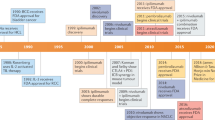
Hodi, F. S. et al. Improved survival with ipilimumab in patients with metastatic melanoma. N. Engl. J. Med. 363, 711–723 (2010). A report of the Phase III trial showing a survival advantage with CTLA4-specific mAb treatment in advanced melanoma; the results supported FDA approval of the first immune checkpoint inhibitor.
Powles, T. et al. MPDL3280A (anti-PD-L1) treatment leads to clinical activity in metastatic bladder cancer. Nature 515, 558–562 (2014). Although not the first published report of efficacy of therapeutically targeting PDL1, this study demonstrates a 43% response rate in metastatic bladder cancer, a good safety profile and a threefold increase in response rates in tumours that express PDL1 on the tumour-infiltrating immune cells.
Hamid, O. et al. Safety and tumor responses with lambrolizumab (anti-PD-1) in melanoma. N. Engl. J. Med. 369, 134–144 (2013).
Brahmer, J. R. et al. Phase I study of single-agent anti-programmed death-1 (MDX-1106) in refractory solid tumors: safety, clinical activity, pharmacodynamics, and immunologic correlates. J. Clin. Oncol. 28, 3167–3175 (2010). This first-in-human trial of a PD1 blocker shows that the therapy is well tolerated and produces a partial response in a patient with melanoma and in a patient with renal cell carcinoma, and a complete response in a patient with colon adenocarcinoma.
Herbst, R. S. et al. Predictive correlates of response to the anti-PD-L1 antibody MPDL3280A in cancer patients. Nature 515, 563–567 (2014).
Garon, E. B. et al. Pembrolizumab for the treatment of non-small-cell lung cancer. N. Engl. J. Med. 372, 2018–2028 (2015).
Le, D. T. et al. PD-1 blockade in tumors with mismatch-repair deficiency. N. Engl. J. Med. http://dx.doi.org/10.1056/nejmoa1500596 (2015).
Robert, C. et al. Pembrolizumab versus ipilimumab in advanced melanoma. N. Engl. J. Med. http://dx.doi.org/10.1056/nejmoa1503093 (2015).
Larkin, J. et al. Combined nivolumab and ipilimumab or monotherapy in untreated melanoma. N. Engl. J. Med. http://dx.doi.org/10.1056/nejmoa1504030 (2015).
Schadendorf, D. et al. Pooled analysis of long-term survival data from Phase II and Phase III trials of ipilimumab in unresectable or metastatic melanoma. J. Clin. Oncol. 33, 1889–1894 (2015). Pooled data from multiple clinical trials of ipilimumab with follow-up for up to 10 years (4,846 patients) demonstrate that the survival curve begins to plateau in year three, indicating durable ongoing responses.
Wolchok, J. D. et al. Nivolumab plus ipilimumab in advanced melanoma. N. Engl. J. Med. 369, 122–133 (2013). The first Phase I trial of combined CTLA4 and PD1 blockade demonstrates that this combination is highly effective, but highly toxic.
Postow, M. A. et al. Nivolumab and ipilimumab versus ipilimumab in untreated melanoma. N. Engl. J. Med. 372, 2006–2017 (2015). The first randomized Phase II trial to compare the efficacy and toxicity of combination versus monotherapy with immune checkpoint inhibitors. It reports 61% response rates with nivolumab plus ipilimumab versus 11% with ipilimumab alone; however, 54% of patients on the combination had grade 3 or 4 adverse events.
Jenkins, M. K. & Schwartz, R. H. Antigen presentation by chemically modified splenocytes induces antigen-specific T cell unresponsiveness in vitro and in vivo. J. Exp. Med. 165, 302–319 (1987).
Suntharalingam, G. et al. Cytokine storm in a phase 1 trial of the anti-CD28 monoclonal antibody TGN1412. N. Engl. J. Med. 355, 1018–1028 (2006).
Antonia, S. J. et al. Nivolumab (anti-PD-1; BMS-936558, ONO-4538) and ipilimumab in first-line NSCLC: interim phase I results. J. Clin. Oncol. 32 (Suppl.), 8023 (2014).
Antonia, S. J. et al. Nivolumab (anti-PD-1; BMS-936558, ONO-4538) in combination with platinum-based doublet chemotherapy (PT-DC) in advanced non-small cell lung cancer (NSCLC). J. Clin. Oncol. 32 (Suppl.), 8113 (2014).
Amin, A. et al. Nivolumab (anti-PD-1; BMS-936558, ONO-4538) in combination with sunitinib or pazopanib in patients (pts) with metastatic renal cell carcinoma (mRCC). J. Clin. Oncol. 32 (Suppl.), 5010 (2014).
Hammers, H. et al. Phase I study of nivolumab in combination with ipilimumab in metastatic renal cell carcinoma (mRCC). J. Clin. Oncol. 32 (Suppl.), 4504 (2014).
Hamid, O. S. et al. Clinical activity, safety, and biomarkers of MPDL3280A, an engineered PD-L1 antibody in patients with locally advanced or metastatic melanoma (mM). J. Clin. Oncol. 31 (Suppl.), 9010 (2013).
Postow, M. A. et al. Immunologic correlates of the abscopal effect in a patient with melanoma. N. Engl. J. Med. 366, 925–931 (2012).
Antonia, S. et al. Phase Ib study of MEDI4736, a programmed cell death ligand-1 (PD-L1) antibody, in combination with tremelimumab, a cytotoxic T-lymphocyte-associated protein-4 (CTLA-4) antibody, in patients (pts) with advanced NSCLC. J. Clin. Oncol. 33 (Suppl.), 3014 (2015).
Parry, R. V. et al. CTLA-4 and PD-1 receptors inhibit T-cell activation by distinct mechanisms. Mol. Cell. Biol. 25, 9543–9553 (2005).
Korman, A. et al. Activity of anti-PD-1 in murine tumor models: role of “host” PD-L1 and synergistic effect of anti-PD-1 and anti-CTLA-4. J. Immunol. 178 (Suppl.), S82 (2007). First report to demonstrate the synergistic effect of blocking both PD1 and CTLA4 in mouse tumour models; the results formed the basis of the development of this combination in patients.
Freeman, G. J. & Sharpe, A. H. A new therapeutic strategy for malaria: targeting T cell exhaustion. Nat. Immunol. 13, 113–115 (2012).
Robert, C. et al. Nivolumab in previously untreated melanoma without BRAF mutation. N. Engl. J. Med. 372, 320–330 (2015).
Dovedi, S. J. et al. Acquired resistance to fractionated radiotherapy can be overcome by concurrent PD-L1 blockade. Cancer Res. 74, 5458–5468 (2014).
Keir, M. E., Butte, M. J., Freeman, G. J. & Sharpe, A. H. PD-1 and its ligands in tolerance and immunity. Annu. Rev. Immunol. 26, 677–704 (2008).
Postow, M. A. et al. Pharmacodynamic effect of ipilimumab on absolute lymphocyte count (ALC) and association with overall survival in patients with advanced melanoma. J. Clin. Oncol. 31 (Suppl.), 9052 (2013).
Le, D. T. & Jaffee, E. M. Regulatory T-cell modulation using cyclophosphamide in vaccine approaches: a current perspective. Cancer Res. 72, 3439–3444 (2012).
Vincent, J. et al. 5-Fluorouracil selectively kills tumor-associated myeloid-derived suppressor cells resulting in enhanced T cell-dependent antitumor immunity. Cancer Res. 70, 3052–3061 (2010).
Sevko, A. et al. Antitumor effect of paclitaxel is mediated by inhibition of myeloid-derived suppressor cells and chronic inflammation in the spontaneous melanoma model. J. Immunol. 190, 2464–2471 (2013).
Hannani, D. et al. Prerequisites for the antitumor vaccine-like effect of chemotherapy and radiotherapy. Cancer J. 17, 351–358 (2011).
Kim, K. et al. Eradication of metastatic mouse cancers resistant to immune checkpoint blockade by suppression of myeloid-derived cells. Proc. Natl Acad. Sci. USA 111, 11774–11779 (2014).
Papa, M. Z., Vetto, J. T., Ettinghausen, S. E., Mule, J. J. & Rosenberg, S. A. Effect of corticosteroid on the antitumor activity of lymphokine-activated killer cells and interleukin 2 in mice. Cancer Res. 46, 5618–5623 (1986).
Mier, J. W. et al. Inhibition of interleukin-2-induced tumor necrosis factor release by dexamethasone: prevention of an acquired neutrophil chemotaxis defect and differential suppression of interleukin-2-associated side effects. Blood 76, 1933–1940 (1990).
Vetto, J. T., Papa, M. Z., Lotze, M. T., Chang, A. E. & Rosenberg, S. A. Reduction of toxicity of interleukin-2 and lymphokine-activated killer cells in humans by the administration of corticosteroids. J. Clin. Oncol. 5, 496–503 (1987).
Wolchok, J. et al. Efficacy and safety results from a Phase III trial of nivolumab (NIVO) alone or combined with ipilimumab (IPI) versus IPI alone in treatment-naive patients (pts) with advanced melanoma (MEL) (CheckMate 067). J. Clin. Oncol. 33 (Suppl.), LBA1 (2015).
Donia, M. et al. BRAF inhibition improves tumor recognition by the immune system: potential implications for combinatorial therapies against melanoma involving adoptive T-cell transfer. Oncoimmunology 1, 1476–1483 (2012).
Frederick, D. T. et al. BRAF inhibition is associated with enhanced melanoma antigen expression and a more favorable tumor microenvironment in patients with metastatic melanoma. Clin. Cancer Res. 19, 1225–1231 (2013).
Jiang, X., Zhou, J., Giobbie-Hurder, A., Wargo, J. & Hodi, F. S. The activation of MAPK in melanoma cells resistant to BRAF inhibition promotes PD-L1 expression that is reversible by MEK and PI3K inhibition. Clin. Cancer Res. 19, 598–609 (2013).
Cooper, Z. A. et al. Response to BRAF inhibition in melanoma is enhanced when combined with immune checkpoint blockade. Cancer Immunol. Res. 2, 643–654 (2014).
Ribas, A., Hodi, F. S., Callahan, M., Konto, C. & Wolchok, J. Hepatotoxicity with combination of vemurafenib and ipilimumab. N. Engl. J. Med. 368, 1365–1366 (2013).
Ribas, A. et al. Phase I study combining anti-PD-L1 (MEDI4736) with BRAF (dabrafenib) and/or MEK (trametinib) inhibitors in advanced melanoma. J. Clin. Oncol. 33 (Suppl.), 3003 (2015).
Talmadge, J. E. & Gabrilovich, D. I. History of myeloid-derived suppressor cells. Nat. Rev. Cancer 13, 739–752 (2013).
Finke, J. et al. MDSC as a mechanism of tumor escape from sunitinib mediated anti-angiogenic therapy. Int. Immunopharmacol. 11, 856–861 (2011).
Bhatt, R. S. et al. Renal cancer resistance to antiangiogenic therapy is delayed by restoration of angiostatic signaling. Mol. Cancer Ther. 9, 2793–2802 (2010).
Salven, P., Orpana, A., Teerenhovi, L. & Joensuu, H. Simultaneous elevation in the serum concentrations of the angiogenic growth factors VEGF and bFGF is an independent predictor of poor prognosis in non-Hodgkin lymphoma: a single-institution study of 200 patients. Blood 96, 3712–3718 (2000).
Zhan, P., Qian, Q. & Yu, L. K. Serum VEGF level is associated with the outcome of patients with hepatocellular carcinoma: a meta-analysis. Hepatobiliary Surg. Nutr. 2, 209–215 (2013).
Yuan, J. et al. Pretreatment serum VEGF is associated with clinical response and overall survival in advanced melanoma patients treated with ipilimumab. Cancer Immunol. Res. 2, 127–132 (2014).
Ohm, J. E. & Carbone, D. P. VEGF as a mediator of tumor-associated immunodeficiency. Immunol. Res. 23, 263–272 (2001).
Gabrilovich, D. I., Ishida, T., Nadaf, S., Ohm, J. E. & Carbone, D. P. Antibodies to vascular endothelial growth factor enhance the efficacy of cancer immunotherapy by improving endogenous dendritic cell function. Clin. Cancer Res. 5, 2963–2970 (1999).
Lieu, C. et al. Safety and efficacy of MPDL3280A (anti-PDL1) in combination with bevacizumab (bev) and/or chemotherapy (chemo) in patients (pts) with locally advanced or metastatic solid tumors. Ann. Oncol. 25 (Suppl. 4): iv361–iv372 (2014). This is the first report of the combination of a PDL1-specific mAb and a VEGF-specific mAb (bevacizumab) in patients, which was highly tolerable in patients.
Ellis, L. M. The role of neuropilins in cancer. Mol. Cancer Ther. 5, 1099–1107 (2006).
Chaudhary, B., Khaled, Y. S., Ammori, B. J. & Elkord, E. Neuropilin 1: function and therapeutic potential in cancer. Cancer Immunol. Immunother. 63, 81–99 (2014).
Papatriantafyllou, M. T cells: neuropilin 1 — distinguishing TReg cell subsets. Nat. Rev. Immunol. 12, 746 (2012).
Grandclement, C. & Borg, C. Neuropilins: a new target for cancer therapy. Cancers (Basel) 3, 1899–1928 (2011).
Weekes, C. D. et al. A Phase I study of the human monoclonal anti-NRP1 antibody MNRP1685A in patients with advanced solid tumors. Invest. New Drugs 32, 653–660 (2014).
Patnaik, A. et al. Phase Ib study evaluating MNRP1685A, a fully human anti-NRP1 monoclonal antibody, in combination with bevacizumab and paclitaxel in patients with advanced solid tumors. Cancer Chemother. Pharmacol. 73, 951–960 (2014).
Delgoffe, G. M. et al. Stability and function of regulatory T cells is maintained by a neuropilin-1-semaphorin-4a axis. Nature 501, 252–256 (2013).
Vilcek, J. & Feldmann, M. Historical review: cytokines as therapeutics and targets of therapeutics. Trends Pharmacol. Sci. 25, 201–209 (2004).
Schaer, D. A., Hirschhorn-Cymerman, D. & Wolchok, J. D. Targeting tumor-necrosis factor receptor pathways for tumor immunotherapy. J. Immunother. Cancer 2, 7 (2014).
Kwon, B. S. et al. Immune responses in 4-1BB (CD137)-deficient mice. J. Immunol. 168, 5483–5490 (2002).
Chester, C. et al. Biomarker characterization using mass cytometry in a phase 1 trial of urelumab (BMS-663513) in subjects with advanced solid tumors and relapsed/refractory B-cell non-Hodgkin lymphoma. J. Clin. Oncol. 32 (Suppl.), 3017 (2014).
Hwu, W. Targeted therapy for metastatic melanoma: from bench to bedside. HemOnc Today [online], (2010).
Segal, N. H. et al. A phase 1 study of PF-05082566 (anti-4-1BB) in patients with advanced cancer. J. Clin. Oncol. 32 (Suppl.), 3007 (2014).
Gopal, A. et al. A phase I study of PF-05082566 (anti-4-1BB) + rituximab in patients with CD20+ NHL. J. Clin. Oncol. 33 (Suppl.), 3004 (2015).
Kohrt, H. E. et al. Targeting CD137 enhances the efficacy of cetuximab. J. Clin. Invest. 124, 2668–2682 (2014).
Ye, Q. et al. CD137 accurately identifies and enriches for naturally occurring tumor-reactive T cells in tumor. Clin. Cancer Res. 20, 44–55 (2014). Demonstrates that in the tumour microenvironment, tumour-reactive T cells express the TNFR 4-1BB, thus bolstering the hypothesis that 4-1BB-specific agonist antibodies will have a synergistic effect with other immune checkpoint therapeutics.
Bulliard, Y. et al. OX40 engagement depletes intratumoral Tregs via activating FcγRs, leading to antitumor efficacy. Immunol. Cell Biol. 92, 475–480 (2014).
Guo, Z. et al. PD-1 blockade and OX40 triggering synergistically protects against tumor growth in a murine model of ovarian cancer. PLoS ONE 9, e89350 (2014).
Guo, Z. et al. Combined TIM-3 blockade and CD137 activation affords the long-term protection in a murine model of ovarian cancer. J. Transl. Med. 11, 215 (2013).
Adler, A. J. & Vella, A. T. Betting on improved cancer immunotherapy by doubling down on CD134 and CD137 co-stimulation. Oncoimmunology 2, e22837 (2013).
Wei, H. et al. Combinatorial PD-1 blockade and CD137 activation has therapeutic efficacy in murine cancer models and synergizes with cisplatin. PLoS ONE 8, e84927 (2013).
Kovacsovics-Bankowski, M. et al. Phase I/II clinical trial of anti-OX40, radiation and cyclophosphamide in patients with prostate cancer: immunological analysis. J. Immunother. Cancer 1, 255 (2013).
Curti, B. D. et al. OX40 is a potent immune-stimulating target in late-stage cancer patients. Cancer Res. 73, 7189–7198 (2013).
Cohen, A. D. et al. Agonist anti-GITR monoclonal antibody induces melanoma tumor immunity in mice by altering regulatory T cell stability and intra-tumor accumulation. PLoS ONE 5, e10436 (2010).
Lu, L. et al. Combined PD-1 blockade and GITR triggering induce a potent antitumor immunity in murine cancer models and synergizes with chemotherapeutic drugs. J. Transl. Med. 12, 36 (2014).
Grewal, I. S. CD70 as a therapeutic target in human malignancies. Expert Opin. Ther. Targets 12, 341–351 (2008).
Ansell, S. M. et al. Phase I evaluation of an agonist anti-CD27 human antibody (CDX-1127) in patients with advanced hematologic malignancies. J. Clin. Oncol. 32 (Suppl.), 3034 (2014).
Infante, J. R. et al. Immunologic activity of an activating anti-CD27 antibody (CDX-1127) in patients (pts) with solid tumors. J. Clin. Oncol. 32 (Suppl.), 3027 (2014).
Twohig, J. P. et al. The death receptor 3/TL1A pathway is essential for efficient development of antiviral CD4+ and CD8+ T-cell immunity. FASEB J. 26, 3575–3586 (2012).
Bossen, C. et al. Interactions of tumor necrosis factor (TNF) and TNF receptor family members in the mouse and human. J. Biol. Chem. 281, 13964–13971 (2006).
Meylan, F. et al. The TNF-family receptor DR3 is essential for diverse T cell-mediated inflammatory diseases. Immunity 29, 79–89 (2008).
Slebioda, T. J. et al. Triggering of TNFRSF25 promotes CD8+ T-cell responses and anti-tumor immunity. Eur. J. Immunol. 41, 2606–2611 (2011).
Schreiber, T. H. & Podack, E. R. Immunobiology of TNFSF15 and TNFRSF25. Immunol. Res. 57, 3–11 (2013).
Schreiber, T. H., Wolf, D., Bodero, M., Gonzalez, L. & Podack, E. R. T cell costimulation by TNFR superfamily (TNFRSF)4 and TNFRSF25 in the context of vaccination. J. Immunol. 189, 3311–3318 (2012).
Bodmer, J. L., Schneider, P. & Tschopp, J. The molecular architecture of the TNF superfamily. Trends Biochem. Sci. 27, 19–26 (2002).
Bajor, D. et al. Combination of agonistic CD40 monoclonal antibody CP-870,893 and anti-CTLA-4 antibody tremelimumab in patients with metastatic melanoma. American Association for Cancer Research Annual Meeting [online], (2015).
Zippelius, A., Schreiner, J., Herzig, P. & Muller, P. Induced PD-L1 expression mediates acquired resistance to agonistic anti-CD40 treatment. Cancer Immunol. Res. 3, 236–244 (2015).
Medina-Echeverz, J. et al. Systemic agonistic anti-CD40 treatment of tumor-bearing mice modulates hepatic myeloid-suppressive cells and causes immune-mediated liver damage. Cancer Immunol. Res. 3, 557–566 (2015).
Cai, G. et al. CD160 inhibits activation of human CD4+ T cells through interaction with herpesvirus entry mediator. Nat. Immunol. 9, 176–185 (2008).
Derre, L. et al. BTLA mediates inhibition of human tumor-specific CD8+ T cells that can be partially reversed by vaccination. J. Clin. Invest. 120, 157–167 (2010).
Pasero, C. & Olive, D. Interfering with coinhibitory molecules: BTLA/HVEM as new targets to enhance anti-tumor immunity. Immunol. Lett. 151, 71–75 (2013).
Watanabe, N. et al. BTLA is a lymphocyte inhibitory receptor with similarities to CTLA-4 and PD-1. Nat. Immunol. 4, 670–679 (2003).
Huang, C. T. et al. Role of LAG-3 in regulatory T cells. Immunity 21, 503–513 (2004).
Woo, S. R. et al. Immune inhibitory molecules LAG-3 and PD-1 synergistically regulate T-cell function to promote tumoral immune escape. Cancer Res. 72, 917–927 (2012). The combination of LAG3 and PD1 blockade in a mouse tumour model is more effective than either alone, prompting the development of this combination in an ongoing clinical trial.
Matsuzaki, J. et al. Tumor-infiltrating NY-ESO-1-specific CD8+ T cells are negatively regulated by LAG-3 and PD-1 in human ovarian cancer. Proc. Natl Acad. Sci. USA 107, 7875–7880 (2010).
Wang-Gillam, A. et al. A Phase I study of IMP321 and gemcitabine as the front-line therapy in patients with advanced pancreatic adenocarcinoma. Invest. New Drugs 31, 707–713 (2013).
Brignone, C. et al. First-line chemoimmunotherapy in metastatic breast carcinoma: combination of paclitaxel and IMP321 (LAG-3Ig) enhances immune responses and antitumor activity. J. Transl. Med. 8, 71 (2010).
Brignone, C., Escudier, B., Grygar, C., Marcu, M. & Triebel, F. A phase I pharmacokinetic and biological correlative study of IMP321, a novel MHC class II agonist, in patients with advanced renal cell carcinoma. Clin. Cancer Res. 15, 6225–6231 (2009).
McIntire, J. J. et al. Identification of Tapr (an airway hyperreactivity regulatory locus) and the linked Tim gene family. Nat. Immunol. 2, 1109–1116 (2001).
Freeman, G. J., Casasnovas, J. M., Umetsu, D. T. & DeKruyff, R. H. TIM genes: a family of cell surface phosphatidylserine receptors that regulate innate and adaptive immunity. Immunol. Rev. 235, 172–189 (2010).
Jin, H. T. et al. Cooperation of Tim-3 and PD-1 in CD8 T-cell exhaustion during chronic viral infection. Proc. Natl Acad. Sci. USA 107, 14733–14738 (2010).
Sakuishi, K. et al. Targeting Tim-3 and PD-1 pathways to reverse T cell exhaustion and restore anti-tumor immunity. J. Exp. Med. 207, 2187–2194 (2010). Promising results from the combination of TIM3 and PD1 blockade in mouse tumours support the development of this combination in patients.
Zhu, C. et al. The Tim-3 ligand galectin-9 negatively regulates T helper type 1 immunity. Nat. Immunol. 6, 1245–1252 (2005).
Huang, Y. H. et al. CEACAM1 regulates TIM-3-mediated tolerance and exhaustion. Nature 517, 386–390 (2015).
DeKruyff, R. H. et al. T cell/transmembrane, Ig, and mucin-3 allelic variants differentially recognize phosphatidylserine and mediate phagocytosis of apoptotic cells. J. Immunol. 184, 1918–1930 (2010).
Sakuishi, K., Jayaraman, P., Behar, S. M., Anderson, A. C. & Kuchroo, V. K. Emerging Tim-3 functions in antimicrobial and tumor immunity. Trends Immunol. 32, 345–349 (2011).
Madireddi, S. et al. Galectin-9 controls the therapeutic activity of 4-1BB-targeting antibodies. J. Exp. Med. 211, 1433–1448 (2014).
Gleason, M. K. et al. Tim-3 is an inducible human natural killer cell receptor that enhances interferon γ production in response to galectin-9. Blood 119, 3064–3072 (2012).
Pillai, S., Netravali, I. A., Cariappa, A. & Mattoo, H. Siglecs and immune regulation. Annu. Rev. Immunol. 30, 357–392 (2012).
Jandus, C. et al. Interactions between Siglec-7/9 receptors and ligands influence NK cell-dependent tumor immunosurveillance. J. Clin. Invest. 124, 1810–1820 (2014).
Laubli, H. et al. Engagement of myelomonocytic Siglecs by tumor-associated ligands modulates the innate immune response to cancer. Proc. Natl Acad. Sci. USA 111, 14211–14216 (2014).
Takamiya, R., Ohtsubo, K., Takamatsu, S., Taniguchi, N. & Angata, T. The interaction between Siglec-15 and tumor-associated sialyl-Tn antigen enhances TGF-β secretion from monocytes/macrophages through the DAP12-Syk pathway. Glycobiology 23, 178–187 (2013).
Pardoll, D. M. The blockade of immune checkpoints in cancer immunotherapy. Nat. Rev. Cancer 12, 252–264 (2012).
Chen, L. & Flies, D. B. Molecular mechanisms of T cell co-stimulation and co-inhibition. Nat. Rev. Immunol. 13, 227–242 (2013).
Zhao, R. et al. HHLA2 is a member of the B7 family and inhibits human CD4 and CD8 T-cell function. Proc. Natl Acad. Sci. USA 110, 9879–9884 (2013).
Sharpe, A. H. Mechanisms of costimulation. Immunol. Rev. 229, 5–11 (2009).
Capece, D., Verzella, D., Fischietti, M., Zazzeroni, F. & Alesse, E. Targeting costimulatory molecules to improve antitumor immunity. J. Biomed. Biotechnol. 2012, 926321 (2012).
Ng Tang, D. et al. Increased frequency of ICOS+ CD4 T cells as a pharmacodynamic biomarker for anti-CTLA-4 therapy. Cancer Immunol. Res. 1, 229–234 (2013).
Fan, X., Quezada, S. A., Sepulveda, M. A., Sharma, P. & Allison, J. P. Engagement of the ICOS pathway markedly enhances efficacy of CTLA-4 blockade in cancer immunotherapy. J. Exp. Med. 211, 715–725 (2014).
Yap, E. H., Rosche, T., Almo, S. & Fiser, A. Functional clustering of immunoglobulin superfamily proteins with protein–protein interaction information calibrated hidden Markov model sequence profiles. J. Mol. Biol. 426, 945–961 (2014).
Le Mercier, I. et al. VISTA regulates the development of protective antitumor immunity. Cancer Res. 74, 1933–1944 (2014).
Janakiram, M. et al. Expression, clinical significance, and receptor identification of the newest B7 family member HHLA2 protein. Clin. Cancer Res. 21, 2359–2366 (2015).
Zhu, Y. et al. B7-H5 costimulates human T cells via CD28H. Nat. Commun. 4, 2043 (2013).
Nguyen, T., Liu, X. K., Zhang, Y. & Dong, C. BTNL2, a butyrophilin-like molecule that functions to inhibit T cell activation. J. Immunol. 176, 7354–7360 (2006).
Valentonyte, R. et al. Sarcoidosis is associated with a truncating splice site mutation in BTNL2. Nat. Genet. 37, 357–364 (2005).
Arnett, H. A. & Viney, J. L. Immune modulation by butyrophilins. Nat. Rev. Immunol. 14, 559–569 (2014).
Fitzgerald, L. M. et al. Germline missense variants in the BTNL2 gene are associated with prostate cancer susceptibility. Cancer Epidemiol. Biomarkers Prev. 22, 1520–1528 (2013).
Vijai, J. et al. A genome-wide association study suggests evidence of variants at 6p21.32 associated with marginal zone lymphoma. Cancer Res. 74 (Suppl. 19), 5071 (2014).
Abeler-Dorner, L., Swamy, M., Williams, G., Hayday, A. C. & Bas, A. Butyrophilins: an emerging family of immune regulators. Trends Immunol. 33, 34–41 (2012).
Rubinstein, R., Ramagopal, U. A., Nathenson, S. G., Almo, S. C. & Fiser, A. Functional classification of immune regulatory proteins. Structure 21, 766–776 (2013).
de Andrade, L. F., Smyth, M. J. & Martinet, L. DNAM-1 control of natural killer cells functions through nectin and nectin-like proteins. Immunol. Cell Biol. 92, 237–244 (2014).
Sema Kurtulus, S. et al. Mechanisms of TIGIT-driven immune suppression in cancer. J. Immunother. Cancer 2 (Suppl. 3), O13 (2014).
Chauvin, J. M. et al. TIGIT and PD-1 impair tumor antigen-specific CD8+ T cells in melanoma patients. J. Clin. Invest. 125, 2046–2058 (2015).
Johnston, R. J. et al. The immunoreceptor TIGIT regulates antitumor and antiviral CD8+ T cell effector function. Cancer Cell 26, 923–937 (2014). TIGIT is identified as an inhibitory receptor mediating exhaustion on CD8+ T cells; the combination of TIGIT and PDL1 blockade is more effective than either alone in preclinical models.
Guillerey, C. et al. Immunosurveillance and therapy of multiple myeloma are CD226 dependent. J. Clin. Invest. 125, 2077–2089 (2015).
Li, A. H. et al. Analysis of loss-of-function variants and 20 risk factor phenotypes in 8,554 individuals identifies loci influencing chronic disease. Nat. Genet. 47, 640–642 (2015).
Beano, A. et al. Correlation between NK function and response to trastuzumab in metastatic breast cancer patients. J. Transl. Med. 6, 25 (2008). Elucidates the critical role of natural killer cells in mediating the activity of trastuzumab in patients with breast cancer.
Vey, N. et al. A Phase 1 trial of the anti-inhibitory KIR mAb IPH2101 for AML in complete remission. Blood 120, 4317–4323 (2012).
Raulet, D. H. Roles of the NKG2D immunoreceptor and its ligands. Nat. Rev. Immunol. 3, 781–790 (2003).
Spear, P., Wu, M. R., Sentman, M. L. & Sentman, C. L. NKG2D ligands as therapeutic targets. Cancer Immun. 13, 8 (2013).
Farnault, L., Sanchez, C., Baier, C., Le Treut, T. & Costello, R. T. Hematological malignancies escape from NK cell innate immune surveillance: mechanisms and therapeutic implications. Clin. Dev. Immunol. 2012, 421702 (2012).
Bonnafous, C. et al. Targeting MICA with therapeutic antibodies for the treatment of cancer (Poster). J. Immunother. Cancer 1 (Suppl. 1), P41 (2013).
Wada, H., Matsumoto, N., Maenaka, K., Suzuki, K. & Yamamoto, K. The inhibitory NK cell receptor CD94/NKG2A and the activating receptor CD94/NKG2C bind the top of HLA-E through mostly shared but partly distinct sets of HLA-E residues. Eur. J. Immunol. 34, 81–90 (2004).
Chretien, A. S. et al. Cancer-induced alterations of NK-mediated target recognition: current and investigational pharmacological strategies aiming at restoring NK-mediated anti-tumor activity. Front. Immunol. 5, 122 (2014).
Sheu, J. & Shih, le-M. HLA-G and immune evasion in cancer cells. J. Formos Med. Assoc. 109, 248–257 (2010).
Alegre, E. et al. Some basic aspects of HLA-G biology. J. Immunol. Res. 2014, 657625 (2014).
González, A. et al. The immunosuppressive molecule HLA-G and its clinical implications. Crit. Rev. Clin. Lab Sci. 49, 63–84 (2012).
Stark, S. & Watzl, C. 2B4 (CD244), NTB-A and CRACC (CS1) stimulate cytotoxicity but no proliferation in human NK cells. Int. Immunol. 18, 241–247 (2006).
Valiante, N. M. & Trinchieri, G. Identification of a novel signal transduction surface molecule on human cytotoxic lymphocytes. J. Exp. Med. 178, 1397–1406 (1993).
Ljunggren, H. G. & Malmberg, K. J. Prospects for the use of NK cells in immunotherapy of human cancer. Nat. Rev. Immunol. 7, 329–339 (2007).
Munn, D. H. Blocking IDO activity to enhance anti-tumor immunity. Front. Biosci. (Elite Ed) 4, 734–745 (2012). This review highlights immune suppression mediated by IDO in both the tumour and its draining lymph nodes.
Gibney, G. T. et al. Preliminary results from a phase 1/2 study of INCB024360 combined with ipilimumab (ipi) in patients (pts) with melanoma. J. Clin. Oncol. 32 (Suppl.), 3010 (2014).
Soliman, H. H. et al. A first in man phase I trial of the oral immunomodulator, indoximod, combined with docetaxel in patients with metastatic solid tumors. Oncotarget 5, 8136–8146 (2014).
[No authors listed.] NewLink Genetics announces exclusive worldwide licensing agreement for development of NLG919, an IDO inhibitor in Phase 1, and research collaboration for the discovery of next generation IDO/TDO inhibitors. NewLink Genetics [online], (2014).
[No authors listed.] Bristol-Myers Squibb to expand its immuno-oncology pipeline with agreement to acquire Flexus Biosciences, Inc. Bristol-Myers Squibb [online], (2015).
Margadant, C. & Sonnenberg, A. Integrin-TGF-β crosstalk in fibrosis, cancer and wound healing. EMBO Rep. 11, 97–105 (2010).
Gueorguieva, I. et al. Defining a therapeutic window for the novel TGF-β inhibitor LY2157299 monohydrate based on a pharmacokinetic/pharmacodynamic model. Br. J. Clin. Pharmacol. 77, 796–807 (2014).
Bhola, N. E. et al. TGF-β inhibition enhances chemotherapy action against triple-negative breast cancer. J. Clin. Invest. 123, 1348–1358 (2013). Shows that TGFß-dependent signals underlie resistance to chemotherapy via multiple mechanisms, including blunting the antitumour immune response.
Bedi, A. et al. Inhibition of TGF-β enhances the in vivo antitumor efficacy of EGF receptor-targeted therapy. Mol. Cancer Ther. 11, 2429–2439 (2012).
Hanks, B. A., Holtzhausen, A., Evans, K., Heid, M. & Blobe, G. C. Combinatorial TGF-β signaling blockade and anti-CTLA-4 antibody immunotherapy in a murine BRAFV600E-PTEN−/− transgenic model of melanoma. J. Clin. Oncol. 32 (Suppl.), 3011 (2014).
Wilson, W. R. & Hay, M. P. Targeting hypoxia in cancer therapy. Nat. Rev. Cancer 11, 393–410 (2011).
Ohta, A. et al. A2A adenosine receptor protects tumors from antitumor T cells. Proc. Natl Acad. Sci. USA 103, 13132–13137 (2006).
Cekic, C. & Linden, J. Adenosine A2A receptors intrinsically regulate CD8+ T cells in the tumor microenvironment. Cancer Res. 74, 7239–7249 (2014).
Jones, G. B. & Yuan, G. Towards next generation adenosine A2A receptor antagonists. Curr. Med. Chem. 21, 3918–3935 (2014).
Yegutkin, G. G. Nucleotide- and nucleoside-converting ectoenzymes: important modulators of purinergic signalling cascade. Biochim. Biophys. Acta 1783, 673–694 (2008).
Mandapathil, M. et al. Generation and accumulation of immunosuppressive adenosine by human CD4+CD25highFOXP3+ regulatory T cells. J. Biol. Chem. 285, 7176–7186 (2010).
Iannone, R., Miele, L., Maiolino, P., Pinto, A. & Morello, S. Adenosine limits the therapeutic effectiveness of anti-CTLA4 mAb in a mouse melanoma model. Am. J. Cancer Res. 4, 172–181 (2014).
Allard, B., Pommey, S., Smyth, M. J. & Stagg, J. Targeting CD73 enhances the antitumor activity of anti-PD-1 and anti-CTLA-4 mAbs. Clin. Cancer Res. 19, 5626–5635 (2013). Shows that blocking CD73 improves the effect of both PD1 and CTLA4 blockade, and preferentially synergizes with PD1 blockade because activation of the adenosine receptor enhances PD1 expression.
Hatfield, S. M. et al. Immunological mechanisms of the antitumor effects of supplemental oxygenation. Sci. Transl. Med. 7, 277ra30 (2015).
Cekic, C., Day, Y. J., Sag, D. & Linden, J. Myeloid expression of adenosine A2A receptor suppresses T and NK cell responses in the solid tumor microenvironment. Cancer Res. 74, 7250–7259 (2014).
Morello, S. & Miele, L. Targeting the adenosine A2b receptor in the tumor microenvironment overcomes local immunosuppression by myeloid-derived suppressor cells. OncoImmunology 3, e27989 (2014).
Hoskin, D. W., Mader, J. S., Furlong, S. J., Conrad, D. M. & Blay, J. Inhibition of T cell and natural killer cell function by adenosine and its contribution to immune evasion by tumor cells (Review). Int. J. Oncol. 32, 527–535 (2008).
Srivastava, M. K. et al. Myeloid suppressor cell depletion augments antitumor activity in lung cancer. PLoS ONE 7, e40677 (2012).
Feig, C. et al. Targeting CXCL12 from FAP-expressing carcinoma-associated fibroblasts synergizes with anti-PD-L1 immunotherapy in pancreatic cancer. Proc. Natl Acad. Sci. USA 110, 20212–20217 (2013).
Luster, T. A. et al. Plasma protein β-2-glycoprotein 1 mediates interaction between the anti-tumor monoclonal antibody 3G4 and anionic phospholipids on endothelial cells. J. Biol. Chem. 281, 29863–29871 (2006).
Shtivelband, M. et al. Randomized, blinded, placebo-controlled phase II trial of docetaxel and bavituximab as second-line therapy in locally advanced or metastatic non-squamous non-small cell lung cancer. J. Clin. Oncol. 31 (Suppl.), 8095 (2013).
Yopp, A. et al. Antibody-mediated blockade of phosphatidylserine enhances the antitumor activity of targeted therapy and immune checkpoint inhibitors by affecting myeloid and lymphocyte populations in the tumor microenvironment. J. Immunother. Cancer 2 (Suppl. 3), P266 (2014). Blocking phosphotidylserine, a ligand of the co-inhibitory receptor TIM3, improves the efficacy of PD1 and CTLA4 tumour immunotherapy.
Huang, X. et al. Antibody-mediated phosphatidylserine blockade significantly enhances the efficacy of downstream immune checkpoint inhibition in K1735 mouse melanoma. J. Immunother. Cancer 2 (Suppl. 3), P205 (2014).
Garon, E. B. et al. Ramucirumab plus docetaxel versus placebo plus docetaxel for second-line treatment of stage IV non-small-cell lung cancer after disease progression on platinum-based therapy (REVEL): a multicentre, double-blind, randomised phase 3 trial. Lancet 384, 665–673 (2014).
Tseng, D. et al. Anti-CD47 antibody-mediated phagocytosis of cancer by macrophages primes an effective antitumor T-cell response. Proc. Natl Acad. Sci. USA 110, 11103–11108 (2013). Targeting CD47 increases phagocytosis, enhances antitumour responses and synergizes with many antitumour mAbs in vivo.
Weiskopf, K. et al. Engineered SIRPα variants as immunotherapeutic adjuvants to anticancer antibodies. Science 341, 88–91 (2013).
Jaiswal, S. et al. CD47 is upregulated on circulating hematopoietic stem cells and leukemia cells to avoid phagocytosis. Cell 138, 271–285 (2009).
Chao, M. P. et al. Anti-CD47 antibody synergizes with rituximab to promote phagocytosis and eradicate non-Hodgkin lymphoma. Cell 142, 699–713 (2010).
Melero, I. et al. Therapeutic vaccines for cancer: an overview of clinical trials. Nat. Rev. Clin. Oncol. 11, 509–524 (2014).
Taube, J. M. et al. Colocalization of inflammatory response with B7-h1 expression in human melanocytic lesions supports an adaptive resistance mechanism of immune escape. Sci. Transl. Med. 4, 127ra37 (2012). Describes the feedback loop whereby tumours dominantly evade the immune response by expressing PDL1 in response to IFN γ produced by activated T cells at the lymphocyte-rich tumour margin.
Jass, J. R. Lymphocytic infiltration and survival in rectal cancer. J. Clin. Pathol. 39, 585–589 (1986).
Sobin, L. & Wittekind, C. TNM Classification of Malignant Tumors 2nd edn (Wiley, 2002).
Mlecnik, B., Bindea, G., Pages, F. & Galon, J. Tumor immunosurveillance in human cancers. Cancer Metastasis Rev. 30, 5–12 (2011).
Galon, J. et al. Cancer classification using the Immunoscore: a worldwide task force. J. Transl. Med. 10, 205 (2012).
Angell, H. & Galon, J. From the immune contexture to the Immunoscore: the role of prognostic and predictive immune markers in cancer. Curr. Opin. Immunol. 25, 261–267 (2013).
Messina, J. L. et al. 12-chemokine gene signature identifies lymph node-like structures in melanoma: potential for patient selection for immunotherapy? Sci. Rep. 2, 765 (2012).
Thompson, R. H. et al. Costimulatory B7-H1 in renal cell carcinoma patients: indicator of tumor aggressiveness and potential therapeutic target. Proc. Natl Acad. Sci. USA 101, 17174–17179 (2004). The first article to show that PDL1 expression on tumour cells and on infiltrating lymphocytes in kidney cancer is associated with worse prognosis and increased cancer-specific death.
Giraldo, N. A. et al. Orchestration and prognostic significance of immune checkpoints in the microenvironment of primary and metastatic renal cell cancer. Clin. Cancer Res. http://dx.doi.org/10.1158/1078-0432.CCR-14-2926 (2015).
Tumeh, P. C. et al. PD-1 blockade induces responses by inhibiting adaptive immune resistance. Nature 515, 568–571 (2014). Shows that tumours that have high CD8 infiltrate and PDL1 expression on the tumour and at the tumour margin are more likely to respond to PD1 immunotherapy.
Porter, D. L., Levine, B. L., Kalos, M., Bagg, A. & June, C. H. Chimeric antigen receptor-modified T cells in chronic lymphoid leukemia. N. Engl. J.Med. 365, 725–733 (2011).
John, L. B. et al. Anti-PD-1 antibody therapy potently enhances the eradication of established tumors by gene-modified T cells. Clin. Cancer Res. 19, 5636–5646 (2013). Shows that chimeric antigen receptor T cells can express PD1, be susceptible to PD1-mediated inhibition of T cell activation, and that PD1 blockade can enhance the activity of chimeric antigen receptor T cells in mouse models.
Wolchok, J. D. et al. Guidelines for the evaluation of immune therapy activity in solid tumors: immune-related response criteria. Clin. Cancer Res. 15, 7412–7420 (2009).
Nordlund, J. J. et al. Vitiligo in patients with metastatic melanoma: a good prognostic sign. J. Am. Acad. Dermatol. 9, 689–696 (1983).
Bystryn, J. C., Rigel, D., Friedman, R. J. & Kopf, A. Prognostic significance of hypopigmentation in malignant melanoma. Arch. Dermatol. 123, 1053–1055 (1987).
Scalzo, S. et al. Primary hypothyroidism associated with interleukin-2 and interferon α-2 therapy of melanoma and renal carcinoma. Eur. J. Cancer 26, 1152–1156 (1990).
Becker, J. C., Winkler, B., Klingert, S. & Brocker, E. B. Antiphospholipid syndrome associated with immunotherapy for patients with melanoma. Cancer 73, 1621–1624 (1994).
Rosenberg, S. A. & White, D. E. Vitiligo in patients with melanoma: normal tissue antigens can be targets for cancer immunotherapy. J. Immunother. Emphasis Tumor Immunol. 19, 81–84 (1996).
Atkins, M. B. et al. Hypothyroidism after treatment with interleukin-2 and lymphokine-activated killer cells. N. Engl. J. Med. 318, 1557–1563 (1988).
Attia, P. et al. Autoimmunity correlates with tumor regression in patients with metastatic melanoma treated with anti-cytotoxic T-lymphocyte antigen-4. J. Clin. Oncol. 23, 6043–6053 (2005).
Chapman, P. B. et al. Improved survival with vemurafenib in melanoma with BRAF V600E mutation. N. Engl. J. Med. 364, 2507–2516 (2011).
Beck, K. E. et al. Enterocolitis in patients with cancer after antibody blockade of cytotoxic T-lymphocyte-associated antigen 4. J. Clin. Oncol. 24, 2283–2289 (2006).
Kyi, C., Carvajal, R. D., Wolchok, J. D. & Postow, M. A. Ipilimumab in patients with melanoma and autoimmune disease. J. Immunother. Cancer 2, 35 (2014).
Hurwitz, A. A. et al. Combination immunotherapy of primary prostate cancer in a transgenic mouse model using CTLA-4 blockade. Cancer Res. 60, 2444–2448 (2000).
Hodi, F. S. et al. Ipilimumab plus sargramostim versus ipilimumab alone for treatment of metastatic melanoma: a randomized clinical trial. JAMA 312, 1744–1753 (2014). Treatment with ipilimumab plus sargramostim (a granulocyte–macrophage colony-stimulating factor) versus ipilimumab alone results in lower toxicity and modestly better overall survival.
Jinushi, M. et al. MFG-E8-mediated uptake of apoptotic cells by APCs links the pro- and antiinflammatory activities of GM-CSF. J. Clin. Invest. 117, 1902–1913 (2007).
Rini, B. I. et al. Phase 1 dose-escalation trial of tremelimumab plus sunitinib in patients with metastatic renal cell carcinoma. Cancer 117, 758–767 (2011).
Maker, A. V. et al. Tumor regression and autoimmunity in patients treated with cytotoxic T lymphocyte-associated antigen 4 blockade and interleukin 2: a phase I/II study. Ann. Surg. Oncol. 12, 1005–1016 (2005).
Robert, C. et al. Ipilimumab plus dacarbazine for previously untreated metastatic melanoma. N. Engl. J. Med. 364, 2517–2526 (2011).
Stewart, R. et al. The role of Fc gamma receptors in the activity of immunomodulatory antibodies for cancer. J. Immunother. Cancer 2, 29 (2014).
Gomez-Roca, C. A. et al. Phase I study of RG7155, a novel anti-CSF1R antibody, in patients with advanced/metastatic solid tumors. J. Clin. Oncol. 33 (Suppl.; abstr.), 3005 (2015).
Simpson, T. R. et al. Fc-dependent depletion of tumor-infiltrating regulatory T cells co-defines the efficacy of anti-CTLA-4 therapy against melanoma. J. Exp. Med. 210, 1695–1710 (2013).
Selby, M. J. et al. Anti-CTLA-4 antibodies of IgG2a isotype enhance antitumor activity through reduction of intratumoral regulatory T cells. Cancer Immunol. Res. 1, 32–42 (2013). Shows that an ADCC-activating Fc region on the CTLA4-specific mAb is essential for antitumour activity in a mouse tumour model, suggesting that the depletion of CTLA4-positive T Reg cells within the tumour is a major mechanism.
Mahoney, K. M. & Atkins, M. B. Prognostic and predictive markers for the new immunotherapies. Oncology (Williston Park) 28 (Suppl. 3), 39–48 (2014). An overview of the differences between prognostic and predictive markers as well as the candidate predictive and pharmacodynamic markers in the field of immune checkpoint blockade, focusing on PDL1 expression.
Green, M. R. et al. Integrative analysis reveals selective 9p24.1 amplification, increased PD-1 ligand expression, and further induction via JAK2 in nodular sclerosing Hodgkin lymphoma and primary mediastinal large B-cell lymphoma. Blood 116, 3268–3277 (2010). This paper shows that genomic amplification of 9p24.1 encoding Janus kinase 2, PDL1 and PDL2 results in PDL1 and PDL2 overexpression in Hodgkin and primary mediastinal lymphomas.
Ansell, S. M. et al. PD-1 blockade with nivolumab in relapsed or refractory Hodgkin's lymphoma. N. Engl. J. Med. 372, 311–319 (2015).
Taube, J. M. et al. Association of PD-1, PD-1 ligands, and other features of the tumor immune microenvironment with response to anti-PD-1 therapy. Clin. Cancer Res. 20, 5064–5074 (2014).
Paz-Ares, L. et al. Phase III, randomized trial (CheckMate 057) of nivolumab (NIVO) versus docetaxel (DOC) in advanced non-squamous cell (non-SQ) non-small cell lung cancer (NSCLC). J. Clin. Oncol. 33 (Suppl.), LBA109 (2015).
Brahmer, J. et al. Nivolumab versus docetaxel in advanced squamous-cell non-small-cell lung cancer. N. Engl. J. Med. http://dx.doi.org/10.1056/NEJMoa1504627 (2015).
Schellens, J. et al. CEA-targeted engineered IL2: clinical confirmation of tumor targeting and evidence of intra-tumoral immune activation. J. Clin. Oncol. 33 (Suppl.), 3016 (2015).
Sznol, M. & Chen, L. Antagonist antibodies to PD-1 and B7-H1 (PD-L1) in the treatment of advanced human cancer-response. Clin. Cancer Res. 19, 5542 (2013).
Grosso, J. et al. Programmed death-ligand 1 (PD-L1) expression in various tumor types. J. Immunother. Cancer 1 (Suppl 1), P53 (2013).
Brown, J. A. et al. Blockade of programmed death-1 ligands on dendritic cells enhances T cell activation and cytokine production. J. Immunol. 170, 1257–1266 (2003). Shows that enhanced T cell activation is a consequence of blockade of the interaction with PD1, not signal transduction by PDL1 or PDL2. The paper also presents data on PDL1 expression in a broad range of solid and haematological malignancies and normal tissues.
Dong, H. et al. Tumor-associated B7-H1 promotes T-cell apoptosis: a potential mechanism of immune evasion. Nat. Med. 8, 793–800 (2002). The first paper to show PDL1 expression in situ on solid tumours, including lung, ovary, colon and melanoma.
Ohigashi, Y. et al. Clinical significance of programmed death-1 ligand-1 and programmed death-1 ligand-2 expression in human esophageal cancer. Clin. Cancer Res. 11, 2947–2953 (2005).
Nomi, T. et al. Clinical significance and therapeutic potential of the programmed death-1 ligand/programmed death-1 pathway in human pancreatic cancer. Clin. Cancer Res. 13, 2151–2157 (2007).
Hamanishi, J. et al. Programmed cell death 1 ligand 1 and tumor-infiltrating CD8+ T lymphocytes are prognostic factors of human ovarian cancer. Proc. Natl Acad. Sci. USA 104, 3360–3365 (2007).
Gao, Q. et al. Overexpression of PD-L1 significantly associates with tumor aggressiveness and postoperative recurrence in human hepatocellular carcinoma. Clin. Cancer Res. 15, 971–979 (2009).
Latchman, Y. et al. PD-L2 is a second ligand for PD-1 and inhibits T cell activation. Nat. Immunol. 2, 261–268 (2001). Identifies PDL2 as a second ligand for PD1, recruitment of SHP2 as a mechanism for PD1-mediated inhibition of T cell activation, and first shows PDL1 expression on solid tumour cell lines.
Shi, M. et al. Expression of programmed cell death 1 ligand 2 (PD-L2) is a distinguishing feature of primary mediastinal (thymic) large B-cell lymphoma and associated with PDCD1LG2 copy gain. Am. J. Surg. Pathol. 38, 1715–1723 (2014).
Sun, Y. et al. B7-H3 and B7-H4 expression in non-small-cell lung cancer. Lung Cancer 53, 143–151 (2006).
Wu, C. P. et al. Relationship between co-stimulatory molecule B7-H3 expression and gastric carcinoma histology and prognosis. World J. Gastroenterol. 12, 457–459 (2006).
Crispen, P. L. et al. Tumor cell and tumor vasculature expression of B7-H3 predict survival in clear cell renal cell carcinoma. Clin. Cancer Res. 14, 5150–5157 (2008).
Loos, M. et al. Expression of the costimulatory molecule B7-H3 is associated with prolonged survival in human pancreatic cancer. BMC Cancer 9, 463 (2009).
Sun, J. et al. Clinical significance and regulation of the costimulatory molecule B7-H3 in human colorectal carcinoma. Cancer Immunol. Immunother. 59, 1163–1171 (2010).
Zang, X. et al. Tumor associated endothelial expression of B7-H3 predicts survival in ovarian carcinomas. Mod. Pathol. 23, 1104–1112 (2010).
Roth, T. J. et al. B7-H3 ligand expression by prostate cancer: a novel marker of prognosis and potential target for therapy. Cancer Res. 67, 7893–7900 (2007).
Boorjian, S. A. et al. T-cell coregulatory molecule expression in urothelial cell carcinoma: clinicopathologic correlations and association with survival. Clin. Cancer Res. 14, 4800–4808 (2008).
Tringler, B. et al. B7-H4 is highly expressed in ductal and lobular breast cancer. Clin. Cancer Res. 11, 1842–1848 (2005). Although the receptor for B7-H4 is unknown, this inhibitory B7 family member is highly expressed in breast cancer, suggesting a mechanism of immune evasion and encouraging drug development.
Krambeck, A. E. et al. B7-H4 expression in renal cell carcinoma and tumor vasculature: associations with cancer progression and survival. Proc. Natl Acad. Sci. USA 103, 10391–10396 (2006).
Kryczek, I. et al. B7-H4 expression identifies a novel suppressive macrophage population in human ovarian carcinoma. J. Exp. Med. 203, 871–881 (2006).
Simon, I. et al. B7-H4 is a novel membrane-bound protein and a candidate serum and tissue biomarker for ovarian cancer. Cancer Res. 66, 1570–1575 (2006).
Tringler, B. et al. B7-H4 overexpression in ovarian tumors. Gynecol. Oncol. 100, 44–52 (2006).
Chen, L. J. et al. B7-H4 expression associates with cancer progression and predicts patient's survival in human esophageal squamous cell carcinoma. Cancer Immunol. Immunother. 60, 1047–1055 (2011).
Arigami, T. et al. Clinical significance of the B7-H4 coregulatory molecule as a novel prognostic marker in gastric cancer. World J. Surg. 35, 2051–2057 (2011).
Jiang, J. et al. Tumor expression of B7-H4 predicts poor survival of patients suffering from gastric cancer. Cancer Immunol. Immunother. 59, 1707–1714 (2010).
Wang, Y. et al. α 1 antichymotrypsin is aberrantly expressed during melanoma progression and predicts poor survival for patients with metastatic melanoma. Pigment Cell. Melanoma Res. 23, 575–578 (2010).
Awadallah, N. S. et al. Detection of B7-H4 and p53 in pancreatic cancer: potential role as a cytological diagnostic adjunct. Pancreas 36, 200–206 (2008).
Quandt, D., Fiedler, E., Boettcher, D., Marsch, W. & Seliger, B. B7-H4 expression in human melanoma: its association with patients' survival and antitumor immune response. Clin. Cancer Res. 17, 3100–3111 (2011).
Schulkens, I. A. et al. Galectin expression profiling identifies galectin-1 and galectin-9δ5 as prognostic factors in stage I/II non-small cell lung cancer. PLoS ONE 9, e107988 (2014).
Lahm, H. et al. Comprehensive galectin fingerprinting in a panel of 61 human tumor cell lines by RT-PCR and its implications for diagnostic and therapeutic procedures. J. Cancer Res. Clin. Oncol. 127, 375–386 (2001).
Lotan, R. et al. Lactose-binding lectin expression in human colorectal carcinomas. Relation to tumor progression. Carbohydr. Res. 213, 47–57 (1991).
Miyazaki, J. et al. Increased expression of galectin-3 in primary gastric cancer and the metastatic lymph nodes. Oncol. Rep. 9, 1307–1312 (2002).
Pallesen, G. & Hamilton-Dutoit, S. J. Ki-1 (CD30) antigen is regularly expressed by tumor cells of embryonal carcinoma. Am. J. Pathol. 133, 446–450 (1988).
Jilaveanu, L. B. et al. CD70 expression patterns in renal cell carcinoma. Hum. Pathol. 43, 1394–1399 (2012).
Yang, Z. Z., Novak, A. J., Ziesmer, S. C., Witzig, T. E. & Ansell, S. M. CD70+ non-Hodgkin lymphoma B cells induce Foxp3 expression and regulatory function in intratumoral CD4+CD25 T cells. Blood 110, 2537–2544 (2007).
Schreiner, B. et al. Expression of the B7-related molecule ICOSL by human glioma cells in vitro and in vivo. Glia 44, 296–301 (2003).
Martin-Orozco, N. et al. Melanoma cells express ICOS ligand to promote the activation and expansion of T-regulatory cells. Cancer Res. 70, 9581–9590 (2010).
Sloan, K. E. et al. CD155/PVR plays a key role in cell motility during tumor cell invasion and migration. BMC Cancer 4, 73 (2004).
Acknowledgements
G.J.F. acknowledges research support from grants P50CA101942, U54CA163125, P01AI054456 and R01AI089955. K.M.M. acknowledges research support from the Claudia Adams Barr Program for Innovative Cancer Research, an American Association for Cancer Research Basic Cancer Research Fellowship (14-40-01-MAHO), and an American Society of Clinical Oncology Young Investigator Award supported by the Kidney Cancer Association.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
G.J.F. has patents pending royalties on the programmed cell death protein 1 (PD1) pathway from Bristol-Myers Squibb, Roche, Merck, EMD-Serono, Boehringer-Ingelheim, AstraZeneca, and Novartis. G.J.F. has served on advisory boards for CoStim, Novartis, Roche, and Bristol-Myers Squibb. K.M.M. and P.D.R. declare no competing interests.
Related links
Glossary
- Immunoediting
-
Immunological processes that identify and eliminate cancer cells. Also known as immune surveillance.
- Cytotoxic T lymphocyte antigen 4
-
(CTLA4). An immune checkpoint that competes with CD28 for its B7 ligands on antigen presenting cells.
- Neoantigens
-
Novel protein sequences that arise during cancer-associated mutagenesis and that can be processed into peptide antigens that are presented by MHC and recognized as foreign by T cells.
- Programmed cell death ligand 1
-
(PDL1). An immune checkpoint ligand expressed on immune cells, some normal tissues and many tumours. PDL1 binds to programmed cell death protein 1 on lymphocytes to inhibit T cell receptor signalling and activation.
- T cell exhaustion
-
A state of T cell non-responsiveness due to chronic activation.
- Programmed cell death protein 1
-
(PD1). An immune checkpoint receptor expressed on activated lymphocytes and highly expressed on exhausted T lymphocytes.
- Interferon-γ
-
(IFNγ). This cytokine triggers diverse cellular responses including macrophage activation and propagation of an immune response, as well as the expression of negative regulatory factors such as programmed cell death 1 ligand 1 and indoleamine 2,3-dioxygenase.
- Immune checkpoints
-
Inhibitory pathways that regulate the adaptive immune responses.
- R-CHOP
-
Acronym for the immunochemotherapy regimen used to treat non-Hodgkin lymphoma: rituximab (R); cyclophosphamide (C); hydroxydaunorubicin (H); Oncovin (O; Genus Pharmaceuticals; vincristine); prednisone (P).
- Breakthrough designations
-
The US Food and Drug Administration (FDA) designation for drugs that, based on preliminary clinical evidence, may substantially improve patient outcomes over available treatments. Breakthrough designation gives access to intensive guidance from the FDA regarding product development.
- Objective response rates
-
Percentage of patients whose tumors decrease in size with therapy. For example, by the RECIST (Response Evaluation Criteria in Solid Tumours) criteria, the percentage of patients whose tumours decrease by 30% or more.
- Superagonist
-
An agent that produces a supra-physiological response, greater than the response seen in normal physiology.
- Co-stimulatory signal
-
A second, non-antigen-specific signal that works with T cell receptor signalling to increase T cell activation.
- Myeloid-derived suppressor cells
-
(MDSCs). A heterogeneous population of myeloid lineage cells with immunosuppresive activities.
- Histone deactylase
-
An intracellular protein that regulates gene transcription by modifying histones and thereby chromosome structure.
- BRAF
-
A serine/threonine kinase often associated with solid tumour oncogenesis and resistance to therapy; mutant BRAF may be an effective target for therapeutics, as seen with vemurafenib in V600E-mutated melanoma.
- Cross-presentation
-
The ability of some antigen presenting cells to take up extracellular proteins or cells and present their antigens in the context of MHC class I.
- TNF receptors
-
(TNFRs). A large family of proteins associated with diverse cellular activities, including the activation of lymphocytes.
- TNFR superfamily
-
(TNFRSF). A group containing approximately 30 type I or type III membrane proteins and several secreted proteins. TNFRSF receptors are characterized by the presence of one to four extracellular, cysteine-rich ligand-binding domains.
- Antibody-dependent cell-mediated cytotoxicity
-
(ADCC). Cell killing mediated by natural killer cells, macrophages, neutrophils and eosinophils through antibodies bound to Fc receptors expressed by these effector cells.
- Natural killer T cells
-
A type of lymphocyte that is thought to have the functions of both activated T cells and natural killer cells.
- T cell memory
-
The process of developing long-lived and self-renewing T cells that retain specificity for an antigen after the antigen has been removed, allowing rapid reactivation of the immune response.
- Fc receptor
-
A type of cell surface receptor that binds to the crystallizable fragment (Fc) region of antibodies to trigger a variety of cellular responses.
- Immunoglobulin superfamily
-
A large group of proteins containing a structural domain related to the immunoglobulin (Ig) domain of antibodies. Ig domains contain 70–110 amino acids with a charcteristic Ig fold giving them a sandwich-like structure composed of two sheets of anti-parallel β-strands.
- NY-ESO-1
-
A tumour antigen that is expressed on various tumours, including some melanoma. It may be used to track the immune response to the tumour by identifying NY-ESO-1-specific T cells using NY-ESO-1 peptide–MHC class I tetramers.
- Phosphatidylserine
-
A phospholipid in the cell membrane, normally found on the inner surface unless the cell is undergoing cell death, in which it can be found on the cell surface and serves as an 'eat me' signal for phagocytes. A ligand for the T cell immunoglobulin mucin 3 immune checkpoint.
- Galectin 9
-
(GAL9). A member of a family of proteins that can bind β-galactoside sugars. Some galectins are overexpressed by tumour cells and some can crosslink extracellular proteins (for example, 4-1BB, T cell immunoglobulin mucin 3) and potentiate their immunomodulatory function.
- Poliovirus receptor
-
(PVR). A protein that is expressed by dendritic cells, but may be overexpressed by some tumours, and can bind T cell immunoglobulin and ITIM domain (TIGIT), a related protein expressed on T cells and natural killer cells.
- Human leukocyte antigen
-
(HLA). The genetic designation for the human MHC
- Integrin
-
A protein that is specialized for cell–cell or cell–extracellular matrix interactions.
- Ectonucleotidases
-
Enzymes, such as CD39 and CD73, that are expressed on the external cell surface and metabolize nucleotides.
- Fast track designation
-
A US Food and Drug Administration (FDA) designation that expedites the FDA review process for a promising medication with the potential to address an unmet medical need for a serious or life-threatening condition with no adequate treatment or cure.
Rights and permissions
About this article
Cite this article
Mahoney, K., Rennert, P. & Freeman, G. Combination cancer immunotherapy and new immunomodulatory targets. Nat Rev Drug Discov 14, 561–584 (2015). https://doi.org/10.1038/nrd4591
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrd4591
This article is cited by
-
Bioinformatics analysis identifies GLUD1 as a prognostic indicator for clear cell renal cell carcinoma
European Journal of Medical Research (2024)
-
The CUL5 E3 ligase complex negatively regulates central signaling pathways in CD8+ T cells
Nature Communications (2024)
-
Inhibition of tumor immune escape by blocking PD-1/PD-L1 engagement with dual-targeting molecularly imprinted polymer layer
Cancer Nanotechnology (2023)
-
A comprehensive review on immune checkpoint inhibitors induced cardiotoxicity characteristics and associated factors
European Journal of Medical Research (2023)
-
Overcoming acquired resistance to cancer immune checkpoint therapy: potential strategies based on molecular mechanisms
Cell & Bioscience (2023)