Abstract
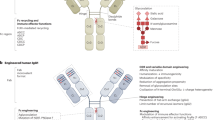
Fully human monoclonal antibodies (mAbs) are a promising and rapidly growing category of targeted therapeutic agents. The first such agents were developed during the 1980s, but none achieved clinical or commercial success. Advances in technology to generate the molecules for study — in particular, transgenic mice and yeast or phage display — renewed interest in the development of human mAbs during the 1990s. In 2002, adalimumab became the first human mAb to be approved by the US Food and Drug Administration (FDA). Since then, an additional six human mAbs have received FDA approval: panitumumab, golimumab, canakinumab, ustekinumab, ofatumumab and denosumab. In addition, 3 candidates (raxibacumab, belimumab and ipilimumab) are currently under review by the FDA, 7 are in Phase III studies and 81 are in either Phase I or II studies. Here, we analyse data on 147 human mAbs that have entered clinical study to highlight trends in their development and approval, which may help inform future studies of this class of therapeutic agents.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Reichert, J. M. Monoclonal antibodies as innovative therapeutics. Curr. Pharm. Biotechnol. 9, 423–430 (2008).
Reichert, J. M., Rosensweig, C. J., Faden, L. B. & Dewitz, M. C. Monoclonal antibody successes in the clinic. Nature Biotech. 23, 1073–1078 (2005).
Reichert, J. M. Antibodies to watch in 2010. MAbs 2, 84–100 (2010).
James, K. & Bell, G. T. Human monoclonal antibody production. Current status and future prospects. J. Immunol. Methods 100, 5–40 (1987).
Olsson, L. & Kaplan, H. S. Human–human hybridomas producing monoclonal antibodies of predefined antigenic specificity. Proc. Natl Acad. Sci. USA 77, 5429–5431 (1980).
Shoenfeld, Y. et al. Production of autoantibodies by human–human hybridomas. J. Clin. Invest. 70, 205–208 (1982).
Olsson, L. et al. Antibody producing human–human hybridomas. II. Derivation and characterization of an antibody specific for human leukemia cells. J. Exp. Med. 159, 537–550 (1984).
Kozbor, D. & Roder, J. C. Requirements for the establishment of high-titered human monoclonal antibodies against tetanus toxoid using the Epstein–Barr virus technique. J. Immunol. 127, 1275–1280 (1981).
Kozbor, D., Lagarde, A. E. & Roder, J. C. Human hybridomas constructed with antigen-specific Epstein–Barr virus-transformed cell lines. Proc. Natl Acad. Sci. USA 79, 6651–6655 (1982).
Beerli, R. R. & Rader, C. Mining human antibody repertoires. MAbs 2, 361–374 (2010).
Teng, N. N. et al. Protection against Gram-negative bacteremia and endotoxemia with human monoclonal IgM antibodies. Proc. Natl Acad. Sci. USA 82, 1790–1794 (1985).
Ziegler, E. J. et al. Treatment of Gram-negative bacteremia and septic shock with HA-1A human monoclonal antibody against endotoxin. A randomized, double-blind, placebo-controlled trial. The HA-1A Sepsis Study Group. N. Engl. J. Med. 324, 429–436 (1991).
Cross, A. S. Antiendotoxin antibodies: a dead end? Ann. Intern. Med. 121, 58–60 (1994).
Luce, J. M. Introduction of new technology into critical care practice: a history of HA-1A human monoclonal antibody against endotoxin. Crit. Care Med. 21, 1233–1241 (1993).
McCloskey, R. V., Straube, R. C., Sanders, C., Smith, S. M. & Smith, C. R. Treatment of septic shock with human monoclonal antibody HA-1A. A randomized, double-blind, placebo-controlled trial. CHESS Trial Study Group. Ann. Intern. Med. 121, 1–5 (1994).
Abbott. Abbott Annual Report 2008. Abbott website [online], (2008).
US Food and Drug Administration. Vectibix Panitumumab Injectable. Application No.: 125147. Medical Review(s). FDA website [online], (2006).
Van Cutsem, E. et al. Open-label phase III trial of panitumumab plus best supportive care compared with best supportive care alone in patients with chemotherapy-refractory metastatic colorectal cancer. J. Clin. Oncol. 25, 1658–1664 (2007).
Mazumdar, S. & Greenwald, D. Golimumab. MAbs 1, 422–431 (2009).
Lachmann, H. J. et al. Use of canakinumab in the cryopyrin-associated periodic syndrome. N. Engl. J. Med. 360, 2416–2425 (2009).
Dhimolea, E. Canakinumab. MAbs 2, 3–13 (2010).
Cingoz, O. Ustekinumab. MAbs 1, 216–221 (2009).
Teeling, J. L. et al. The biological activity of human CD20 monoclonal antibodies is linked to unique epitopes on CD20. J. Immunol. 177, 362–371 (2006).
Glennie, M. J., French, R. R., Cragg, M. S. & Taylor, R. P. Mechanisms of killing by anti-CD20 monoclonal antibodies. Mol. Immunol. 44, 3823–3837 (2007).
Zhang, B. Ofatumumab. MAbs 1, 326–331 (2009).
Pageau, S. C. Denosumab. MAbs 1, 210–215 (2009).
Mazumdar, S. Raxibacumab. MAbs 1, 531–538 (2009).
Dall'Era, M. & Wofsy, D. Connective tissue diseases: belimumab for systemic lupus erythematosus: breaking through? Nature Rev. Rheumatol. 6, 124–125 (2010).
Wallace, D. J. et al. A phase II, randomized, double-blind, placebo-controlled, dose-ranging study of belimumab in patients with active systemic lupus erythematosus. Arthritis Rheum. 61, 1168–1178 (2009).
Hodi, F. S. et al. Improved survival with ipilimumab in patients with metastatic melanoma. N. Engl. J. Med. 363, 711–723 (2010).
Reichert, J. M. & Dewitz, M. C. Anti-infective monoclonal antibodies: perils and promise of development. Nature Rev. Drug Discov. 5, 191–195 (2006).
Reichert, J. M. & Valge-Archer, V. E. Development trends for monoclonal antibody cancer therapeutics. Nature Rev. Drug Discov. 6, 349–356 (2007).
Green, L. L. et al. Antigen-specific human monoclonal antibodies from mice engineered with human Ig heavy and light chain YACs. Nature Genet. 7, 13–21 (1994).
Lonberg, N. et al. Antigen-specific human antibodies from mice comprising four distinct genetic modifications. Nature 368, 856–859 (1994).
Green, L. L. Antibody engineering via genetic engineering of the mouse: XenoMouse strains are a vehicle for the facile generation of therapeutic human monoclonal antibodies. J. Immunol. Methods 231, 11–23 (1999).
Lonberg, N. Human antibodies from transgenic animals. Nature Biotech. 23, 1117–1125 (2005).
McCafferty, J., Griffiths, A. D., Winter, G. & Chiswell, D. J. Phage antibodies: filamentous phage displaying antibody variable domains. Nature 348, 552–554 (1990).
Vaughan, T. J. et al. Human antibodies with sub-nanomolar affinities isolated from a large non-immunized phage display library. Nature Biotech. 14, 309–314 (1996).
Clackson, T., Hoogenboom, H. R., Griffiths, A. D. & Winter, G. Making antibody fragments using phage display libraries. Nature 352, 624–628 (1991).
Chirino, A. J., Ary, M. L. & Marshall, S. A. Minimizing the immunogenicity of protein therapeutics. Drug Discov. Today 9, 82–90 (2004).
Baert, F. et al. Influence of immunogenicity on the long-term efficacy of infliximab in Crohn's disease. N. Engl. J. Med. 348, 601–608 (2003).
De Groot, A. S. & Scott, D. W. Immunogenicity of protein therapeutics. Trends Immunol. 28, 482–490 (2007).
Bartelds, G. M. et al. Clinical response to adalimumab: relationship to anti-adalimumab antibodies and serum adalimumab concentrations in rheumatoid arthritis. Ann. Rheum. Dis. 66, 921–926 (2007).
Bender, N. K. et al. Immunogenicity, efficacy and adverse events of adalimumab in RA patients. Rheumatol. Int. 27, 269–274 (2007).
Hwang, W. Y. & Foote, J. Immunogenicity of engineered antibodies. Methods 36, 3–10 (2005).
Saif, M. W. & Cohenuram, M. Role of panitumumab in the management of metastatic colorectal cancer. Clin. Colorectal Cancer 6, 118–124 (2006).
Saif, M. W., Peccerillo, J. & Potter, V. Successful re-challenge with panitumumab in patients who developed hypersensitivity reactions to cetuximab: report of three cases and review of literature. Cancer Chemother. Pharmacol. 63, 1017–1022 (2009).
Chung, C. H. et al. Cetuximab-induced anaphylaxis and IgE specific for galactose-α-1,3-galactose. N. Engl. J. Med. 358, 1109–1117 (2008).
Lecluse, L. L. A. et al. Extent and clinical consequences of antibody formation against adalimumab in patients with plaque psoriasis. Arch. Dermatol. 146, 127–132 (2010).
Weinblatt, M. E. et al. Adalimumab, a fully human anti-tumor necrosis factor monoclonal antibody, for the treatment of rheumatoid arthritis in patients taking concomitant methotrexate: the ARMADA trial. Arthritis Rheum. 48, 35–45 (2003).
Nechansky, A. HAHA — nothing to laugh about. Measuring the immunogenicity (human anti-human antibody response) induced by humanized monoclonal antibodies applying ELISA and SPR technology. J. Pharm. Biomed. Anal. 51, 252–254 (2009).
Lofgren, J. A. et al. Comparing ELISA and surface plasmon resonance for assessing clinical immunogenicity of panitumumab. J. Immunol. 178, 7467–7472 (2007).
Jefferis, R. & LeFranc, M.-P. Human immunoglobulin allotypes — possible implications for immunogenicity. MAbs 1, 332–338 (2009).
Gilles, J. G. et al. Natural autoantibodies and anti-idiotypes. Semin. Thromb. Hemost. 26, 151–155 (2000).
Emmi, L. The role of intravenous immunoglobulin therapy in autoimmune and inflammatory disorders. Neurol. Sci. 23 (Suppl. 1), 1–8 (2002).
Harding, F. A. et al. The immunogenicity of humanized and fully human antibodies: residual immunogenicity residues in the CDR regions. Mabs 2, 256–265 (2010).
Shankar, G., Pendley, C. & Stein, K. E. A risk-based bioanalytical strategy for the assessment of antibody immune responses against biological drugs. Nature Biotech. 25, 555–561 (2007).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Glossary
- Allotype
-
Antibody allotypes are defined by their polymorphism within the immunoglobulin heavy and light chains. Natural allelic genetic variation in the constant region of genes in humans may predispose a given patient to anti-drug antibody responses if the drug is a foreign allotype.
- Ankylosing spondylitis
-
A chronic condition of unknown aetiology that is characterized by inflammation of the joints of the spine and pelvis. Disease progression may result in fusion of the joints.
- Anti-idiotypic antibody
-
An antibody that targets the hypervariable antigen-binding domain of an exogenous immunoglobulin, including therapeutic monoclonal antibodies. As the constant regions are fairly conserved, with the exception of allotypic differences, many anti-immunoglobulin responses will be directed against the highly variable, antigen-binding domain.
- Cryopyrin-associated periodic syndromes
-
(CAPS). A group of rare, inherited autoimmune disorders associated with over-secretion of interleukin-1 that may cause inflammation of the skin, eyes, bones, joints and meninges.
- Phage-display technologies
-
A method involving the use of bacteriophages to select desirable antibody variable domains based on their binding properties.
- Pre-transplant desensitization
-
In the recipient patient, reduction of antibody-producing cells or the amount of circulating antibodies that might target foreign tissue prior to transplantation of an organ.
- Systemic lupus erythematosus
-
A chronic, inflammatory autoimmune disease affecting connective tissue throughout the body.
Rights and permissions
About this article
Cite this article
Nelson, A., Dhimolea, E. & Reichert, J. Development trends for human monoclonal antibody therapeutics. Nat Rev Drug Discov 9, 767–774 (2010). https://doi.org/10.1038/nrd3229
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrd3229
This article is cited by
-
Fluid protein condensates for bio-inspired applications
Nature Reviews Bioengineering (2023)
-
Facile repurposing of peptide–MHC-restricted antibodies for cancer immunotherapy
Nature Biotechnology (2023)
-
Targeting galectin-driven regulatory circuits in cancer and fibrosis
Nature Reviews Drug Discovery (2023)
-
Simple chitin-based cell culture platform for production of biopharmaceuticals
Biotechnology Letters (2023)
-
Cell-penetrating porcine single-chain antibodies (transbodies) against nonstructural protein 1β (NSP1β) of porcine reproductive and respiratory syndrome virus inhibit virus replication
Archives of Virology (2023)