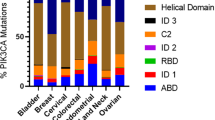
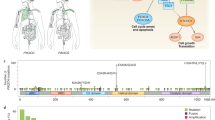
Key Points
-
The phosphatidylinositol-3-kinase (PI3K) pathway plays a crucial role in cell growth and survival and is activated in various cancers. Multiple components of the pathway are frequently targeted by amplification, mutation, and, less commonly, translocation in neoplasia; there is also crosstalk with the p53 and retinoblastoma pathways to comprise a signalling network that promotes tumour initiation and progression.
-
Despite major interest in this pathway for cancer drug discovery, no drugs have yet been approved that act specifically against PI3K or its downstream regulator, AKT. However, several drugs that were developed for other purposes either directly or indirectly target PI3K signaling, such as the rapamycin analogs, the ether lipids perifosine and miltefosine, and inhibitors of the epidermal growth factor receptor (EGFR), HER2, c-kit, platelet-derived growth factor receptor (PDGFR) and bcr–abl.
-
Because of the crucial role of the PI3K pathway in normal cell growth and in the cellular response to stress, the main challenge to developing PI3K pathway-targeted drugs is to identify inhibitors with a usable therapeutic index.
-
It is likely that PI3K inhibitors will need to be used in combination with other drugs that induce cell stress, such as other signaling inhibitors, radio- and chemotherapy.
-
Points at which therapeutic intervention might be useful in the PI3K pathway include PI3K itself, the downstream regulator AKT, although toxicity has thus far precluded use of specific inhibitors of this crucial signaling node, and other downstream components such as mTOR, integrin-linked kinase (ILK), phosphoinositide-dependent kinase-1 (PDK-1), p70S6 kinase, and Forkhead/FOXO1.
-
As with other molecularly targeted agents such as imatinib mesylate (Gleevec) and trastuzumab (Herceptin), the success of PI3K inhibitors will probably depend on the selection of cancer patients likely to be responders based on genomic aberrations. The co-development of molecular markers determining early response will allow triage of non-responders to other therapies and thereby increase the utility of targeted agents.
Abstract
Evolving studies with several different targeted therapeutic agents are demonstrating that patients with genomic alterations of the target, including amplification, translocation and mutation, are more likely to respond to the therapy. Recent studies indicate that numerous components of the phosphatidylinositol-3-kinase (PI3K)/AKT pathway are targeted by amplification, mutation and translocation more frequently than any other pathway in cancer patients, with resultant activation of the pathway. This warrants exploiting the PI3K/AKT pathway for cancer drug discovery.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Fruman, D. A., Meyers, R. E. & Cantley, L. C. Phosphoinositide kinases. Annu. Rev. Biochem. 67, 481–507 (1998). Excellent review of the structure and function of PI3Ks.
Hiles, I. D. et al. Phosphatidylinositol 3-kinase: structure and expression of the 110 kd catalytic subunit. Cell 70, 419–429 (1992).
Jimenez, C. et al. Identification and characterization of a new oncogene derived from the regulatory subunit of phosphoinositide 3-kinase. EMBO J. 17, 743–753 (1998).
Pawson, T. & Nash, P. Protein–protein interactions define specificity in signal transduction. Genes. Dev. 14, 1027–1047 (2000).
Davies, S. P., Reddy, H., Caivano, M. & Cohen, P. Specificity and mechanism of action of some commonly used protein kinase inhibitors. Biochem. J. 351, 95–105 (2000).
Oudit, G. Y. et al. The role of phosphoinositide-3 kinase and PTEN in cardiovascular physiology and disease. J. Mol. Cell Cardiol. 37, 449–471 (2004).
Bi, L., Okabe, I., Bernard, D. J., Wynshaw-Boris, A. & Nussbaum, R. L. Proliferative defect and embryonic lethality in mice homozygous for a deletion in the p110α subunit of phosphoinositide 3-kinase. J. Biol. Chem. 274, 10963–10968 (1999).
Brachmann, S. M., Ueki, K., Engelman, J. A., Kahn, R. C. & Cantley, L. C. Phosphoinositide 3-kinase catalytic subunit deletion and regulatory subunit deletion have opposite effects on insulin sensitivity in mice. Mol. Cell. Biol. 25, 1596–1607 (2005).
Okkenhaug, K. et al. Impaired B and T cell antigen receptor signaling in p110δ PI 3-kinase mutant mice. Science 297, 1031–1034 (2002).
Bilancio, A. et al. Key role of the p110δ isoform of PI3K in B cell antigen and IL4 receptor signalling- comparative analysis of genetic and pharmacological interference with p110δ function in B cells. Blood. 2005 Sep 22; [Epub ahead of print].
Northcott, C. A. et al. Upregulated function of phosphatidylinositol-3-kinase in genetically hypertensive rats: a moderator of arterial hypercontractility. Clin. Exp. Pharmacol. Physiol. 32, 851–858 (2005).
Condliffe, A. M. et al. Sequential activation of class IB and class IA PI3K is important for the primed respiratory burst of human but not murine neutrophils. Blood 106, 1432–1440 (2005).
Sujobert, P. et al. Essential role for the p110δ isoform in phosphoinositide 3-kinase activation and cell proliferation in acute myeloid leukemia. Blood 106, 1063–1066 (2005).
Yip, S. C. et al. Over-expression of the p110β but not p110α isoform of PI 3-kinase inhibits motility in breast cancer cells. Cell Motil. Cytoskeleton 59, 180–188 (2004).
Geng, L. et al. A specific antagonist of the p110δ catalytic component of phosphatidylinositol 3′-kinase, IC486068, enhances radiation-induced tumor vascular destruction. Cancer Res. 64, 4893–4899 (2004)
Campbell, M. et al. Glucose-potentiated chemotaxis in human vascular smooth muscle is dependent on cross-talk between the PI3K and MAPK signaling pathways. Circ. Res. 95, 380–388 (2004).
Gukovsky, I. et al. Phosphatidylinositide 3-kinase γ regulates key pathologic responses to cholecystokinin in pancreatic acinar cells. Gastroenterology 126, 554–566 (2004).
Leverrier, Y. et al. Class I phosphoinositide 3-kinase p110β is required for apoptotic cell and Fcγ receptor-mediated phagocytosis by macrophages. J. Biol. Chem. 278, 38437–38442 (2003).
Chantry, D. et al. p110δ, a novel phosphatidylinositol 3-kinase catalytic subunit that associates with p85 and is expressed predominantly in leukocytes. J. Biol. Chem. 272, 19236–19241 (1997).
Perrino, C. et al. Targeted inhibition of β-adrenergic receptor kinase-1-associated phosphoinositide-3 kinase activity preserves β-adrenergic receptor signaling and prolongs survival in heart failure induced by calsequestrin overexpression. J. Am. Coll. Cardiol. 45, 1862–1870 (2005).
Jackson, S. P. et al. PI3-kinase p110β: a new target for antithrombotic therapy. Nature Med. 11, 507–514 (2005).
Coffer, P. J. & Woodgett, J. R. Molecular cloning and characterisation of a novel putative protein-serine kinase related to the cAMP-dependent and protein kinase C families. Eur. J. Biochem. 201, 475–81 (1991).
Bellacosa, A., Testa, J. R., Staal, S. P. & Tsichlis, P. N. A retroviral oncogene, akt, encoding a serine-threonine kinase containing an SH2-like region. Science 254, 274–77 (1991).
Murthy, S. S., Tosolini, A., Taguchi, T. & Testa, J. R. Mapping of AKT3, encoding a member of the Akt/protein kinase B family, to human and rodent chromosomes by fluorescence in situ hybridization. Cytogenet. Cell Genet. 88, 39–40 (2000).
Alessi, D. R. et al. Mechanism of activation of protein kinase B by insulin and IGF-1. EMBO J. 15, 6541–551 (1996).
Blume-Jensen, P. & Hunter, T. Oncogenic kinase signalling. Nature 411, 355–65 (2001).
Lynch, D. K., Ellis, C. A., Edwards, P. A. & Hiles, I. D. Integrin-linked kinase regulates phosphorylation of serine 473 of protein kinase B by an indirect mechanism. Oncogene 18, 8024–32 (1999).
Kawakami, Y. et al. Protein kinase C βII regulates Akt phosphorylation on Ser-473 in a cell type- and stimulus-specific fashion. J. Biol. Chem. 279, 47720–5 (2004).
Sarbassov dos, D., Guertin, D. A., Ali, S. M. & Sabatini, D. M. Phosphorylation and regulation of Akt/PKB by the rictor-mTOR complex. Science 307, 1098–101 (2005).
Arboleda, M. J. et al. Overexpression of AKT2/protein kinase Bβ leads to up-regulation of β1 integrins, increased invasion, and metastasis of human breast and ovarian cancer cells. Cancer Res. 63, 196–206 (2003).
Yang, Z. Z. et al. Protein kinase B α/Akt1 regulates placental development and fetal growth. J. Biol. Chem. 278, 32124–31 (2003).
Peng, X. D. et al. Dwarfism, impaired skin development, skeletal muscle atrophy, delayed bone development, and impeded adipogenesis in mice lacking Akt1 and Akt2. Genes Dev. 17, 1352–65 (2003).
Cho, H., Thorvaldsen, J. L., Chu, Q., Feng, F. & Birnbaum, M. J. Akt1/PKBα is required for normal growth but dispensable for maintenance of glucose homeostasis in mice. J. Biol. Chem. 276, 38349–52 (2001).
Testa, J. R. & Bellacosa, A. AKT plays a central role in tumorigenesis. Proc. Natl Acad. Sci. USA 98, 10983–985 (2001).
Vivanco, I. & Sawyers, C. L. The phosphatidylinositol 3-Kinase AKT pathway in human cancer. Nature Rev. Cancer 2, 489–501 (2002).
Luo, J., Manning, B. D. & Cantley, L. C. Targeting the PI3K-Akt pathway in human cancer: rationale and promise. Cancer Cell 4, 257–62 (2003).
Chen, X. et al. Constitutively active Akt is an important regulator of TRAIL sensitivity in prostate cancer. Oncogene 20, 6073–83 (2001).
Wang, Q. et al. Regulation of TRAIL expression by the phosphatidylinositol 3-kinase/Akt/GSK-3 pathway in human colon cancer cells. J. Biol. Chem. 277, 36602–610 (2002).
Carroll, P. E. et al. Centrosome hyperamplification in human cancer: chromosome instability induced by p53 mutation and/or Mdm2 overexpression. Oncogene 18, 1935–44 (1999).
Toi, M., Saji, S., Suzuki, A., Yamamoto, Y., Tominaga, T. MDM2 in Breast Cancer. Breast Cancer 25, 264–68 (1997).
Sherr, C. J. & Weber, J. D. The ARF/p53 pathway. Curr. Opin. Genet. Dev. 10, 94–99 (2000).
Brunet, A. et al. Akt promotes cell survival by phosphorylating and inhibiting a Forkhead transcription factor. Cell 96, 857–68 (1999). Excellent review of some of the downstream mechanisms of AKT action.
Cahill, C. M. et al. Phosphatidylinositol 3-kinase signaling inhibits DAF-16 DNA binding and function via 14–3-3-dependent and 14–3-3-independent pathways. J. Biol. Chem. 276, 13402–10 (2001).
Wendel, H. G. et al. Survival signalling by Akt and eIF4E in oncogenesis and cancer therapy. Nature 428, 332–337 (2004).
Schmelzle, T. & Hall, M. N. TOR, a central controller of cell growth. Cell 103, 253–262 (2000). Excellent review of mTOR and its activity.
Lazaris-Karatzas, A., Montine, K. S. & Sonenberg, N. Malignant transformation by a eukaryotic initiation factor subunit that binds to mRNA 5′ cap. Nature 345, 544–547 (1990).
Polunovsky, V. A. et al. Translational control of the antiapoptotic function of Ras. J. Biol. Chem. 275, 24776–24780 (2000).
Potter, C. J., Pedraza, L. G. & Xu, T. Akt regulates growth by directly phosphorylating Tsc2. Nature Cell Biol. 4, 658–665 (2002).
Tee, A. R. et al. Tuberous sclerosis complex-1 and-2 gene products function together to inhibit mammalian target of rapamycin (mTOR)-mediated downstream signaling. Proc. Natl Acad. Sci. USA 99, 13571–576 (2002).
Dan, H. C. et al. Phosphatidylinositol 3-kinase/Akt pathway regulates tuberous sclerosis tumor suppressor complex by phosphorylation of tuberin. J. Biol. Chem. 277, 35364–35370 (2002).
Hengstschlager, M., Rosner, M., Fountoulakis, M. & Lubec, G. Tuberous sclerosis genes regulate cellular 14-3-3 protein levels. Biochem. Biophys. Res. Commun. 312, 676–83 (2003).
Harrington, L. S. et al. The TSC1–2 tumor suppressor controls insulin-PI3K signaling via regulation of IRS proteins. J. Cell Biol. 166, 213–23 (2004).
Zhang, H. et al. Loss of Tsc1/Tsc2 activates mTOR and disrupts PI3K-Akt signaling through downregulation of PDGFR. J. Clin. Invest. 112, 1223–1233 (2003).
Sun, S. Y. et al. Activation of Akt and eIF4E survival pathways by rapamycin-mediated mammalian target of rapamycin inhibition. Cancer Res. 65, 7052–7058 (2005).
Hay, N. The Akt-mTOR tango and its relevance to cancer. Cancer Cell. 8, 179–183 (2005).
Hardie, D. G., Scott, J. W., Pan, D. A. & Hudson, E. R. Management of cellular energy by the AMP-activated protein kinase system. FEBS Lett. 546, 113–120 (2003).
Shaw, R. J. et al. The tumor suppressor LKB1 kinase directly activates AMP-activated kinase and regulates apoptosis in response to energy stress. Proc. Natl Acad. Sci. USA 101, 3329–3335 (2004).
Shaw, R. J. et al. The LKB1 tumor suppressor negatively regulates mTOR signaling. Cancer Cell 6, 91–99 (2004).
Fujita, N., Sato, S. & Tsuruo, T. Phosphorylation of p27Kip1 at threonine 198 by p90 ribosomal protein S6 kinases promotes its binding to 14-3-3 and cytoplasmic localization. J. Biol. Chem. 278, 49254–49260 (2003).
Shin, I. et al. PKB/Akt mediates cell-cycle progression by phosphorylation of p27Kip1 at threonine 157 and modulation of its cellular localization. Nature Med. 8, 1145–1152 (2002).
Zhou, B. P. et al. Cytoplasmic localization of p21Cip1/WAF1 by Akt-induced phosphorylation in HER-2/neu-overexpressing cells. Nature Cell Biol. 3, 245–252 (2001).
Diehl, J. A., Cheng, M., Roussel, M. F. & Sherr, C. J. Glycogen synthase kinase-3β regulates cyclin D1 proteolysis and subcellular localization. Genes Dev. 12, 3499–3511 (1998).
Kim, W. Y. & Kaelin, W. G. Role of VHL gene mutation in human cancer. J. Clin. Oncol. 22, 4991–5004 (2004).
Skinner, H. D., Zheng, J. Z., Fang, J., Agani, F. & Jiang, B. H. Vascular endothelial growth factor transcriptional activation is mediated by hypoxia-inducible factor 1α, HDM2, and p70S6K1 in response to phosphatidylinositol 3-kinase/AKT signaling. J. Biol. Chem. 279, 45643–51 (2004).
Zhang, D. & Brodt, P. Type 1 insulin-like growth factor regulates MT1-MMP synthesis and tumor invasion via PI 3-kinase/Akt signaling. Oncogene 22, 974–82 (2003).
Kimura, A. et al. Induction of hTERT expression and phosphorylation by estrogen via Akt cascade in human ovarian cancer cell lines. Oncogene 23, 4505–15 (2004).
Remy, I., Montmarquette, A. & Michnick, S. W. PKB/Akt modulates TGF-β signalling through a direct interaction with Smad3. Nature Cell Biol. 6, 358–65 (2004).
Shou, J. et al. Mechanisms of tamoxifen resistance: increased estrogen receptor-HER2/neu cross-talk in ER/HER2-positive breast cancer. J. Natl Cancer Inst. 96, 926–35 (2004).
Mondesire, W. H. et al. Targeting mammalian target of rapamycin synergistically enhances chemotherapy-induced cytotoxicity in breast cancer cells. Clin. Cancer Res. 10, 7031–42 (2004).
Knuefermann, C. et al. HER2/PI-3K/Akt activation leads to a multidrug resistance in human breast adenocarcinoma cells. Oncogene 22, 3205–12 (2003).
Gupta, A. K. et al. Radiation sensitization of human cancer cells in vivo by inhibiting the activity of PI3K using LY294002. Int. J. Radiat. Oncol. Biol. Phys. 56, 846–53 (2003).
Brognard, J., Clark, A. S., Ni, Y. & Dennis, P. A. Akt/protein kinase B is constitutively active in non-small cell lung cancer cells and promotes cellular survival and resistance to chemotherapy and radiation. Cancer Res. 61, 3986–97 (2001).
Kim, D. et al. AKT/PKB signaling mechanisms in cancer and chemoresistance. Front Biosci. 10, 975–87 (2005).
Stoica, G. E. et al. Effect of estradiol on estrogen receptor-α gene expression and activity can be modulated by the ErbB2/PI 3-K/Akt pathway. Oncogene 22, 7998–8011 (2003).
Mendez, P., Azcoitia, I. & Garcia-Segura, L. M. Estrogen receptor α forms estrogen-dependent multimolecular complexes with insulin-like growth factor receptor and phosphatidylinositol 3-kinase in the adult rat brain. Brain Res. Mol. Brain Res. 112, 170–6 (2003).
Nagata, Y. et al. PTEN activation contributes to tumor inhibition by trastuzumab, and loss of PTEN predicts trastuzumab resistance in patients. Cancer Cell 6, 117–127 (2004). Excellent review of the role of PTEN in trastuzumab resistance in breast cancer.
Hanahan, D. & Weinberg, R. A. The hallmarks of cancer. Cell 100, 57–70 (2000). Excellent review of neoplastic progression due to the collective role of multiple genetic and molecular abnormalities.
Bachman, K. E. et al. The PIK3CA Gene is Mutated with High Frequency in Human Breast Cancers. Cancer Biol. Ther. 3, 772–775 (2004).
Kang, S., Bader, A. G. & Vogt, P. K. Phosphatidylinositol 3-kinase mutations identified in human cancer are oncogenic. Proc. Natl Acad. Sci. USA 102, 802–807 (2005). Excellent paper which demonstrates that PI3K mutations are oncogenic in human cancers.
Samuels, Y. et al. High frequency of mutations of the PIK3CA gene in human cancers. Science 304, 554 (2004).
Campbell, I. G. et al. Mutation of the PIK3CA gene in ovarian and breast cancer. Cancer Res. 64, 7678–7681 (2004).
Shayesteh, L. et al. PIK3CA is implicated as an oncogene in ovarian cancer. Nature Genet. 21, 99–102 (1999).
Woenckhaus, J. et al. Genomic gain of PIK3CA and increased expression of p110α are associated with progression of dysplasia into invasive squamous cell carcinoma. J. Pathol. 198, 335–342 (2002).
Ma, Y. Y. et al. PIK3CA as an oncogene in cervical cancer. Oncogene 19, 2739–2744 (2000).
Mizoguchi, M., Nutt, C. L., Mohapatra, G. & Louis, D. N. Genetic alterations of phosphoinositide 3-kinase subunit genes in human glioblastomas. Brain Pathol. 14, 372–377 (2004).
Broderick, D. K. et al. Mutations of PIK3CA in anaplastic oligodendrogliomas, high-grade astrocytomas, and medulloblastomas. Cancer Res. 64, 5048–5050 (2004).
Jucker, M. et al. Expression of a mutated form of the p85α regulatory subunit of phosphatidylinositol 3-kinase in a Hodgkin's lymphoma-derived cell line (CO). Leukemia 16, 894–901 (2002).
Cheng, J. Q. et al. Amplification of AKT2 in human pancreatic cells and inhibition of AKT2 expression and tumorigenicity by antisense RNA. Proc. Natl Acad. Sci. USA 93, 3636–3341 (1996).
Ruggeri, B. A., Huang, L., Wood, M., Cheng, J. Q. & Testa, J. R. Amplification and overexpression of the AKT2 oncogene in a subset of human pancreatic ductal adenocarcinomas. Mol. Carcinog. 21, 81–86 (1998).
Bellacosa, D. et al. Molecular alterations of the AKT2 oncogene in ovarian and breast carcinomas. Int. J. Cancer 64, 280–285 (1995).
Staal, S. P. Molecular cloning of the akt oncogene and its human homologues AKT1 and AKT2: amplification of AKT1 in a primary human gastric adenocarcinoma. Proc. Natl Acad. Sci. USA 84, 5034–5037 (1987).
Eng, C. PTEN: one gene, many syndromes. Hum. Mutat. 22, 183–198 (2003).
Li, Y. L., Tian, Z., Wu, D. Y., Fu, B. Y. & Xin, Y. Loss of heterozygosity on 10q23. 3 and mutation of tumor suppressor gene PTEN in gastric cancer and precancerous lesions. World J. Gastroenterol. 11, 285–288 (2005).
Li, J. et al. PTEN, a putative protein tyrosine phosphatase gene mutated in human brain, breast, and prostate cancer. Science 275, 1943–1947 (1997).
Cairns, P. et al. Frequent inactivation of PTEN/MMAC1 in primary prostate cancer. Cancer Res. 57, 4997–5000 (1997).
Garcia, J. M. et al. Allelic loss of the PTEN region (10q23) in breast carcinomas of poor pathophenotype. Breast Cancer Res. Treat. 57, 237–243 (1999).
Khan, S. et al. PTEN promoter is methylated in a proportion of invasive breast cancers. Int. J. Cancer 112, 407–410 (2004).
Goel, A. et al. Frequent inactivation of PTEN by promoter hypermethylation in microsatellite instability-high sporadic colorectal cancers. Cancer Res. 64, 3014–3021 (2004).
Stephens, P. et al. A screen of the complete protein kinase gene family identifies diverse patterns of somatic mutations in human breast cancer. Nature Genet. 37, 590–592 (2005).
Davies, H. et al. Somatic mutations of the protein kinase gene family in human lung cancer. Cancer Res. 65, 7591–7595 (2005).
Parsons, D. W. et al. Colorectal cancer: mutations in a signalling pathway. Nature 436, 792 (2005).
Wang, Z. et al. Mutational analysis of the tyrosine phosphatome in colorectal cancers. Science. 304, 1164–1166 (2004).
Rodriguez-Viciana, P. et al. Role of phosphoinositide 3-OH kinase in cell transformation and control of the actin cytoskeleton by Ras. Cell 89, 457–467 (1997).
Almoguera, C. et al. Most human carcinomas of the exocrine pancreas contain mutant c-K-ras genes. Cell 53, 549–554 (1988).
Yanez, L., Groffen, J. & Valenzuela, D. M. c-K-ras mutations in human carcinomas occur preferentially in codon 12. Oncogene 1, 315–318 (1987).
Nelson, M. A., Wymer, J. & Clements, N. Jr. Detection of K-ras gene mutations in non-neoplastic lung tissue and lung cancers. Cancer Lett 103, 115–121 (1996).
Davies, H. A. et al. Mutations of the BRAF gene in human cancer. Nature. 417, 949–954 (2002).
Rolitsky, C. D., Theil, K. S., McGaughy, V. R., Copeland, L. J. & Niemann, T. H. HER-2/neu amplification and overexpression in endometrial carcinoma. Int. J. Gynecol. Pathol. 18, 138–143 (1999).
Slamon, D. J. et al. Studies of the HER-2/neu proto-oncogene in human breast and ovarian cancer. Science 244, 707–712 (1989).
Stephens, P. et al. Lung cancer: intragenic ERBB2 kinase mutations in tumours. Nature 431, 525–526 (2004). Excellent paper which demonstrates that HER2 mutations occur in 10% human lung cancers
Klos, K. S. et al. Combined trastuzumab and paclitaxel treatment better inhibits ErbB-2-mediated angiogenesis in breast carcinoma through a more effective inhibition of Akt than either treatment alone. Cancer 98, 1377–1385 (2003).
Nicholson, K. M., Streuli, C. H. & Anderson, N. G. Autocrine signalling through erbB receptors promotes constitutive activation of protein kinase B/Akt in breast cancer cell lines. Breast Cancer Res. Treat. 81, 117–128 (2003).
Ekstrand, A. J., Sugawa, N., James, C. D. & Collins, V. P. Amplified and rearranged epidermal growth factor receptor genes in human glioblastomas reveal deletions of sequences encoding portions of the N- and/or C-terminal tails. Proc. Natl Acad. Sci. USA 89, 4309–4313 (1992).
Sordella, R., Bell, D. W., Haber, D. A. & Settleman, J. Gefitinib-sensitizing EGFR mutations in lung cancer activate anti-apoptotic pathways. Science 305, 1163–1167 (2004).
Tang, C. K., Gong, X. Q., Moscatello, D. K., Wong, A. J. & Lippman, M. E. Epidermal growth factor receptor vIII enhances tumorigenicity in human breast cancer. Cancer Res. 60, 3081–3087 (2000).
Barber, T. D., Vogelstein, B., Kinzler, K. W. & Velculescu, V. E. Somatic mutations of EGFR in colorectal cancers and glioblastomas. N. Engl. J. Med. 351, 2883 (2004).
Okamoto, I. et al. Expression of constitutively activated EGFRvIII in non-small cell lung cancer. Cancer Sci. 94, 50–56 (2003).
Rosenzweig, K. E., Youmell, M. B., Palayoor, S. T. & Price, B. D. Radiosensitization of human tumor cells by the phosphatidylinositol-3-kinase inhibitors wortmannin and LY294002 correlates with inhibition of DNA-dependent protein kinase and prolonged G2-M delay. Clin. Cancer Res. 3, 1149–1156 (1997).
Ng, S. S., Tsao, M. S., Nicklee, T. & Hedley, D. W. Wortmannin inhibits pkb/akt phosphorylation and promotes gemcitabine antitumor activity in orthotopic human pancreatic cancer xenografts in immunodeficient mice. Clin. Cancer Res. 7, 3269–3275 (2001).
Kim, S. H. et al. Potentiation of chemosensitivity in multidrug-resistant human leukemia CEM cells by inhibition of DNA-dependent protein kinase using wortmannin. Leuk. Res. 24, 917–925 (2000).
Schultz, R. M. et al. In vitro and in vivo antitumor activity of the phosphatidylinositol-3-kinase inhibitor, wortmannin. Anticancer Res. 15, 1135–1139 (1995).
Hu, L., Zaloudek, C., Mills, G. B., Gray, J. & Jaffe, R. B. In vivo and in vitro ovarian carcinoma growth inhibition by a phosphatidylinositol 3-kinase inhibitor (LY294002). Clin. Cancer Res. 6, 880–886 (2000).
Hu, L., Hofmann, J., Lu, Y., Mills, G. B. & Jaffe, R. B. Inhibition of phosphatidylinositol 3′-kinase increases efficacy of paclitaxel in in vitro and in vivo ovarian cancer models. Cancer Res. 62, 1087–1092 (2002).
Shokat, K. et al. Isoform-specific phosphoinositide 3-kinase inhibitors from an arylmorpholine scaffold. Bioorg. Med. Chem. 12, 4749–4759 (2004).
Ward, S. G. & Finan, P. Isoform-specific phosphoinositide 3-kinase inhibitors as therapeutic agents. Curr. Opin. Pharmacol. 3, 426–434 (2003).
Wymann, M. P., Zvelebil, M. & Laffargue, M. Phosphoinositide 3-kinase signalling — which way to target?, Trends. Pharmacol. Sci. 24, 366–376 (2003).
Roberson, A., Jackson, S., Kenche, V., Yaip, C., Parbaharan, H. & Thompson, P. Therapeutic Morpholino-Substituted Compounds. WO 01/53266 A1, Thrombogenix, 2001.
Giranda, V. et al. Novel ATP-competitive Akt inhibitors slow the progression of tumors in vivo. EORTC/AACR Molecular Therapeutics Geneva 2005 Abstract # 246
Defeo-Jones, D. et al. Tumor cell sensitization to apoptotic stimuli by selective inhibition of specific Akt/PKB family members. Mol. Cancer Ther. 4, 271–279 (2005).
Lindsley, C. W. et al. Allosteric Akt (PKB) inhibitors: discovery and SAR of isozyme selective inhibitors. Bioorg. Med. Chem. Lett. 15, 761–764 (2005).
Barnett, S. F. et al. Identification and characterization of pleckstrin-homology-domain-dependent and isoenzyme-specific Akt inhibitors. Biochem. J. 385, 399–408 (2005).
Kondapaka, S. B., Singh, S. S., Dasmahapatra, G. P., Sausville, E. A. & Roy, K. K. Perifosine, a novel alkylphospholipid, inhibits protein kinase B activation. Mol. Cancer Ther. 2, 1093–1103 (2003).
Van Ummersen, L. et al. A phase I trial of perifosine (NSC 639966) on a loading dose/maintenance dose schedule in patients with advanced cancer. Clin. Cancer Res. 10, 7450–7456 (2004).
Eng, C. P., Sehgal, S. N. & Vezina, C. Activity of rapamycin (AY-22, 989) against transplanted tumors. J. Antibiot. (Tokyo) 37, 1231–1237 (1984).
Dudkin, L. et al. Biochemical correlates of mTOR inhibition by the rapamycin ester CCI-779 and tumor growth inhibition. Clin. Cancer Res. 7, 1758–1764 (2001).
Shi, Y. et al. Rapamycin enhances apoptosis and increases sensitivity to cisplatin in vitro. Cancer Res. 55, 1982–1988 (1995).
deGraffenried, L. A. et al. Inhibition of mTOR activity restores tamoxifen response in breast cancer cells with aberrant Akt Activity. Clin. Cancer Res. 10, 8059–8067 (2004).
Atkins, M. B. et al. Randomized phase II study of multiple dose levels of CCI-779, a novel mammalian target of rapamycin kinase inhibitor, in patients with advanced refractory renal cell carcinoma. J. Clin. Oncol. 22, 909–918 (2004).
Chan, S. et al. First report: a phase 2 study of the safety and activity of CCI-779 for patients with locally advanced or metastatic breast cancer failing prior chemotherapy. Proc. Am. Soc. Clin. Oncol. 21, abstract 175 (2002).
Galanis, E. et al. NCCTG phase II trial of CCI-779 in recurrent glioblastoma multiforme (GBM). Proc. Am. Soc. Clin. Oncol. 22, 1503 (2004).
DeGraffenried, L. A. et al. Reduced PTEN expression in breast cancer cells confers susceptibility to inhibitors of the PI3 kinase/Akt pathway. Ann. Oncol. 15, 1510–1516 (2004).
Mills, G. B., Lu, Y. & Kohn, E. C. Linking molecular therapeutics to molecular diagnostics: inhibition of the FRAP/RAFT/TOR component of the PI3K pathway preferentially blocks PTEN mutant cells in vitro and in vivo. Proc. Natl Acad. Sci. USA 98, 10031–10033 (2001).
Kau, T. R. et al. A chemical genetic screen identifies inhibitors of regulated nuclear export of a Forkhead transcription factor in PTEN-deficient tumor cells. Cancer Cell. 4, 463–476 (2003).
Kim, K. M. & Lee, Y. J. Amiloride augments TRAIL-induced apoptotic death by inhibiting phosphorylation of kinases and phosphatases associated with the P13K-Akt pathway. Oncogene. 24, 355–366 (2005).
Slamon, D. J. et al. Use of chemotherapy plus a monoclonal antibody against HER2 for metastatic breast cancer that overexpresses HER2. N. Engl. J. Med. 344, 783–792 (2001).
Paez, J. G. et al. EGFR mutations in lung cancer: correlation with clinical response to gefitinib therapy. Science 304, 1497–1500 (2004).
Mukohara, T. et al. Differential effects of gefitinib and cetuximab on non-small-cell lung cancers bearing epidermal growth factor receptor mutations. J. Natl. Cancer Inst. 97, 1185–1194 (2005).
Haas-Kogan, D. A. et al. Biomarkers to predict response to epidermal growth factor receptor inhibitors. Cell Cycle. 4, (2005) [Epub ahead of print].
Giaccone, G. et al. A phase III clinical trial of ZD 1839 ('Iressa') in combination with gemcitabine and cisplatin in chemotherapy-naïve patients with advanced non-small cell lung cancer (INTACT-1). Ann. Oncol. 13, 2 (2002).
Johnson, D. H. et al. ZD1839 ('Iressa') in combination with paclitaxel and carboplatin in chemotherapy-naïve patients with advanced non-small cell lung cancer (NSCLC): initial results from a phase III trial (INTACT-2). Ann. Oncol. 13, 127 (2002).
Lynch, T. J. et al. Activating mutations in the epidermal growth factor receptor underlying responsiveness of non-small-cell lung cancer to gefitinib. N. Engl. J. Med. 350, 2129–2139 (2004).
Shepherd, F. A. et al. A randomized placebo-controlled trial of erlotinib in patients with advanced non-small cell lung cancer (NSCLC) following failure of 1st line or 2nd line chemotherapy. A National Cancer Institute of Canada Clinical Trials Group (NCIC CTG) trial. Proc. Am. Soc. Clin. Oncol. 22, 7022 (2004).
Moore, M. J. et al. Erlotinib improves survival when added to gemcitabine in patients with advanced pancreatic cancer. A phase III trial of the National Cancer Institute of Canada Clinical Trials Group [NCIC-CTG]. ASCO Gastrointestinal Cancers Symposium abstract 77 (2005).
Kohn, E. C. et al. Targeted Therapeutics in Breast Cancer: Challenges to Success. Diseases of the Breast (Editor Marc Lippman) In Press.
Kohn, E. C. et al. Molecular Therapeutics: promise and challenges. Semin Oncol 31, 39–53 (2004).
Drees, B. E., Mills, G. B., Rommel, C. & Prestwich G. D. Therapeutic potential of phosphoinositide 3-kinase inhibitors. Expert Opinion on therapeutic patents In Press.
Meric-Bernstam, F. & Mills, G. B. Mammalian target of rapamycin. Semin Oncol 31 (Suppl. 16), 10–17 (2004).
Debiec-Rychter, M. et al. Mechanisms of resistance to imatinib mesylate in gastrointestinal stromal tumors and activity of the PKC412 inhibitor against imatinib-resistant mutants. Gastroenterology 128, 270–279 (2005).
Bhalla, U. S., Ram, P. T. & Iyengar, R. MAP kinase phosphatase as a locus of flexibility in a mitogen-activated protein kinase signaling network. Science 297, 1018–1023 (2002).
Bhalla, U. S. & Iyengar, R. Emergent properties of networks of biological signaling pathways. Science 283, 381–387 (1999).
Eker, S. et al. Pathway logic: symbolic analysis of biological signaling. Pac Symp Biocomput 400–412 (2002).
Ferrell J. E. Jr., & Machleder, E. M. The biochemical basis of an all-or-none cell fate switch in Xenopus oocytes. Science 280, 895–898 (1998).
Hoffman, A. et al. The IκB–NF–κB signaling module: temporal control and selective gene activation. Science 298, 1241–1245 (2002).
Huang, C. Y. & Ferrell, J. E. Jr. Ultrasensitivity in the mitogen-activated protein kinase cascade. Proc. Natl Acad. Sci. USA 93, 10078–10083 (1996).
Ma'ayan, A. et al. Formation of regulatory patterns during signal propagation in a Mammalian cellular network. Science 309, 1078–1083 (2005).
Park, C. S., Schneider, I. C. & Haugh, J. M. Kinetic analysis of platelet-derived growth factor receptor/phosphoinositide 3-kinase/Akt signaling in fibroblasts. J. Biol. Chem. 278, 37064–37072 (2003).
Sachs, K. et al. Causal protein-signaling networks derived from multiparameter single-cell data. Science 308, 523–529 (2005).
Liu, X. et al. Rapamycin inhibits Akt-mediated oncogenic transformation and tumor growth. Anticancer Res. 24, 2697–2704 (2004).
Wendel, H. G. et al. Survival signalling by Akt and eIF4E in oncogenesis and cancer therapy. Nature 428, 332–337 (2004).
Podsypanina, K. et al. An inhibitor of mTOR reduces neoplasia and normalizes p70/S6 kinase activity in Pten+/− mice. Proc. Natl Acad. Sci. USA 98, 10320–10325 (2001).
Neshat, M. S. et al. Enhanced sensitivity of PTEN-deficient tumors to inhibition of FRAP/mTOR. Proc. Natl Acad. Sci. USA 98, 10314–10319 (2001).
Pardanani, A. et al. FIP1L1-PDGFRA fusion: prevalence and clinicopathologic correlates in 89 consecutive patients with moderate to severe eosinophilia. Blood 104, 3038–3045 (2004).
Druker, B. J. et al. Effects of a selective inhibitor of the Abl tyrosine kinase on the growth of Bcr-Abl positive cells. Nature Med. 2, 561–566 (1996).
Samuels, Y. et al. Mutant PIK3CA promotes cell growth and invasion of human cancer cells. Cancer Cell. 7, 561–573 (2005).
Lu, Y., Zi, X. & Pollak, M. Molecular mechanisms underlying IGF-I-induced attenuation of the growth-inhibitory activity of trastuzumab (Herceptin) on SKBR3 breast cancer cells. Int. J. Cancer 108, 334–341 (2004).
Ritter, C. A. et al. Mechanisms of resistance development against trastuzumab (Herceptin) in an in vivo breast cancer model. Int. J. Clin. Pharmacol. Ther. 42, 642–643 (2004).
Thompson, J. E. & Thompson, C. B. Putting the rap on Akt. J. Clin. Oncol. 22, 4217–4226 (2004). Excellent review of AKT involvement in cancer cell metabolism.
Warburg, O., Posener, K. & Negelein, E. Uber den stoffwechsel der tumoren. Biochem. Z. 152, 319–344 (1924).
Rathmell, J. C. et al. Akt-directed glucose metabolism can prevent Bax conformation change and promote growth factor-independent survival. Mol. Cell. Biol. 23, 7315–7328 (2003).
Bomanji, J. B., Costa, D. C. & Ell, P. J. Clinical role of positron emission tomography in oncology. Lancet Oncol. 2, 157–164 (2001).
Samuelson, A. V. & Lowe, S. W. Selective induction of p53 and chemosensitivity in RB-deficient cells by E1A mutants unable to bind the RB-related proteins. Proc. Natl Acad. Sci. USA 94, 12094–12099 (1997).
George, D. J. & Kaelin, W. G. Jr. The von Hippel–Lindau protein, vascular endothelial growth factor, and kidney cancer. N. Engl. J. Med. 349, 419–421 (2003). Excellent review of roles of vHL and VEGF in renal cell carcinoma.
Acknowledgements
The authors' research is supported by National Institutes of Health (NIH) grants to G.B.M. as well as by a Department Of Defence grant to P.T.R., and Aventis Pharmaceutical M.D. Anderson fellowship award to B.T.H., and a training fellowship from the Keck Centern Pharmacoinformatic Training Program at the Gulf Coast Consortia (NIH grant) to D.L.S.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
G.B.M. has financial relationships with QLT Inc., Semafore Pharmaceuticals, Eli Lilly & Co., Keryx Biopharmaceuticals and Abbott Laboratories.
Related links
Related links
DATABASES
Entrez Gene
OMIM
dermatofibrosarcoma protuberans
FURTHER INFORMATION
Nucleic Acids Reaseach Database List
The Sanger Institute Catalogues of Somatic Mutations in Cancer
Glossary
- HAMARTOMAS
-
Abnormal growth of mature normal cells and tissues in an organ composed of identical elements.
- REVERSE PHASE PROTEIN ARRAY
-
An array that immobilizes the whole repertoire of patient proteins that represent the state of individual tissue cell populations undergoing disease transitions.
Rights and permissions
About this article
Cite this article
Hennessy, B., Smith, D., Ram, P. et al. Exploiting the PI3K/AKT Pathway for Cancer Drug Discovery. Nat Rev Drug Discov 4, 988–1004 (2005). https://doi.org/10.1038/nrd1902
Issue Date:
DOI: https://doi.org/10.1038/nrd1902
This article is cited by
-
Targeting PI3K/Akt in Cerebral Ischemia Reperfusion Injury Alleviation: From Signaling Networks to Targeted Therapy
Molecular Neurobiology (2024)
-
Atractylodin induces apoptosis through downregulation of PI3Kγ-mediated PI3K/Akt/mTOR/p70S6K signalling in colon cancer cells and suppresses the tumour formation in xenograft mice model
Macromolecular Research (2024)
-
Enhancement of PD-L1-attenuated CAR-T cell function through breast cancer-associated fibroblasts-derived IL-6 signaling via STAT3/AKT pathways
Breast Cancer Research (2023)
-
GBAP1 functions as a tumor promotor in hepatocellular carcinoma via the PI3K/AKT pathway
BMC Cancer (2023)
-
KCNAB2 overexpression inhibits human non-small-cell lung cancer cell growth in vitro and in vivo
Cell Death Discovery (2023)