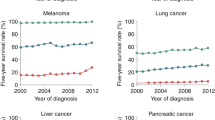
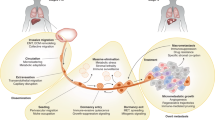
Abstract
Metastasis is the leading cause of cancer death. The metastatic cascade is a complex yet inefficient process that we have only begun to understand in recent years. Several of the early steps of this cascade are not readily targetable in the clinic. Past therapeutic developmental strategies have not distinguished between micrometastases and overt metastases. This lack of understanding is apparent in therapies that have been developed for patients with metastatic disease that are not efficacious in patients with micrometastatic disease; that is, in the adjuvant setting. Moreover, drugs that target distant metastases often do not work in the adjuvant setting. This Review will discuss our current understanding of the metastatic cascade as it relates to therapy, emerging therapeutic targets in the metastatic process, and how novel antimetastatic therapies might be developed for clinical use.
Key Points
-
Targeting the right steps of the metastatic cascade is vital for successful clinical development in cancer
-
Metastatic colonization is the pivotal transition between micrometastases and macrometastases; it constitutes the major target in the metastatic cascade
-
Targetable processes and cells of the metastatic cascade include: the tumor microenvironment, the genetic background, angiogenesis, the immune axis, dormancy, and cancer-stem cells
-
Metastases inhibitor trials should go beyond testing efficacy and safety, and include monitoring micrometastatic burden as well as assessing the ideal duration for the suppression of a specific target
-
The basic assumption that what works in macrometastases will work for micrometastases needs to be revisited
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Giantonio, B. J. et al. Bevacizumab in combination with oxaliplatin, fluorouracil, and leucovorin (FOLFOX4) for previously treated metastatic colorectal cancer: results from the Eastern Cooperative Oncology Group Study E3200. J. Clin. Oncol. 25, 1539–1544 (2007).
Allegra, C. J. et al. Phase III trial assessing bevacizumab in stages II and III carcinoma of the colon: results of NSABP protocol C-08. J. Clin. Oncol. 29, 11–16 (2011).
Van Cutsem, E., Lambrechts, D., Prenen, H., Jain, R. K. & Carmeliet, P. Lessons from the adjuvant bevacizumab trial on colon cancer: what next? J. Clin. Oncol. 29, 1–4 (2011).
Hanahan, D. & Weinberg, R. A. The hallmarks of cancer. Cell 100, 57–70 (2000).
Kerbel, R. S. Tumor angiogenesis: past, present and the near future. Carcinogenesis 21, 505–515 (2000).
Leenders, W. P., Kusters, B. & de Waal, R. M. Vessel co-option: how tumors obtain blood supply in the absence of sprouting angiogenesis. Endothelium 9, 83–87 (2002).
Van Cutsem, E. et al. Cetuximab and chemotherapy as initial treatment for metastatic colorectal cancer. N. Engl. J. Med. 360, 1408–1417 (2009).
Ebos, J. M. & Kerbel, R. S. Antiangiogenic therapy: impact on invasion, disease progression and metastasis. Nat. Rev. Clin. Oncol. 8, 210–221 (2011).
Berger, J. C., Vander Griend, D. J., Robinson, V. L., Hickson, J. A. & Rinker-Schaeffer, C. W. Metastasis suppressor genes: from gene identification to protein function and regulation. Cancer Biol. Ther. 4, 805–812 (2005).
Ma, L. et al. Therapeutic silencing of miR-10b inhibits metastasis in a mouse mammary tumor model. Nat. Biotechnol. 28, 341–347 (2010).
Palmieri, D. et al. Vorinostat inhibits brain metastatic colonization in a model of triple-negative breast cancer and induces DNA double-strand breaks. Clin. Cancer Res. 15, 6148–6157 (2009).
Chambers, A. F., MacDonald, I. C., Schmidt, E. E., Morris, V. L. & Groom, A. C. Clinical targets for anti-metastasis therapy. Adv. Cancer Res. 79, 91–121 (2000).
Steeg, P. S. Tumor metastasis: mechanistic insights and clinical challenges. Nat. Med. 12, 895–904 (2006).
Weiss, L., Ward, P. M. & Holmes, J. C. Liver-to-lung traffic of cancer cells. Int. J. Cancer 32, 79–83 (1983).
Weiss, L., Voit, A. & Lane, W. W. Metastatic patterns in patients with carcinomas of the lower esophagus and upper rectum. Invasion Metastasis 4, 47–60 (1984).
Weiss, L. et al. Haematogenous metastatic patterns in colonic carcinoma: an analysis of 1541 necropsies. J. Pathol. 150, 195–203 (1986).
Ma, L., Teruya-Feldstein, J. & Weinberg, R. A. Tumour invasion and metastasis initiated by microRNA-10b in breast cancer. Nature 449, 682–688 (2007).
Palmieri, D. et al. Medroxyprogesterone acetate elevation of Nm23-H1 metastasis suppressor expression in hormone receptor-negative breast cancer. J. Natl Cancer Inst. 97, 632–642 (2005).
Nguyen, D. X., Bos, P. D. & Massague, J. Metastasis: from dissemination to organ-specific colonization. Nat. Rev. Cancer 9, 274–284 (2009).
Taylor, J. L. et al. New paradigms for the function of JNKK1/MKK4 in controlling growth of disseminated cancer cells. Cancer Lett. 272, 12–22 (2008).
Guise, T. A. et al. Basic mechanisms responsible for osteolytic and osteoblastic bone metastases. Clin. Cancer Res. 12, 6213s–6216s (2006).
Mundy, G. R. Metastasis to bone: causes, consequences and therapeutic opportunities. Nat. Rev. Cancer 2, 584–593 (2002).
Gnant, M. et al. Endocrine therapy plus zoledronic acid in premenopausal breast cancer. N. Engl. J. Med. 360, 679–691 (2009).
Coleman, R. E. et al. Adjuvant treatment with zoledronic acid in stage II/III breast cancer. The AZURE Trial (BIG 01/04) [abstract S4-5]. 70 (24 Suppl.), 86s (2010).
Fizazi, K. et al. Randomized phase II trial of denosumab in patients with bone metastases from prostate cancer, breast cancer, or other neoplasms after intravenous bisphosphonates. J. Clin. Oncol. 27, 1564–1571 (2009).
Lipton, A. et al. Randomized active-controlled phase II study of denosumab efficacy and safety in patients with breast cancer-related bone metastases. J. Clin. Oncol. 25, 4431–4437 (2007).
US National Library of Medicine. ClinicalTrials.gov [online], (2011).
Sossey-Alaoui, K. et al. Down-regulation of WAVE3, a metastasis promoter gene, inhibits invasion and metastasis of breast cancer cells. Am. J. Pathol. 170, 2112–2121 (2007).
Steeg, P. S., Bevilacqua, G., Pozzatti, R., Liotta, L. A. & Sobel, M. E. Altered expression of NM23, a gene associated with low tumor metastatic potential, during adenovirus 2 Ela inhibition of experimental metastasis. Cancer Res. 48, 6550–6554 (1988).
Smith, S. C. & Theodorescu, D. Learning therapeutic lessons from metastasis suppressor proteins. Nat. Rev. Cancer 9, 253–264 (2009).
Marshall, J. C., Lee, J. H. & Steeg, P. S. Clinical–translational strategies for the elevation of Nm23-H1 metastasis suppressor gene expression. Mol. Cell Biochem. 329, 115–120 (2009).
US National Library of Medicine. ClinicalTrials.gov [online], (2010).
Titus, B. et al. Endothelin axis is a target of the lung metastasis suppressor gene RhoGDI2. Cancer Res. 65, 7320–7327 (2005).
Said, N., Smith, S., Sanchez-Carbayo, M. & Theodorescu, D. Tumor endothelin-1 enhances metastatic colonization of the lung in mouse xenograft models of bladder cancer. J. Clin. Invest. 121, 132–147 (2011).
Dykxhoorn, D. M. MicroRNAs and metastasis: little RNAs go a long way. Cancer Res. 70, 6401–6406 (2010).
Gumireddy, K. et al. In vivo selection for metastasis promoting genes in the mouse. Proc. Natl Acad. Sci. USA 104, 6696–6701 (2007).
Armeanu-Ebinger, S. et al. Differential expression of invasion promoting genes in childhood rhabdomyosarcoma. Int. J. Oncol. 38, 993–1000 (2011).
Folkman, J. & Klagsbrun, M. Angiogenic factors. Science 235, 442–447 (1987).
Bishop, G. A. & Hostager, B. S. Molecular mechanisms of CD40 signaling. Arch. Immunol. Ther. Exp. (Warsz.) 49, 129–137 (2001).
Conway, E. M., Collen, D. & Carmeliet, P. Molecular mechanisms of blood vessel growth. Cardiovasc. Res. 49, 507–521 (2001).
Gale, N. W. & Yancopoulos, G. D. Growth factors acting via endothelial cell-specific receptor tyrosine kinases: VEGFs, angiopoietins, and ephrins in vascular development. Genes Dev. 13, 1055–1066 (1999).
Nelson, A. R., Fingleton, B., Rothenberg, M. L. & Matrisian, L. M. Matrix metalloproteinases: biologic activity and clinical implications. J. Clin. Oncol. 18, 1135–1149 (2000).
Carmeliet, P. Fibroblast growth factor-1 stimulates branching and survival of myocardial arteries: a goal for therapeutic angiogenesis? Circ. Res. 87, 176–178 (2000).
Lindahl, P. et al. Role of platelet-derived growth factors in angiogenesis and alveogenesis. Curr. Top. Pathol. 93, 27–33 (1999).
Loges, S., Mazzone, M., Hohensinner, P. & Carmeliet, P. Silencing or fueling metastasis with VEGF inhibitors: antiangiogenesis revisited. Cancer Cell 15, 167–170 (2009).
Ellis, L. M. Mechanisms of action of bevacizumab as a component of therapy for metastatic colorectal cancer. Semin. Oncol. 33, S1–S7 (2006).
Ebos, J. M. et al. Accelerated metastasis after short-term treatment with a potent inhibitor of tumor angiogenesis. Cancer Cell 15, 232–239 (2009).
Pàez-Ribes, M. et al. Antiangiogenic therapy elicits malignant progression of tumors to increased local invasion and distant metastasis. Cancer Cell 15, 220–231 (2009).
Schneider, B. P. & Sledge, G. W. Jr. Anti-VEGF therapy as adjuvant therapy: clouds on the horizon? Breast Cancer Res. 11, 303 (2009).
Kim, D. W., Lu, B. & Hallahan, D. E. Receptor tyrosine kinase inhibitors as anti-angiogenic agents. Curr. Opin. Investig. Drugs 5, 597–604 (2004).
Karin, M. Nuclear factor-κB in cancer development and progression. Nature 441, 431–436 (2006).
Karin, M., Yamamoto, Y. & Wang, Q. M. The IKK NF-κB system: a treasure trove for drug development. Nat. Rev. Drug Discov. 3, 17–26 (2004).
Rosenberg, S. A. et al. Treatment of 283 consecutive patients with metastatic melanoma or renal cell cancer using high-dose bolus interleukin 2. JAMA 271, 907–913 (1994).
Coppin, C. et al. Immunotherapy for advanced renal cell cancer. Cochrane Database Syst. Rev. (1):CD001425 (2005).
Liu, B., DeFilippo, A. M. & Li, Z. Overcoming immune tolerance to cancer by heat shock protein vaccines. Mol. Cancer Ther. 1, 1147–1151 (2002).
Seavey, M. M. et al. A novel human Her-2/neu chimeric molecule expressed by Listeria monocytogenes can elicit potent HLA-A2 restricted CD8-positive T cell responses and impact the growth and spread of Her-2/neu-positive breast tumors. Clin. Cancer Res. 15, 924–932 (2009).
Holmes, J. P. et al. Results of the first phase I clinical trial of the novel II-key hybrid preventive HER-2/neu peptide (AE37) vaccine. J. Clin. Oncol. 26, 3426–3433 (2008).
Pichichero, M. E. Improving vaccine delivery using novel adjuvant systems. Hum. Vaccin. 4, 262–270 (2008).
Hodi, F. S. Cytotoxic T-lymphocyte-associated antigen-4. Clin. Cancer Res. 13, 5238–5242 (2007).
Demicheli, R., Abbattista, A., Miceli, R., Valagussa, P. & Bonadonna, G. Time distribution of the recurrence risk for breast cancer patients undergoing mastectomy: further support about the concept of tumor dormancy. Breast Cancer Res. Treat. 41, 177–185 (1996).
Karrison, T. G., Ferguson, D. J. & Meier, P. Dormancy of mammary carcinoma after mastectomy. J. Natl Cancer Inst. 91, 80–85 (1999).
Vessella, R. L., Pantel, K. & Mohla, S. Tumor cell dormancy: an NCI workshop report. Cancer Biol. Ther. 6, 1496–1504 (2007).
Stearns, A. T., Hole, D., George, W. D. & Kingsmore, D. B. Comparison of breast cancer mortality rates with those of ovarian and colorectal carcinoma. Br. J. Surg. 94, 957–965 (2007).
Saphner, T., Tormey, D. C. & Gray, R. Annual hazard rates of recurrence for breast cancer after primary therapy. J. Clin. Oncol. 14, 2738–2746 (1996).
Willis, L. et al. Breast cancer dormancy can be maintained by small numbers of micrometastases. Cancer Res. 70, 4310–4317 (2010).
Townson, J. L. & Chambers, A. F. Dormancy of solitary metastatic cells. Cell Cycle 5, 1744–1750 (2006).
Naumov, G. N. et al. Ineffectiveness of doxorubicin treatment on solitary dormant mammary carcinoma cells or late-developing metastases. Breast Cancer Res. Treat. 82, 199–206 (2003).
Koebel, C. M. et al. Adaptive immunity maintains occult cancer in an equilibrium state. Nature 450, 903–907 (2007).
Aguirre-Ghiso, J. A., Estrada, Y., Liu, D. & Ossowski, L. ERK (MAPK) activity as a determinant of tumor growth and dormancy; regulation by p38 (SAPK). Cancer Res. 63, 1684–1695 (2003).
Aguirre-Ghiso, J. A., Ossowski, L. & Rosenbaum, S. K. Green fluorescent protein tagging of extracellular signal-regulated kinase and p38 pathways reveals novel dynamics of pathway activation during primary and metastatic growth. Cancer Res. 64, 7336–7345 (2004).
Barkan, D. et al. Inhibition of metastatic outgrowth from single dormant tumor cells by targeting the cytoskeleton. Cancer Res. 68, 6241–6250 (2008).
Goss, P. E. & Chambers, A. F. Does tumour dormancy offer a therapeutic target? Nat. Rev. Cancer 10, 871–877 (2010).
Maitland, N. J. & Collins, A. T. Cancer stem cells—a therapeutic target? Curr. Opin. Mol. Ther. 12, 662–673 (2010).
Patel, S. A., Ndabahaliye, A., Lim, P. K., Milton, R. & Rameshwar, P. Challenges in the development of future treatments for breast cancer stem cells. Breast Cancer (London) 2, 1–11 (2010).
Takebe, N., Harris, P. J., Warren, R. Q. & Ivy, S. P. Targeting cancer stem cells by inhibiting Wnt, Notch, and Hedgehog pathways. Nat. Rev. Clin. Oncol. 8, 97–106 (2011).
Von Hoff, D. D. et al. Inhibition of the hedgehog pathway in advanced basal-cell carcinoma. N. Engl. J. Med. 361, 1164–1172 (2009).
Rudin, C. M. et al. Treatment of medulloblastoma with hedgehog pathway inhibitor GDC-0449. N. Engl. J. Med. 361, 1173–1178 (2009).
US National Library of Medicine. ClinicalTrials.gov [online], (2010).
US National Library of Medicine. ClinicalTrials.gov [online], (2011).
US National Library of Medicine. ClinicalTrials.gov [online], (2009).
US National Library of Medicine. ClinicalTrials.gov [online], (2010).
US National Library of Medicine. ClinicalTrials.gov [online], (2008).
Miller, K. D. et al. A randomized phase II pilot trial of adjuvant marimastat in patients with early-stage breast cancer. Ann. Oncol. 13, 1220–1224 (2002).
Miller, K. D. et al. A randomized phase II feasibility trial of BMS-275291 in patients with early stage breast cancer. Clin. Cancer Res. 10, 1971–1975 (2004).
Pegram, M. D., Pietras, R., Bajamonde, A., Klein, P. & Fyfe, G. Targeted therapy: wave of the future. J. Clin. Oncol. 23, 1776–1781 (2005).
Kobayashi, H. et al. Dynamic micro-magnetic resonance imaging of liver micrometastasis in mice with a novel liver macromolecular magnetic resonance contrast agent DAB-Am64-(1B4M-Gd)64 . Cancer Res. 61, 4966–4970 (2001).
Branca, R. T. et al. Molecular MRI for sensitive and specific detection of lung metastases. Proc. Natl Acad. Sci. USA 107, 3693–3697 (2010).
Maitland, M. L., Hudoba, C., Snider, K. L. & Ratain, M. J. Analysis of the yield of phase II combination therapy trials in medical oncology. Clin. Cancer Res. 16, 5296–5302 (2010).
Pantel, K., Alix-Panabieres, C. & Riethdorf, S. Cancer micrometastases. Nat. Rev. Clin. Oncol. 6, 339–351 (2009).
Pantel, K. & Riethdorf, S. Pathology: are circulating tumor cells predictive of overall survival? Nat. Rev. Clin. Oncol. 6, 190–191 (2009).
Braun, S. et al. A pooled analysis of bone marrow micrometastasis in breast cancer. N. Engl. J. Med. 353, 793–802 (2005).
Meng, S. et al. Circulating tumor cells in patients with breast cancer dormancy. Clin. Cancer Res. 10, 8152–8162 (2004).
US National Library of Medicine. ClinicalTrials.gov [online], (2011).
De Gramont, A. et al. AVANT: Results from randomized, three-arm multinational phase III study to investigate bevacizumab with either XELOX or FOLFOX4 versus FOLFOX4 alone as adjuvant treatment for colon cancer [abstract]. J. Clin. Oncol. 29 (Suppl. 4), a362 (2011).
Goss, P. E. et al. Randomized trial of letrozole following tamoxifen as extended adjuvant therapy in receptor-positive breast cancer: updated findings from NCIC CTG MA.17. J. Natl Cancer Inst. 97, 1262–1271 (2005).
US National Library of Medicine. ClinicalTrials.gov [online], (2010).
US National Library of Medicine. ClinicalTrials.gov [online], (2009).
Ciuleanu, T. et al. Maintenance pemetrexed plus best supportive care versus placebo plus best supportive care for non-small-cell lung cancer: a randomised, double-blind, phase 3 study. Lancet 374, 1432–1440 (2009).
Sargent, D. J. & Hayes, D. F. Assessing the measure of a new drug: is survival the only thing that matters? J. Clin. Oncol. 26, 1922–1923 (2008).
Cortazar, P., Johnson, R., Justice, R. et al. Adjuvant breast cancer: FDA approval overview [abstract]. J. Clin. Oncol. 27 (Suppl.), e11529 (2009).
Author information
Authors and Affiliations
Contributions
L. A. Mina and G. W. Sledge Jr contributed equally to researching the data for this article, discussion of the content and writing of the manuscript. Both authors also reviewed and edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Mina, L., Sledge, G. Rethinking the metastatic cascade as a therapeutic target. Nat Rev Clin Oncol 8, 325–332 (2011). https://doi.org/10.1038/nrclinonc.2011.59
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrclinonc.2011.59
This article is cited by
-
Repurposing fluphenazine to suppress melanoma brain, lung and bone metastasis by inducing G0/G1 cell cycle arrest and apoptosis and disrupting autophagic flux
Clinical & Experimental Metastasis (2023)
-
LncRNA RPPH1 promotes colorectal cancer metastasis by interacting with TUBB3 and by promoting exosomes-mediated macrophage M2 polarization
Cell Death & Disease (2019)
-
The Prognostic Value of Cancer Stem Cell Markers (Notch1, ALDH1, and CD44) in Primary Colorectal Carcinoma
Journal of Gastrointestinal Cancer (2019)
-
The role of claudins in cancer metastasis
Oncogene (2017)
-
Pathological complete response and prognosis after neoadjuvant chemotherapy for HER2-positive breast cancers before and after trastuzumab era: results from a real-life cohort
British Journal of Cancer (2016)