Key Points
-
Nearly 70% of cases of life-threatening arrhythmias or sudden cardiac death (SCD) in individuals with a structurally normal heart are unexplained, even after molecular autopsy
-
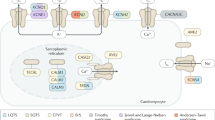
Autoimmune cardiac channelopathies are a novel and increasingly recognized mechanism of cardiac arrhythmias, which are mediated by circulating autoantibodies interfering with the function of various cardiac ion channels
-
By targeting calcium, potassium, or sodium channels, arrhythmogenic autoantibodies induce substantial, but potentially reversible, electrophysiological changes predisposing to the development of conduction disturbances and life-threatening tachyarrhythmias
-
Pathogenic autoantibodies against ion channels are detectable in patients with manifest autoimmune diseases and apparently healthy individuals, and might have a causal role in a subset of unexplained arrhythmias or SCDs
-
Specific autoantibody testing in patients with 'idiopathic' rhythm disturbances could lead to novel treatment opportunities, primarily immunomodulatory therapies
Abstract
Cardiac arrhythmias confer a considerable burden of morbidity and mortality in industrialized countries. Although coronary artery disease and heart failure are the prevalent causes of cardiac arrest, in 5–15% of patients, structural abnormalities at autopsy are absent. In a proportion of these patients, mutations in genes encoding cardiac ion channels are documented (inherited channelopathies), but, to date, the molecular autopsy is negative in nearly 70% of patients. Emerging evidence indicates that autoimmunity is involved in the pathogenesis of cardiac arrhythmias. In particular, several arrhythmogenic autoantibodies targeting specific calcium, potassium, or sodium channels in the heart have been identified. Experimental and clinical studies demonstrate that these autoantibodies can promote conduction disturbances and life-threatening tachyarrhythmias by inducing substantial electrophysiological changes. In this Review, we propose the term 'autoimmune cardiac channelopathies' to define this novel pathogenic mechanism of cardiac arrhythmias, which could be more frequent and clinically relevant than previously appreciated. Indeed, pathogenic autoantibodies against ion channels are detectable not only in patients with manifest autoimmune disease, but also in apparently healthy individuals, which suggests a causal role in some cases of unexplained arrhythmias and cardiac arrest. Considering this possibility and performing specific testing in patients with 'idiopathic' rhythm disturbances could create novel treatment opportunities.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Change history
06 July 2017
The version of this article initially published online included the following errors. In Box 1, phase 3 of the action potential was described as involving 'depolarization' instead of 'repolarization'. In the legend for Figure 1, IKs was defined as the 'rapid' component instead of the 'slow' component of the delayed rectifier K+ current. In the study from reference 115, the rabbits were described as being immunized with a peptide 'against' instead of 'from' the KCNQ1 K+ channel. Finally, there were errors with the references at the end of Box 1, and in the main text on pages 10 and 12. All these errors have been corrected for the print, HTML, and PDF versions of the article.
References
Zipes, D. P., et al. ACC/AHA/ESC 2006 guidelines for management of patients with ventricular arrhythmias and the prevention of sudden cardiac death: a report of the American College of Cardiology/American Heart Association Task Force and the European Society of Cardiology Committee for Practice Guidelines (writing committee to develop guidelines for management of patients with ventricular arrhythmias and the prevention of sudden cardiac death). J. Am. Coll. Cardiol. 48, e247–e346 (2006).
Bowker, T. J., et al. A national survey of sudden unexpected cardiac or unexplained death in adults (SADS). Heart 75, 80 (1996).
Puranik, R., Chow, C. K., Duflou, J. A., Kilborn, M. J. & McGuire, M. A. Sudden death in the young. Heart Rhythm 2, 1277–1282 (2005).
Cerrone, M. & Priori, S. G. Genetics of sudden death: focus on inherited channelopathies. Eur. Heart J. 32, 2109–2118 (2011).
Semsarian, C., Ingles, J. & Wilde, A. A. Sudden cardiac death in the young: the molecular autopsy and a practical approach to surviving relatives. Eur. Heart J. 36, 1290–1296 (2015).
Lazzerini, P. E. et al. Autoantibody-mediated cardiac arrhythmias: mechanisms and clinical implications. Basic Res. Cardiol. 103, 1–11 (2008).
Lee, H. C., Huang, K. T., Wang, X. L. & Shen, W. K. Autoantibodies and cardiac arrhythmias. Heart Rhythm 8, 1788–1795 (2011).
Karnabi, E. & Boutjdir, M. Role of calcium channels in congenital heart block. Scand. J. Immunol. 72, 226–234 (2010).
Qu, Y. & Boutjdir, M. in From Prediction to Prevention of Autoimmune Diseases (eds Conrad, K. et al.) 289–310 (Pabst, 2011).
Grant, A. O. Cardiac ion channels. Circ. Arrhythm. Electrophysiol. 2, 185–194 (2009).
Shaw, R. M. & Colecraft, H. M. L-type calcium channel targeting and local signalling in cardiac myocytes. Cardiovasc. Res. 98, 77–186 (2013).
Ono, K. & Iijima, T. Cardiac T-type Ca2+ channels in the heart. J. Mol. Cell. Cardiol. 48, 65–70 (2010).
Lazzerini, P. E. et al. Arrhythmogenic effects of anti-Ro/SSA antibodies on the adult heart: more than expected? Autoimmun. Rev. 9, 40–44 (2009).
Xiao, H. et al. Arrhythmogenic autoantibodies against calcium channel lead to sudden death in idiopathic dilated cardiomyopathy. Eur. J. Heart Fail. 13, 264–270 (2011). This study shows that in patients with idiopathic dilated cardiomyopathy, agonist-like autoantibodies against L-type calcium channels are frequently detected and independently predict ventricular tachycardia and sudden cardiac death.
Xiao, H. et al. Agonist-like autoantibodies against calcium channel in patients with dilated cardiomyopathy. Heart Vessels 27, 486–492 (2012).
Yu, H. et al. Calcium channel autoantibodies predicted sudden cardiac death and all-cause mortality in patients with ischemic and nonischemic chronic heart failure. Dis. Markers 2014, 796075 (2014).
Catterall, W. A., Perez-Reyes, E., Snutch, T. P. & Striessnig, J. International Union of Pharmacology. XLVIII. Nomenclature and structure–function relationships of voltage-gated calcium channels. Pharmacol. Rev. 57, 411–425 (2005).
Josephson, I. R. & Varadi, G. The beta subunit increases Ca2+ currents and gating charge movements of human cardiac L-type Ca2+ channels. Biophys. J. 70, 1285–1293 (1996).
Brito-Zerón, P., Izmirly, P. M., Ramos-Casals, M., Buyon, J. P. & Khamashta, M. A. The clinical spectrum of autoimmune congenital heart block. Nat. Rev. Rheumatol. 11, 301–312 (2015).
Ambrosi, A., Sonesson, S. E. & Wahren-Herlenius, M. Molecular mechanisms of congenital heart block. Exp. Cell Res. 325, 2–9 (2014).
Lazzerini, P. E., Capecchi, P. L. & Laghi Pasini, F. Anti-Ro/SSA antibodies and cardiac arrhythmias in the adult: facts and hypotheses. Scand. J. Immunol. 72, 213–222 (2010).
Litsey, S. E., Noonan, J. A., O'Connor, W. N., Cottrill, C. M. & Mitchell, B. Maternal connective tissue disease and congenital heart block. Demonstration of immunoglobulin in cardiac tissue. N. Engl. J. Med. 312, 98–100 (1985).
Lee, L. A., Coulter, S., Erner, S. & Chu, H. Cardiac immunoglobulin deposition in congenital heart block associated with maternal anti-Ro autoantibodies. Am. J. Med. 83, 793–796 (1987).
Clancy, R. M., Kapur, R. P., Molad, Y., Askanase, A. D. & Buyon, J. P. Immunohistologic evidence supports apoptosis, IgG deposition, and novel macrophage/fibroblast crosstalk in the pathologic cascade leading to congenital heart block. Arthritis Rheum. 50, 173–182 (2004).
Buyon, J. P. et al. Autoimmune-associated congenital heart block: demographics, mortality, morbidity and recurrence rates obtained from a national neonatal lupus registry. J. Am. Coll. Cardiol. 31, 1658–1666 (1998).
Brucato, A. et al. Risk of congenital complete heart block in newborns of mothers with anti-Ro/SSA antibodies detected by counterimmunoelectrophoresis: a prospective study of 100 women. Arthritis Rheum. 44, 1832–1835 (2001).
Julkunen, H. & Eronen, M. The rate of recurrence of isolated congenital heart block: a population-based study. Arthritis Rheum. 44, 487–488 (2001).
Gladman, G. et al. Fetal echocardiographic screening of pregnancies of mothers with anti-Ro and/or anti-La antibodies. Am. J. Perinatol. 19, 73–80 (2002).
Cimaz, R., Spence, D. L., Hornberger, L. & Silverman, E. D. Incidence and spectrum of neonatal lupus erythematosus: a prospective study of infants born to mothers with anti-Ro autoantibodies. J. Pediatr. 142, 678–683 (2003).
Kaaja, R. & Julkunen, H. Prevention of recurrence of congenital heart block with intravenous immunoglobulin & corticosteroid therapy: comment on the editorial by Buyon et al. Arthritis Rheum. 48, 280–281 (2003).
Solomon, D. G., Rupel, A. & Buyon, J. P. Birth order, gender and recurrence rate in autoantibody-associated congenital heart block: implications for pathogenesis and family counseling. Lupus 12, 646–647 (2003).
Costedoat-Chalumeau, N. et al. Outcome of pregnancies in patients with anti-SSA/Ro antibodies: a study of 165 pregnancies, with special focus on electrocardiographic variations in the children and comparison with a control group. Arthritis Rheum. 50, 3187–3194 (2004).
Grava, C. et al. Isolated congenital heart block in undifferentiated connective tissue disease and in primary Sjögren's syndrome: a clinical study of 81 pregnancies in 41 patients [Italian]. Reumatismo 57, 180–186 (2005).
Gerosa, M. et al. Electrocardiographic abnormalities in infants born from mothers with autoimmune diseases-a multicentre prospective study. Rheumatology 46, 1285–1289 (2007).
Friedman, D. M., et al. Utility of cardiac monitoring in fetuses at risk for congenital heart block: the PR Interval and Dexamethasone Evaluation (PRIDE) prospective study. Circulation 117, 485–493 (2008).
Pisoni, C. N. et al. Failure of intravenous immunoglobulin to prevent congenital heart block: findings of a multicenter, prospective, observational study. Arthritis Rheum. 62, 1147–1152 (2010).
Friedman, D. M. et al. Evaluation of fetuses in a study of intravenous immunoglobulin as preventive therapy for congenital heart block: results of a multicenter, prospective, open-label clinical trial. Arthritis Rheum. 62, 1138–1146 (2010).
Jaeggi, E. T. et al. Prolongation of the atrioventricular conduction in fetuses exposed to maternal anti-Ro/SSA and anti-La/SSB antibodies did not predict progressive heart block. A prospective observational study on the effects of maternal antibodies on 165 fetuses. J. Am. Coll. Cardiol. 57, 1487–1492 (2011).
Skog, A., Lagnefeldt, L., Conner, P., Wahren-Herlenius, M. & Sonesson, S. E. Outcome in 212 anti-Ro/SSA-positive pregnancies and population-based incidence of congenital heart block. Acta Obstet. Gynecol. Scand. 95, 98–105 (2016).
Hayashi, N. et al. Prevalence of disease-specific antinuclear antibodies in general population: estimates from annual physical examinations of residents of a small town over a 5-year period. Mod. Rheumatol. 18, 153–160 (2008).
Satoh, M. et al. Prevalence and sociodemographic correlates of antinuclear antibodies in the United States. Arthritis Rheum. 64, 2319–2327 (2012).
Guo, Y. P. et al. The prevalence of antinuclear antibodies in the general population of china: a cross-sectional study. Curr. Ther. Res. Clin. Exp. 76, 116–119 (2014).
Franceschini, F. & Cavazzana, I. Anti-Ro/SSA and anti-La/SSB antibodies. Autoimmunity 38, 55–63 (2005).
Buyon, J. P. et al. Identification of mothers at risk for congenital heart block and other neonatal lupus syndromes in their children. Comparison of enzyme-linked immunosorbent assay and immunoblot for measurement of anti-SS-A/Ro and anti-SS-B/La antibodies. Arthritis Rheum. 36, 1263–1273 (1993).
Boutjdir, M. et al. Serum and immunoglobulin G from the mother of a child with congenital heart block induce conduction abnormalities and inhibit L-type calcium channels in a rat heart model. Pediatr. Res. 44, 11–19 (1998).
Boutjdir, M. et al. Arrhythogenicity of IgG and anti-52-kD SSA/Ro affinity-purified antibodies from mothers of children with congenital heart block. Circ. Res. 80, 354–362 (1997). This study shows that purified IgG antibodies and anti-Ro/SSA-52 kD autoantibodies from sera of mothers whose children have congenital heart block induce complete atrioventricular block in the human fetal heart and inhibit the L-type calcium current.
Xiao, G. Q., Qu, Y., Hu, K. & Boutjdir, M. Down-regulation of L-type calcium channel in pups born to 52 kDa SSA/Ro immunized rabbits. FASEB J. 15, 1539–1545 (2001).
Garcia, S. et al. Cellular mechanism of the conduction abnormalities induced by serum from anti-Ro/SSA-positive patients in rabbit hearts. J. Clin. Invest. 93, 718–724 (1994).
Viana, V. S. et al. Induction of in vitro heart block is not restricted to affinity purified anti-52 kDa Ro/SSA antibody from mothers of children with neonatal lupus. Lupus 7, 141–147 (1998).
Hamilton, R. M., Lee-Poy, M., Kruger, K. & Silverman, E. D. Investigative methods of congenital complete heart block. J. Electrocardiol. 30 (Suppl.), 69–74 (1998).
Hu, K., Qu, Y., Yue, Y. & Boutjdir, M. Functional basis of sinus bradycardia in congenital heart block. Circ. Res. 94, e32–e38 (2004).
Xiao, G. Q., Hu, K. & Boutjdir, M. Direct inhibition of expressed cardiac l- and t-type calcium channels by IgG from mothers whose children have congenital heart block. Circulation 103, 1599–1604 (2001). IgG antibodies from mothers positive for anti-Ro/SSA autoantibodies and whose children have congenital heart block inhibited L-type and T-type calcium channels, but not sodium or delayed-rectifier potassium channels, expressed in Xenopus laevis oocytes.
Qu, Y., Baroudi, G., Yue, Y. & Boutjdir, M. Novel molecular mechanism involving α1D (Cav 1.3) L-type calcium channel in autoimmune-associated sinus bradycardia. Circulation 111, 3034–3041 (2005).
Qu, Y., Xiao, G. Q., Chen, L. & Boutjdir, M. Autoantibodies from mothers of children with congenital heart block downregulate cardiac L-type Ca channels. J. Mol. Cell. Cardiol. 33, 1153–1163 (2001).
Karnabi, E. et al. Congenital heart block: identification of autoantibody binding site on the extracellular loop (domain I, S5-S6) of alpha(1D) L-type Ca channel. J. Autoimmun. 34, 80–86 (2010).
Salomonsson, S. et al. Ro/SSA autoantibodies directly bind cardiomyocytes, disturb calcium homeostasis, and mediate congenital heart block. J. Exp. Med. 201, 11–17 (2005).
Clancy, R. M. & Buyon, J. P. More to death than dying: apoptosis in the pathogenesis of SSA/Ro-SSB/La- associated congenital heart block. Rheum. Dis. Clin. North. Am. 30, 589–602 (2004).
Qu, Y. & Boutjdir, M. Gene expression of SERCA2a and L-and T-type Ca channels during human heart development. Pediatr. Res. 50, 569–574 (2001).
Itzhaki, I., Schiller, J., Beyar, R., Satin, J. & Gepstein, L. Calcium handling in embryonic stem cell-derived cardiac myocytes: of mice and men. Ann. NY Acad. Sci. 1080, 207–215 (2006).
Seguchi, M., Jarmakani, J. M., George, B. L. & Harding, J. A. Effect of Ca2+ antagonists on mechanical function in the neonatal heart. Pediatr. Res. 20, 838–842 (1986).
Karnabi, E., Qu, Y., Mancarella, S. & Boutjdir, M. Rescue and worsening of congenital heart block-associated electrocardiographic abnormalities in two transgenic mice. J. Cardiovasc. Electrophysiol. 22, 922–930 (2011).
Lazzerini, P. E., Capecchi, P. L. & Laghi-Pasini, F. The “invulnerability” of the adult conduction system to anti-Ro/SSA antibodies? A matter of calcium channel expression on the cardiomyocyte. J. Cardiovasc. Electrophysiol. 22, E88 (2011).
Lazzerini, P. E., Capecchi, P. L. & Laghi-Pasini, F. Isolated heart block of unknown origin in adults and anti-Ro/SSA antibodies: clinical evidence, putative mechanisms and therapeutic implications. Heart Rhythm 12, 449–454 (2015).
Santos-Pardo, I., Martínez-Morillo, M., Villuendas, R. & Bayes-Genis, A. Anti-Ro antibodies and reversible atrioventricular block. N. Engl. J. Med. 368, 2335–2337 (2013).
Saribayev, M. et al. Corticosteroid treatment normalizes QTc prolongation and improves heart block in an elderly patient with anti-Ro-positive systemic lupus erythematosus. Aging Clin. Exp. Res. 26, 337–339 (2014).
Lazzerini, P. E. et al. Isolated atrioventricular block of unknown origin in the adult and autoimmunity: diagnostic and therapeutic considerations exemplified by three anti-Ro/SSA-associated cases. Heart Rhythm Case Rep. 1, 293–299 (2015).
Villuendas, R. et al. Autoimmunity and atrioventricular block of unknown etiology in adults: the role of anti-Ro/SSA antibodies. J. Am. Coll. Cardiol. 63, 1335–1336 (2014).
Bergman, G., et al. Late development of complete atrioventricular block may be immune mediated and congenital in origin. Acta Paediatr. 103, 275–281 (2014).
Caforio, A. L. et al.; European Society of Cardiology Working Group on Myocardial and Pericardial Diseases. Current state of knowledge on aetiology, diagnosis, management, and therapy of myocarditis: a position statement of the European Society of Cardiology Working Group on Myocardial and Pericardial Diseases. Eur. Heart J. 34, 2636–2648 (2013).
Hamilton, R. M. & Azevedo, E. R. Sudden cardiac death in dilated cardiomyopathies. Pacing Clin. Electrophysiol. 32 (Suppl. 2), S32–S40 (2009).
Schultheiss, H. P. et al. Antibody-mediated enhancement of calcium permeability in cardiac myocytes. J. Exp. Med. 168, 2105–2119 (1988).
Kühl, U., Melzner, B., Schäfer, B., Schultheiss, H. P. & Strauer, B. E. The Ca-channel as cardiac autoantigen. Eur. Heart J. 12 (Suppl. D), 99–104 (1991).
Liao, Y. H. et al. A novel autoantibody against Ca2+ channel in patients with idiopathic dilated cardiomyopathy [abstract]. Circulation 114 (Suppl. 18), 2965 (2006).
Splawski, I. et al. CaV1.2 calcium channel dysfunction causes a multi-system disorder including arrhythmia and autism. Cell 119, 19–31 (2004).
Liao, Y. H. Interventional study of diltiazem in dilated cardiomyopathy: a report of multiple centre clinical trial in China. Chinese Cooperative Group Diltiazem Intervention Trial Dilated Cardiomyopathy. Int. J. Cardiol. 64, 25–30 (1998).
Restivo, M., Kozhevnikov, D. O. & Boutjdir, M. Optical mapping of activation patterns in an animal model of congenital heart block. Am. J. Physiol. Heart Circ. Physiol. 280, H1889–H1895 (2001).
Mazel, J. A., El-Sherif, N., Buyon, J. & Boutjdir, M. Electrocardiographic abnormalities in a murine model injected with IgG from mothers of children with congenital heart block. Circulation 99, 1914–1918 (1999).
Menon, A., Silverman, E. D., Gow, R. M. & Hamilton, R. M. Chronotropic competence of the sinus node in congenital complete heart block. Am. J. Cardiol. 82, 1119–1121 (1998).
Askanase, A. D. et al. Spectrum and progression of conduction abnormalities in infants born to mothers with anti-SSA/Ro-SSB/La antibodies. Lupus 11, 145–151 (2002).
Brucato, A., Cimaz, R., Catelli, L. & Meroni, P. Anti-Ro-associated sinus bradycardia in newborns. Circulation 102, E88–E89 (2000).
Chockalingam, P. et al. Persistent fetal sinus bradycardia associated with maternal anti-SSA/Ro and anti-SSB/La antibodies. J. Rheumatol. 38, 2682–2685 (2011).
Strandberg, L. S. et al. Congenital heart block maternal sera autoantibodies target an extracellular epitope on the α1G T-type calcium channel in human fetal hearts. PLoS ONE 8, e72668 (2013). This study shows that antibodies from mothers whose children have congenital heart block can target T-type calcium channels in human fetal cardiomyocytes.
Markham, A. J. et al. Reactivity to the p305 epitope of the α1G T-type calcium channel and autoimmune-associated congenital heart block. J. Am. Heart Assoc. 4, e001836 (2015).
Nerbonne, J. M. Molecular basis of functional myocardial potassium channel diversity. Card. Electrophysiol. Clin. 8, 257–273 (2016).
Drew, B. J. et al. Prevention of torsade de pointes in hospital settings: a scientific statement from the American Heart Association and the American College of Cardiology Foundation. Circulation 121, 1047–1060 (2010).
El-Sherif, N. & Boutjdir, M. Role of pharmacotherapy in cardiac ion channelopathies. Pharmacol. Ther. 155, 1321–1342 (2015).
Lazzerini, P. E., Capecchi, P. L. & Laghi-Pasini, F. Long QT syndrome: an emerging role for inflammation and immunity. Front. Cardiovasc. Med. 2, 26 (2015).
Boutjdir, M., Lazzerini, P. E., Capecchi, P. L., Laghi-Pasini, F. & El-Sherif, N. Potassium channel block and novel autoimmune-associated long QT syndrome. Card. Electrophysiol. Clin. 8, 373–384 (2016).
Cimaz, R. et al. QT interval prolongation in asymptomatic anti-SSA/Ro-positive infants without congenital heart block. Arthritis Rheum. 43, 1049–1053 (2000).
Gordon, P. A., Khamashta, M. A., Hughes, G. R. & Rosenthal, E. Increase in the heart rate-corrected QT interval in children of anti-Ro-positive mothers, with a further increase in those with siblings with congenital heart block: comment on the article by Cimaz et al. Arthritis Rheum. 44, 242–243 (2001).
Cimaz, R. et al. Concomitant disappearance of electrocardiographic abnormalities and of acquired maternal autoantibodies during the first year of life in infants who had QT interval prolongation and anti-SSA/Ro positivity without congenital heart block at birth. Arthritis Rheum. 48, 266–268 (2003).
Lazzerini, P. E. et al. Prolongation of the corrected QT interval in adult patients with anti-Ro/SSA-positive connective tissue diseases. Arthritis Rheum. 50, 1248–1252 (2004).
Lazzerini, P. E. et al. Comparison of frequency of complex ventricular arrhythmias in patients with positive versus negative anti-Ro/SSA and connective tissue disease. Am. J. Cardiol. 100, 1029–1034 (2007).
Jaeggi, E., Laskin, C., Hamilton, R., Kingdom, J. & Silverman, E. The importance of the level of maternal anti-Ro/SSA antibodies as a prognostic marker of the development of cardiac neonatal lupus erythematosus. J. Am. Coll. Cardiol. 55, 2778–2784 (2010).
Bourré-Tessier, J. et al. Prolonged corrected QT interval in anti-Ro/SSA-positive adults with systemic lupus erythematosus. Arthritis Care Res. 63, 1031–1037 (2011).
Lazzerini, P. E. et al. Anti-Ro/SSA-associated corrected QT interval prolongation in adults: the role of antibody level and specificity. Arthritis Care Res. 63, 1463–1470 (2011).
Pisoni, C. N. et al. Elevated IL-1β levels in anti-Ro/SSA connective tissue diseases patients with prolonged corrected QTc interval. Clin. Exp. Rheumatol. 33, 715–720 (2015).
Sham, S., Medheshwaran, M., Tamilselvam, T. & Rajeswari, S. Correlation of QT interval with disease activity in newly detected SLE patients at baseline and during flare. Indian J. Rheumatol. 10, 121–124 (2015).
Tufan, A. N. et al. Prolonged Tpeak–Tend interval in anti-Ro52 antibody-positive connective tissue diseases. Rheumatol. Int. 37, 67–73 (2017).
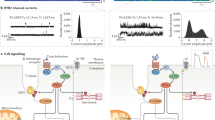
Yue, Y. et al. Pathogenesis of the novel autoimmune-associated long QT syndrome. Circulation 132, 230–240 (2015). This study was the first to establish an animal model for autoimmune-associated long QT syndrome and demonstrated that long QT was caused by action potential prolongation and direct inhibition of hERG1 potassium channel by anti-Ro/SSA autoantibodies from patients with autoimmune diseases.
Fabris, F. et al. Induction of autoimmune response to the extracellular loop of the HERG channel pore induces QTc prolongation in guinea-pigs. J. Physiol. 594, 6175–6187 (2016).
Motta, M., Rodriguez-Perez, C., Tincani, A., Lojacono, A. & Chirico, G. Outcome of infants from mothers with anti-SSA/Ro antibodies. J. Perinatol. 27, 278–283 (2007).
Costedoat-Chalumeau, N. et al. Corrected QT interval in anti-SSA–positive adults with connective tissue disease: Comment on the article by Lazzerini et al. Arthritis Rheum. 52, 676–677 (2005).
Teixeira, R. A. et al. Evidence for cardiac safety and antiarrhythmic potential of chloroquine in systemic lupus erythematosus. Europace 16, 887–892 (2014).
Massie, C. et al. Canadian Scleroderma Research Group (CSRG). Absence of an association between anti-Ro antibodies and prolonged QTc interval in systemic sclerosis: a multicenter study of 689 patients. Semin. Arthritis Rheum. 44, 338–344 (2014).
Gordon, P. A., Rosenthal, E., Khamashta, M. A. & Hughes, G. R. Absence of conduction defects in the electrocardiograms [correction of echocardiograms] of mothers with children with congenital complete heart block. J. Rheumatol. 28, 366–369 (2001).
Nomura, A. et al. Prolongation of heart rate-corrected QT interval is a predictor of cardiac autonomic dysfunction in patients with systemic lupus erythematosus. Rheumatol. Int. 34, 643–647 (2014).
Bourré-Tessier, J. et al. Electrocardiographic findings in systemic lupus erythematosus: data from an international inception cohort. Arthritis Care Res. (Hoboken) 67, 128–135 (2015).
Dugar, M., Cox, S., Limaye, V., Gordon, T. P. & Roberts-Thomson, P. J. Diagnostic utility of anti-Ro52 detection in systemic autoimmunity. Postgrad. Med. J. 86, 79–82 (2010).
Patel, C. & Antzelevitch, C. Pharmacological approach to the treatment of long and short QT syndromes. Pharmacol. Ther. 118, 138–151 (2008).
Lazzerini, P. E., Capecchi, P. L., Boutjdir, M. & Laghi-Pasini, F. Comment on “Absence of an association between anti-Ro antibodies and prolonged QTc interval in systemic sclerosis: a multicenter study of 689 patients” Semin. Arthritis Rheum. 44, e16–e17 (2015).
Nakamura, K. et al. Anti-KCNH2 antibody-induced long QT syndrome: novel acquired form of long QT syndrome. J. Am. Coll. Cardiol. 50, 1808–1809 (2007). This paper first described the association between torsades de pointes and anti-Ro/SSA autoantibodies in an apparently healthy individual who was asymptomatic for autoimmune disease, providing evidence of a direct mechanistic link between circulating anti-Ro/SSA antibodies and QTc prolongation.
Lazzerini, P. E. et al. Arrhythmogenicity of anti-Ro/SSA antibodies in patients with Torsades de Pointes. Circ. Arrhythm. Electrophysiol. 9, e003419 (2016). This paper shows that circulating anti-Ro/SSA-52 kD autoantibodies are frequently detected in patients with torsades de pointes, cross-react with the hERG1 potassium channels, and inhibit the rapid component of the delayed-rectifier potassium current.
Li, J. et al. Anti-KCNQ1 K+ channel autoantibodies increase IKs current and are associated with QT interval shortening in dilated cardiomyopathy. Cardiovasc. Res. 98, 496–503 (2013).
Li, J. et al. Induced KCNQ1 autoimmunity accelerates cardiac repolarization in rabbits: potential significance in arrhythmogenesis and antiarrhythmic therapy. Heart Rhythm 11, 2092–2100 (2014). This paper shows that immunization of rabbits with a peptide corresponding to the S5–S6 extracellular loop of KCNQ1 potassium channels results in enhancement of the slow component of the delayed-rectifier potassium current and decrease in action potential, leading to corrected QT interval shortening and increased susceptibility to ventricular tachyarrhythmias.
Niwa, N. & Nerbonne, J. M. Molecular determinants of cardiac transient outward potassium current (Ito) expression and regulation. J. Mol. Cell. Cardiol. 48, 12–25 (2010).
Suzuki, S. et al. Autoimmune targets of heart and skeletal muscles in myasthenia gravis. Arch. Neurol. 66, 1334–1338 (2009).
Suzuki, S. et al. Novel autoantibodies to a voltage-gated potassium channel Kv1.4 in a severe form of myasthenia gravis. J. Neuroimmunol. 170, 141–149 (2005).
Romi, F. et al. Anti-voltage-gated potassium channel Kv1.4 antibodies in myasthenia gravis. J. Neurol. 259, 1312–1316 (2012).
Suzuki, S. et al. Cardiac involvements in myasthenia gravis associated with anti-Kv1.4 antibodies. Eur. J. Neurol. 21, 223–230 (2014). This paper shows that circulating anti-K v 1.4 channel autoantibodies are present in a substantial percentage of patients with myasthenia gravis who frequently have corrected QT interval prolongation and in some cases died from lethal QT-associated arrhythmias.
Yoo, S. et al. Localization of Na+ channel isoforms at the atrioventricular junction and atrioventricular node in the rat. Circulation 114, 1360–1371 (2006).
Korkmaz, S. et al. Provocation of an autoimmune response to cardiac voltage-gated sodium channel NaV1.5 induces cardiac conduction defects in rats. J. Am. Coll. Cardiol. 62, 340–349 (2013). This paper shows that immunization of rats with a peptide corresponding to the S5–S6 extracellular loop of Na v 1.5 channels induces cardiac conduction defects, which are associated with reduced expression of sodium channels in the myocardium and decreased sodium current in cardiomyocytes.
Viswanathan, P. C., Benson, D. W. & Balser, J. R. A common SCN5A polymorphism modulates the biophysical effects of an SCN5A mutation. J. Clin. Invest. 111, 341–346 (2003).
Wang, D. W., Viswanathan, P. C., Balser, J. R., George, A. L. Jr & Benson, D. W. Clinical, genetic, and biophysical characterization of SCN5A mutations associated with atrioventricular conduction block. Circulation 105, 341–346 (2002).
Saxena, A., Izmirly, P. M., Mendez, B., Buyon, J. P. & Friedman, D. M. Prevention and treatment in utero of autoimmune-associated congenital heart block. Cardiol. Rev. 22, 263–237 (2014).
Frustaci, A. et al. Histological substrate of atrial biopsies in patients with lone atrial fibrillation. Circulation 96, 1180–1184 (1997).
Frustaci, A. et al. Cardiac histological substrate in patients with clinical phenotype of Brugada syndrome. Circulation 112, 3680–3687 (2005).
Acknowledgements
The authors were supported by award number I01BX002137 from Biomedical Laboratory Research and Development Service of Veterans Affairs Office of Research and Development to M.B., and by FAS-Salute ToRSADE project (FAS Salute 2014, Regione Toscana).
Author information
Authors and Affiliations
Contributions
P.E.L. researched data for the article and wrote the manuscript. All the authors discussed the content of the article and reviewed/edited the manuscript before submission. P.L.C., F.L.-P., and M.B. are co-senior authors of this manuscript
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Lazzerini, P., Capecchi, P., Laghi-Pasini, F. et al. Autoimmune channelopathies as a novel mechanism in cardiac arrhythmias. Nat Rev Cardiol 14, 521–535 (2017). https://doi.org/10.1038/nrcardio.2017.61
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrcardio.2017.61
This article is cited by
-
Fibroblast and Immune Cell Cross-Talk in Cardiac Fibrosis
Current Cardiology Reports (2023)
-
Heart Failure in Chronic Infectious and Inflammatory Conditions: Mechanistic Insights from Clinical Heterogeneity
Current Heart Failure Reports (2022)
-
Electroimmunology and cardiac arrhythmia
Nature Reviews Cardiology (2021)
-
The Role of Autoantibodies in Arrhythmogenesis
Current Cardiology Reports (2021)
-
Evaluation of cardiac function and systolic dyssynchrony of fetuses exposed to maternal autoimmune diseases using speckle tracking echocardiography
Clinical Rheumatology (2021)