Key Points
-
The heartbeat originates from spontaneous depolarizations in the sinoatrial node, which lead to the spread of electrical signals throughout the heart via a specialized conduction system
-
Failure of the sinoatrial node pacemaker or disease of the conduction system results in slow heart rates that can cause fainting or sudden death
-
Current therapies rely on electronic pacemakers to provide an adequate heart rate to satisfy haemodynamic needs
-
Electronic pacemaker technologies continue to evolve; however, electronic pacemakers have limitations including battery life, system failure, inability to provide true autonomic response, and device-related infections
-
Biological pacemakers, currently at the preclinical stage, might be an alternative to electronic devices for selected patients in the future
Abstract
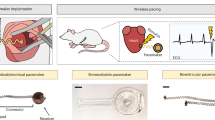
Electrogenesis in the heart begins in the sinoatrial node and proceeds down the conduction system to originate the heartbeat. Conduction system disorders lead to slow heart rates that are insufficient to support the circulation, necessitating implantation of electronic pacemakers. The typical electronic pacemaker consists of a subcutaneous generator and battery module attached to one or more endocardial leads. New leadless pacemakers can be implanted directly into the right ventricular apex, providing single-chamber pacing without a subcutaneous generator. Modern pacemakers are generally reliable, and their programmability provides options for different pacing modes tailored to specific clinical needs. Advances in device technology will probably include alternative energy sources and dual-chamber leadless pacing in the not-too-distant future. Although effective, current electronic devices have limitations related to lead or generator malfunction, lack of autonomic responsiveness, undesirable interactions with strong magnetic fields, and device-related infections. Biological pacemakers, generated by somatic gene transfer, cell fusion, or cell transplantation, provide an alternative to electronic devices. Somatic reprogramming strategies, which involve transfer of genes encoding transcription factors to transform working myocardium into a surrogate sinoatrial node, are furthest along in the translational pipeline. Even as electronic pacemakers become smaller and less invasive, biological pacemakers might expand the therapeutic armamentarium for conduction system disorders.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Miranda, J. O., Ramalho, C., Henriques-Coelho, T. & Areias, J. C. Fetal programming as a predictor of adult health or disease: the need to reevaluate fetal heart function. Heart Fail. Rev. 22, 861–877 (2017).
Friedman, D., Duncanson, L., Glickstein, J. & Buyon, J. A review of congenital heart block. Images Paediatr. Cardiol. 5, 36–48 (2003).
Marban, E. Cardiac channelopathies. Nature 415, 213–218 (2002).
Bers, D. M. Cardiac excitation-contraction coupling. Nature 415, 198–205 (2002).
Crick, S. J. et al. Innervation of the human cardiac conduction system. A quantitative immunohistochemical and histochemical study. Circulation 89, 1697–1708 (1994).
Anderson, R. H., Yanni, J., Boyett, M. R., Chandler, N. J. & Dobrzynski, H. The anatomy of the cardiac conduction system. Clin. Anat. 22, 99–113 (2009).
Anderson, R. H. & Ho, S. Y. The architecture of the sinus node, the atrioventricular conduction axis, and the internodal atrial myocardium. J. Cardiovasc. Electrophysiol. 9, 1233–1248 (1998).
Epstein, J. A. Franklin, H. Epstein Lecture. Cardiac development and implications for heart disease. N. Engl. J. Med. 363, 1638–1647 (2010).
van Weerd, J. H. & Christoffels, V. M. The formation and function of the cardiac conduction system. Development 143, 197–210 (2016).
Ionta, V. et al. SHOX2 overexpression favors differentiation of embryonic stem cells into cardiac pacemaker cells, improving biological pacing ability. Stem Cell Rep. 4, 129–142 (2015).
Kapoor, N., Liang, W., Marban, E. & Cho, H. C. Direct conversion of quiescent cardiomyocytes to pacemaker cells by expression of Tbx18. Nat. Biotechnol. 31, 54–62 (2013).
Liang, W., Cho, H. C. & Marban, E. Wnt signalling suppresses voltage-dependent Na+ channel expression in postnatal rat cardiomyocytes. J. Physiol. 593, 1147–1157 (2015).
Christoffels, V. M. & Moorman, A. F. Development of the cardiac conduction system: why are some regions of the heart more arrhythmogenic than others? Circul. Arrhythmia Electrophysiol. 2, 195–207 (2009).
Eisner, D. A. & Cerbai, E. Beating to time: calcium clocks, voltage clocks, and cardiac pacemaker activity. Am. J. Physiol. Heart Circ. Physiol. 296, H561–H562 (2009).
DiFrancesco, D. Characterization of single pacemaker channels in cardiac sino-atrial node cells. Nature 324, 470–473 (1986).
Mangoni, M. E. et al. Functional role of L-type Cav1.3 Ca2+ channels in cardiac pacemaker activity. Proc. Natl Acad. Sci. USA 100, 5543–5548 (2003).
Huser, J. et al. Functional coupling between glycolysis and excitation-contraction coupling underlies alternans in cat heart cells. J. Physiol. 524, 795–806 (2000).
Bogdanov, K. Y., Vinogradova, T. M. & Lakatta, E. G. Sinoatrial nodal cell ryanodine receptor and Na+-Ca2+ exchanger: molecular partners in pacemaker regulation. Circul. Res. 88, 1254–1258 (2001).
Groenke, S. et al. Complete atrial-specific knockout of sodium-calcium exchange eliminates sinoatrial node pacemaker activity. PloS ONE 8, e81633 (2013).
Torrente, A. G. et al. Burst pacemaker activity of the sinoatrial node in sodium-calcium exchanger knockout mice. Proc. Natl Acad. Sci. USA 112, 9769–9774 (2015).
DiFrancesco, D. & Borer, J. S. The funny current: cellular basis for the control of heart rate. Drugs 67 (Suppl. 2), 15–24 (2007).
Walsh, K. B. & Kass, R. S. Regulation of a heart potassium channel by protein kinase A and C. Science 242, 67–69 (1988).
Monfredi, O., Maltsev, V. A. & Lakatta, E. G. Modern concepts concerning the origin of the heartbeat. Physiology 28, 74–92 (2013).
Vinogradova, T. M. et al. High basal protein kinase A-dependent phosphorylation drives rhythmic internal Ca2+ store oscillations and spontaneous beating of cardiac pacemaker cells. Circul. Res. 98, 505–514 (2006).
Bleiziffer, S. et al. Predictors for new-onset complete heart block after transcatheter aortic valve implantation. JACC Cardiovasc. Interv. 3, 524–530 (2010).
Izmirly, P. M. et al. Clinical and pathologic implications of extending the spectrum of maternal autoantibodies reactive with ribonucleoproteins associated with cutaneous and now cardiac neonatal lupus from SSA/Ro and SSB/La to U1RNP. Autoimmun. Rev. 16, 980–983 (2017).
Ramos, S., Matturri, L., Rossi, L. & Rossi, M. Scleroatrophy of the atrioventricular junctional specialized tissue (Lenegre-Lev Disease) in chronic chagas' heart disease. Acta Cardiol. 50, 483–487 (1995).
Epstein, A. E. et al. 2012 ACCF/AHA/HRS focused update incorporated into the ACCF/AHA/HRS 2008 guidelines for device-based therapy of cardiac rhythm abnormalities: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines and the Heart Rhythm Society. J. Am. Coll. Cardiol. 61, e6–e75 (2013).
Greenspon, A. J. et al. Trends in permanent pacemaker implantation in the United States from 1993 to 2009: increasing complexity of patients and procedures. J. Am. Coll. Cardiol. 60, 1540–1545 (2012).
Aquilina, O. A brief history of cardiac pacing. Images Paediatr. Cardiol. 8, 17–81 (2006).
van Hemel, N. M. & van der Wall, E. E. 8 October 1958, D Day for the implantable pacemaker. Neth. Heart J. 16 (Suppl. 1), S3–S4 (2008).
Larsson, B., Elmqvist, H., Rydén, L. & Schüller, H. Lessons from the first patient with an implanted pacemaker: 1958–2001. Pacing Clin. Electrophysiol. 26, 114–124 (2003).
Chardack, W. M., Gage, A. A. & Greatbatch, W. A transistorized, self-contained, implantable pacemaker for the long-term correction of complete heart block. Surgery 48, 643–654 (1960).
Parsonnet, V., Driller, J., Cook, D. & Rizvi, S. A. Thirty-one years of clinical experience with “nuclear-powered” pacemakers. Pacing Clin. Electrophysiol. 29, 195–200 (2006).
Smyth, N. P., Keshishian, J. D., Garcia, J. M., Kelly, L. C. & Proctor, D. Clinical experience with the isotopic cardiac pacemaker. Ann. Thorac. Surg. 28, 14–21 (1979).
Burr, L. H. The lithium iodide-powered cardiac pacemaker. Clinical experience with 250 implantations. J. Thorac. Cardiovasc. Surg. 73, 421–423 (1977).
Mond, H. G. & Freitag, G. The cardiac implantable electronic device power source: evolution and revolution. Pacing Clin. Electrophysiol. 37, 1728–1745 (2014).
Boriani, G. et al. Role of ventricular Autocapture function in increasing longevity of DDDR pacemakers: a prospective study. Europace 8, 216–220 (2006).
Biffi, M. et al. Actual pacemaker longevity: the benefit of stimulation by automatic capture verification. Pacing Clin. Electrophysiol. 33, 873–881 (2010).
Milasinovic, G. et al. Percent ventricular pacing with managed ventricular pacing mode in standard pacemaker population. Europace 10, 151–155 (2008).
Gillis, A. M. et al. Reducing unnecessary right ventricular pacing with the managed ventricular pacing mode in patients with sinus node disease and AV block. Pacing Clin. Electrophysiol. 29, 697–705 (2006).
Saito, M. et al. Effect of right ventricular pacing on right ventricular mechanics and tricuspid regurgitation in patients with high-grade atrioventricular block and sinus rhythm (from the protection of left ventricular function during right ventricular pacing study). Am. J. Cardiol. 116, 1875–1882 (2015).
Ahmed, F. Z. et al. One-month global longitudinal strain identifies patients who will develop pacing-induced left ventricular dysfunction over time: the Pacing and Ventricular Dysfunction (PAVD) Study. PloS ONE 12, e0162072 (2017).
Madhavan, M., Mulpuru, S. K., McLeod, C. J., Cha, Y. M. & Friedman, P. A. Advances and future directions in cardiac pacemakers: part 2 of a 2-part series. J. Am. Coll. Cardiol. 69, 211–235 (2017).
Moss, A. J. et al. Cardiac-resynchronization therapy for the prevention of heart-failure events. N. Engl. J. Med. 361, 1329–1338 (2009).
Bristow, M. R. et al. Cardiac-resynchronization therapy with or without an implantable defibrillator in advanced chronic heart failure. N. Engl. J. Med. 350, 2140–2150 (2004).
Leclercq, C. et al. A randomized comparison of triple-site versus dual-site ventricular stimulation in patients with congestive heart failure. J. Am. Coll. Cardiol. 51, 1455–1462 (2008).
Turakhia, M. P. et al. Reduced mortality associated with quadripolar compared to bipolar left ventricular leads in cardiac resynchronization therapy. JACC Clin. Electrophysiol. 2, 426–433 (2016).
Mulpuru, S. K., Cha, Y. M. & Asirvatham, S. J. Synchronous ventricular pacing with direct capture of the atrioventricular conduction system: functional anatomy, terminology, and challenges. Heart Rhythm 13, 2237–2246 (2016).
Vijayaraman, P., Dandamudi, G., Worsnick, S. & Ellenbogen, K. A. Acute His-bundle injury current during permanent His-bundle pacing predicts excellent pacing outcomes. Pacing Clin. Electrophysiol. 38, 540–546 (2015).
Mulpuru, S. K., Madhavan, M., McLeod, C. J., Cha, Y. M. & Friedman, P. A. Cardiac pacemakers: function, troubleshooting, and management: part 1 of a 2-part series. J. Am. Coll. Cardiol. 69, 189–210 (2017).
Boriani, G. & Padeletti, L. Management of atrial fibrillation in bradyarrhythmias. Nat. Rev. Cardiol. 12, 337–349 (2015).
Hauser, R. G. et al. Clinical experience with pacemaker pulse generators and transvenous leads: an 8-year prospective multicenter study. Heart Rhythm 4, 154–160 (2007).
Sohail, M. R. et al. Management and outcome of permanent pacemaker and implantable cardioverter-defibrillator infections. J. Am. Coll. Cardiol. 49, 1851–1859 (2007).
Cingolani, E. & Marbán, E. Recreating the sinus node by somatic reprogramming: a dream come true? Rev. Esp. Cardiol. 68, 743–745 (2015).
Baddour, L. M. et al. Update on cardiovascular implantable electronic device infections and their management: a scientific statement from the American Heart Association. Circulation 121, 458–477 (2010).
Basar, N. et al. Upper-extremity deep vein thrombosis and downhill esophageal varices caused by long-term pacemaker implantation. Tex. Heart Inst. J. 37, 714–716 (2010).
Delling, F. N. et al. Tricuspid regurgitation and mortality in patients with transvenous permanent pacemaker leads. Am. J. Cardiol. 117, 988–992 (2016).
Miller, M. A., Neuzil, P., Dukkipati, S. R. & Reddy, V. Y. Leadless cardiac pacemakers: back to the future. J. Am. Coll. Cardiol. 66, 1179–1189 (2015).
Reddy, V. Y. et al. Permanent leadless cardiac pacing: results of the LEADLESS trial. Circulation 129, 1466–1471 (2014).
Piccini, J. P. et al. Long-term outcomes in leadless Micra transcatheter pacemakers with elevated thresholds at implantation: results from the Micra Transcatheter Pacing System Global Clinical Trial. Heart Rhythm 14, 685–691 (2017).
Singh, H. R., Batra, A. S. & Balaji, S. Pacing in children. Ann. Pediatr. Cardiol. 6, 46–51 (2013).
Fortescue, E. B. et al. Patient, procedural, and hardware factors associated with pacemaker lead failures in pediatrics and congenital heart disease. Heart Rhythm 1, 150–159 (2004).
Dagdeviren, C. et al. Conformal piezoelectric systems for clinical and experimental characterization of soft tissue biomechanics. Nat. Mater. 14, 728–736 (2015).
Dagdeviren, C. et al. Conformal piezoelectric energy harvesting and storage from motions of the heart, lung, and diaphragm. Proc. Natl Acad. Sci. USA 111, 1927–1932 (2014).
Haeberlin, A. et al. The first batteryless, solar-powered cardiac pacemaker. Heart Rhythm 12, 1317–1323 (2015).
Kypta, A. et al. Leadless cardiac pacemaker implantation after lead extraction in patients with severe device infection. J. Cardiovasc. Electrophysiol. 27, 1067–1071 (2016).
Bornzin, G. A. et al. Dual-chamber leadless intra-cardiac medical device with intra-cardiac extension. U.S. Patent 8634912 (2014).
Schaldach, M. & Hutten, H. Intracardiac impedance to determine sympathetic activity in rate responsive pacing. Pacing Clin. Electrophysiol. 15, 1778–1786 (1992).
Ellenbogen, K. A., Kay, G. N. & Wilkoff, B. L. Clinical Cardiac Pacing (W. B. Saunders Company, 1995).
Lloyd, M. et al. Rate adaptive pacing in an intracardiac pacemaker. Heart Rhythm 14, 200–205 (2017).
Cho, H. C. & Marbán, E. Biological therapies for cardiac arrhythmias: can genes and cells replace drugs and devices? Circul. Res. 106, 674–685 (2010).
Rosen, M. R. Gene therapy and biological pacing. N. Engl. J. Med. 371, 1158–1159 (2014).
Rosen, M. R., Robinson, R. B., Brink, P. R. & Cohen, I. S. The road to biological pacing. Nature reviews. Cardiology 8, 656–666 (2011).
Cingolani, E. Biological pacemakers: Ready for the clinic? Trends Cardiovasc. Med. 25, 674–675 (2015).
Gutierrez, M. et al. Maternal connective tissue disease associated with congenital AV block [Spanish]. Rev. Med. Chil. 117, 789–793 (1989).
Scott, J. S. & Taylor, P. V. Congenital AV-block: role of anti-Ro and anti-La antibodies. Springer Semin. Immunopathol. 11, 397–408 (1989).
Edelberg, J. M., Aird, W. C. & Rosenberg, R. D. Enhancement of murine cardiac chronotropy by the molecular transfer of the human β2 adrenergic receptor cDNA. J. Clin. Invest. 101, 337–343 (1998).
Edelberg, J. M., Huang, D. T., Josephson, M. E. & Rosenberg, R. D. Molecular enhancement of porcine cardiac chronotropy. Heart 86, 559–562 (2001).
Miake, J., Marban, E. & Nuss, H. B. Biological pacemaker created by gene transfer. Nature 419, 132–133 (2002).
Miake, J., Marban, E. & Nuss, H. B. Functional role of inward rectifier current in heart probed by Kir2.1 overexpression and dominant-negative suppression. J. Clin. Invest. 111, 1529–1536 (2003).
Tristani-Firouzi, M. et al. Functional and clinical characterization of KCNJ2 mutations associated with LQT7 (Andersen syndrome). J. Clin. Invest. 110, 381–388 (2002).
Baruscotti, M., Bucchi, A. & Difrancesco, D. Physiology and pharmacology of the cardiac pacemaker (“funny”) current. Pharmacol. Ther. 107, 59–79 (2005).
Qu, J. et al. Expression and function of a biological pacemaker in canine heart. Circulation 107, 1106–1109 (2003).
Plotnikov, A. N. et al. Biological pacemaker implanted in canine left bundle branch provides ventricular escape rhythms that have physiologically acceptable rates. Circulation 109, 506–512 (2004).
Rosen, M. R., Brink, P. R., Cohen, I. S. & Robinson, R. B. Genes, stem cells and biological pacemakers. Cardiovasc. Res. 64, 12–23 (2004).
Ruhparwar, A. et al. Transplanted fetal cardiomyocytes as cardiac pacemaker. Eur. J. Cardiothorac. Surg. 21, 853–857 (2002).
Xu, C., Police, S., Rao, N. & Carpenter, M. K. Characterization and enrichment of cardiomyocytes derived from human embryonic stem cells. Circul. Res. 91, 501–508 (2002).
Xue, T. et al. Functional integration of electrically active cardiac derivatives from genetically engineered human embryonic stem cells with quiescent recipient ventricular cardiomyocytes: insights into the development of cell-based pacemakers. Circulation 111, 11–20 (2005).
Kehat, I. et al. Electromechanical integration of cardiomyocytes derived from human embryonic stem cells. Nat. Biotechnol. 22, 1282–1289 (2004).
Protze, S. I. et al. Sinoatrial node cardiomyocytes derived from human pluripotent cells function as a biological pacemaker. Nat. Biotechnol. 35, 56–68 (2017).
Chauveau, S. et al. Induced pluripotent stem cell-derived cardiomyocytes provide in vivo biological pacemaker function. Circul. Arrhythmia Electrophysiol. 10, e004508 (2017).
Plotnikov, A. N. et al. Xenografted adult human mesenchymal stem cells provide a platform for sustained biological pacemaker function in canine heart. Circulation 116, 706–713 (2007).
Liechty, K. W. et al. Human mesenchymal stem cells engraft and demonstrate site-specific differentiation after in utero transplantation in sheep. Nat. Med. 6, 1282–1286 (2000).
Cho, H. C., Kashiwakura, Y. & Marban, E. Creation of a biological pacemaker by cell fusion. Circul. Res. 100, 1112–1115 (2007).
Bucchi, A. et al. Wild-type and mutant HCN channels in a tandem biological-electronic cardiac pacemaker. Circulation 114, 992–999 (2006).
Marban, E. & Cingolani, E. Direct reprogramming: bypassing stem cells for therapeutics. JAMA 314, 19–20 (2015).
Hu, Y. F., Dawkins, J. F., Cho, H. C., Marban, E. & Cingolani, E. Biological pacemaker created by minimally invasive somatic reprogramming in pigs with complete heart block. Sci. Transl Med. 6, 245ra94 (2014).
Potapova, I. et al. Human mesenchymal stem cells as a gene delivery system to create cardiac pacemakers. Circul. Res. 94, 952–959 (2004).
Tse, H. F. et al. Bioartificial sinus node constructed via in vivo gene transfer of an engineered pacemaker HCN Channel reduces the dependence on electronic pacemaker in a sick-sinus syndrome model. Circulation 114, 1000–1011 (2006).
Cingolani, E. et al. Biological pacemaker created by percutaneous gene delivery via venous catheters in a porcine model of complete heart block. Heart Rhythm 9, 1310–1318 (2012).
Liu, Q. et al. The role of capsid-endothelial interactions in the innate immune response to adenovirus vectors. Hum. Gene Ther. 14, 627–643 (2003).
Muruve, D. A. The innate immune response to adenovirus vectors. Hum. Gene Ther. 15, 1157–1166 (2004).
Chua, J. D. et al. Diagnosis and management of infections involving implantable electrophysiologic cardiac devices. Ann. Intern. Med. 133, 604–608 (2000).
Cabell, C. H. et al. Increasing rates of cardiac device infections among Medicare beneficiaries: 1990–1999. Am. Heart J. 147, 582–586 (2004).
Athan, E. et al. Clinical characteristics and outcome of infective endocarditis involving implantable cardiac devices. JAMA 307, 1727–1735 (2012).
Sohail, M. R., Henrikson, C. A., Braid-Forbes, M. J., Forbes, K. F. & Lerner, D. J. Mortality and cost associated with cardiovascular implantable electronic device infections. Arch. Intern. Med. 171, 1821–1828 (2011).
Mirowski, M., Mower, M. M., Staewen, W. S., Tabatznik, B. & Mendeloff, A. I. Standby automatic defibrillator. An approach to prevention of sudden coronary death. Arch. Intern. Med. 126, 158–161 (1970).
Lown, B. & Axelrod, P. Implanted standby defibrillators. Circulation 46, 637–639 (1972).
Mirowski, M. et al. Termination of malignant ventricular arrhythmias with an implanted automatic defibrillator in human beings. N. Engl. J. Med. 303, 322–324 (1980).
Mond, H. G. & Proclemer, A. The 11th world survey of cardiac pacing and implantable cardioverter-defibrillators: calendar year 2009 — a World Society of Arrhythmia's project. Pacing Clin. Electrophysiol. 34, 1013–1027 (2011).
Bolli, R. Dandum semper est tempus: the crucial importance of (and increasing disregard for) the test of time. Circul. Res. 117, 755–757 (2015).
Plotnikov, A. N. et al. HCN212-channel biological pacemakers manifesting ventricular tachyarrhythmias are responsive to treatment with If blockade. Heart Rhythm 2, 282–288 (2008).
Shlapakova, I. N. et al. Biological pacemakers in canines exhibit positive chronotropic response to emotional arousal. Heart Rhythm 12, 1835–1840 (2010).
Boink, G. J. et al. HCN2/SkM1 gene transfer into canine left bundle branch induces stable, autonomically responsive biological pacing at physiological heart rates. J. Am. Coll. Cardiol. 61, 1192–1201 (2013).
Acknowledgements
The authors' work on biological pacemakers in funded by the US NIH RO1 HL135866 and NIH RO1 HL048509.
Author information
Authors and Affiliations
Contributions
All authors researched the data for the article, provided substantial contributions to discussions of its content, wrote the article, and undertook review and/or editing of the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Glossary
- Action potential
-
A rise and fall in the electrical potential of the surface membrane of the cell produced by the flow of ions across the cell membrane; pacemaker cells generate spontaneous action potentials that in turn can elicit action potentials in neighbouring cells.
- Diastolic depolarization
-
The spontaneous depolarization of the resting membrane potential during diastole; a unique property of pacemaker cardiomyocytes.
- Asynchronous pacing
-
Fixed rate pacing delivered by the implanted pacemaker independent of any atrial or ventricular activity.
- Synchronous pacing
-
Pacing delivered from the implanted pacemaker on demand if no atrial or ventricular activity is sensed.
- Sick sinus syndrome
-
A sinoatrial node disease that results in slow heart rhythms.
- Optical mapping
-
A laboratory technique that allows high-resolution mapping of the electrical activity of the heart.
- Syngeneic
-
Genetically similar or identical, and hence immunologically compatible.
- Heterokaryons
-
Multinucleate cells that contain genetically different nuclei.
- Electroanatomical mapping
-
A technique used both in the clinic and experimentally that allows mapping of the electrical activity of the heart in vivo.
- AV synchrony
-
The sequential contraction of the atria and the ventricles.
- Hydrops fetalis
-
A serious fetal condition defined as abnormal accumulation of fluid in two or more fetal compartments, including ascites, pleural effusion, pericardial effusion, and skin oedema.
Rights and permissions
About this article
Cite this article
Cingolani, E., Goldhaber, J. & Marbán, E. Next-generation pacemakers: from small devices to biological pacemakers. Nat Rev Cardiol 15, 139–150 (2018). https://doi.org/10.1038/nrcardio.2017.165
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrcardio.2017.165
This article is cited by
-
Harnessing cell reprogramming for cardiac biological pacing
Journal of Biomedical Science (2023)
-
Hydrogel interfaces for merging humans and machines
Nature Reviews Materials (2022)
-
The Essential Need for a Validated Potency Assay for Cell-Based Therapies in Cardiac Regenerative and Reparative Medicine. A Practical Approach to Test Development
Stem Cell Reviews and Reports (2021)
-
Sinoatrial node pacemaker cells share dominant biological properties with glutamatergic neurons
Protein & Cell (2021)
-
Sinoatrial node pacemaker cells: cardiomyocyte- or neuron-like cells?
Protein & Cell (2021)